
Exhibit 99.1

ANNUAL INFORMATION FORM
FOR THE YEAR ENDED DECEMBER 31, 2017
March 27, 2018
Contents
| I. | INTRODUCTION AND FORWARD-LOOKING STATEMENTS | 1 |
| II. | CORPORATE STRUCTURE | 3 |
| III. | THE COMPANY | 4 |
| IV. | Risk Factors and Uncertainties | 18 |
| V. | DIVIDENDS | 35 |
| VI. | DESCRIPTION OF CAPITAL STRUCTURE | 35 |
| VII. | Market For Securities | 36 |
| VIII. | Directors And Officers | 37 |
| IX. | AUDIT COMMITTEE | 41 |
| X. | Legal Proceedings and Regulatory Actions | 43 |
| XI. | Interest of Management and Others in Material Transactions | 44 |
| XII. | Transfer Agent and Registrar | 44 |
| XIII. | Material Contracts | 44 |
| XIV. | Interest of Experts | 44 |
| XV. | Additional Information | 45 |
| I. | INTRODUCTION AND FORWARD-LOOKING STATEMENTS |
The information contained in this Annual Information Form is stated as at December 31, 2017, unless otherwise indicated. If the context otherwise requires or unless otherwise indicated, “Aptose”, the “Company”, “we”, “us” and “our” refer collectively to Aptose Biosciences Inc., 5955 Airport Road Suite #228, Mississauga, Ontario, Canada, L4V 1R9, and to its subsidiaries, Aptose Biosciences U.S. Inc., Aptose Suisse GmbH and NuChem Pharmaceuticals Inc.
Unless otherwise indicated, all dollar amounts are expressed in US dollars and references to “$” are US dollars.
Certain statements in this Annual Information Form (“AIF”) may constitute “forward-looking” statements which involve known and unknown risks, uncertainties and other factors which may cause the actual results, performance or achievements of the Company, or industry results, to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. When used in this AIF, such statements use such words as “anticipate”, “contemplate”, “continue”, “believe”, “plan”, “estimate”, “expect”, “intend”, “will”, “should”, “may”, “hope” and other similar terminology. These statements reflect current expectations regarding future events and operating performance and speak only as of the date of this AIF. Forward-looking statements include, among others:
| · | our ability to obtain the substantial capital we require to fund research and operations; |
| · | our business strategy; |
| · | our clinical development plans; |
| · | our plans to secure and maintain strategic partnerships to assist in the further development of our product candidates and to build our pipeline; |
| · | our plans to conduct clinical trials and preclinical programs; |
| · | our ability to accrue appropriate numbers and types of patients; |
| · | our ability to file and maintain intellectual property to protect our pharmaceutical assets; |
| · | our reliance on external contract research/manufacturing organizations for certain activities; |
| · | potential exposure to legal actions and potential need to take action against other entities; |
| · | our expectations regarding the progress and the successful and timely completion of the various stages of our drug discovery, drug synthesis and formulation, preclinical and clinical studies and the regulatory approval process; |
| · | our plans, objectives, expectations and intentions; and |
| · | other statements including words such as “anticipate”, “contemplate”, “continue”, “believe”, “plan”, “estimate”, “expect”, “intend”, “will”, “should”, “may”, and other similar expressions. |
The forward-looking statements reflect our current views with respect to future events, are subject to significant risks and uncertainties, and are based upon a number of estimates and assumptions that, while considered reasonable by us, are inherently subject to significant business, economic, competitive, political and social uncertainties and contingencies. Many factors could cause our actual results, performance or achievements to be materially different from any future results, performance, or achievements that may be expressed or implied by such forward-looking statements, including, among others:
| 1 |
| · | our ability to obtain the substantial capital we require to fund research and operations; |
| · | our lack of product revenues and history of operating losses; |
| · | our early stage of development, particularly the inherent risks and uncertainties associated with (i) developing new drug candidates generally, (ii) demonstrating the safety and efficacy of these drug candidates in clinical studies in humans, and (iii) obtaining regulatory approval to commercialize these drug candidates; |
| · | our drug candidates require time-consuming and costly preclinical and clinical testing and regulatory approvals before commercialization; |
| · | clinical studies and regulatory approvals of our drug candidates are subject to delays, and may not be completed or granted on expected timetables, if at all, and such delays may increase our costs and could delay our ability to generate revenue; |
| · | the regulatory approval process; |
| · | our ability to recruit patients for clinical trials; |
| · | the progress of our clinical trials; |
| · | our ability to find and enter into agreements with potential partners; |
| · | our ability to attract and retain key personnel; |
| · | our ability to obtain and maintain patent protection; |
| · | our ability to protect our intellectual property rights and not infringe on the intellectual property rights of others; |
| · | our reliance on external contract research/manufacturing organizations for certain activities; |
| · | our ability to comply with applicable governmental regulations and standards; |
| · | development or commercialization of similar products by our competitors, many of which are more established and have or have access to greater financial resources than us; |
| · | commercialization limitations imposed by intellectual property rights owned or controlled by third parties; |
| · | our business is subject to potential product liability and other claims; |
| · | potential exposure to legal actions and potential need to take action against other entities; |
| · | our ability to maintain adequate insurance at acceptable costs; |
| · | further equity financing may substantially dilute the interests of our shareholders; |
| · | changing market conditions; and |
| 2 |
| · | other risks detailed from time-to-time in our on-going quarterly filings, annual information forms, annual reports and annual filings with Canadian securities regulators and the United States Securities and Exchange Commission (“SEC”), and those which are discussed under the heading “Risk Factors and Uncertainties” in this document. |
Should one or more of these risks or uncertainties materialize, or should the assumptions set out in the section entitled “ Risk Factors and Uncertainties” underlying those forward-looking statements prove incorrect, actual results may vary materially from those described herein. These forward-looking statements are made as of the date of this AIF, and we do not intend, and do not assume any obligation, to update these forward-looking statements, except as required by law. Such statements may not prove to be accurate as actual results and future events could differ materially from those anticipated in such statements. Investors are cautioned that forward-looking statements are not guarantees of future performance and accordingly investors are cautioned not to put undue reliance on forward-looking statements due to the inherent uncertainty therein. New factors emerge from time to time, and it is not possible for management of the Company to predict all of these factors or to assess in advance the impact of each such factor on the Company’s business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statement.
| II. | CORPORATE STRUCTURE |
Lorus Therapeutics Inc. (“Old Lorus”) was incorporated on September 5, 1986 under the name RML Medical Laboratories Inc. pursuant to the Business Corporations Act (Ontario). On October 28, 1991, RML Medical Laboratories Inc. amalgamated with Mint Gold Resources Ltd., resulting in Old Lorus becoming a reporting issuer (as defined under applicable securities law) in Ontario, on such date. On August 25, 1992, Old Lorus changed its name to IMUTEC Corporation. On November 27, 1996, Old Lorus changed its name to Imutec Pharma Inc., and on November 19, 1998, Old Lorus changed its name to Lorus Therapeutics Inc. On October 1, 2005, Old Lorus continued under the Canada Business Corporations Act.
On July 10, 2007 (the “Arrangement Date”), Old Lorus completed a plan of arrangement and corporate reorganization with, among others, 6650309 Canada Inc. (“New Lorus”), 6707157 Canada Inc. and Pinnacle International Lands, Inc. As a result of the plan of arrangement and reorganization, each common share of Old Lorus was exchanged for one common share of New Lorus and the assets (excluding certain deferred tax assets) and liabilities of Old Lorus (including all of the shares of its subsidiaries) were transferred, directly or indirectly, to New Lorus and/or its subsidiaries. New Lorus continued the business of Old Lorus after the Arrangement Date with the same officers and employees and continued to be governed by the same board of directors as Old Lorus prior to the Arrangement Date.
On August 28, 2014, New Lorus changed its name from Lorus Therapeutics Inc. to Aptose Biosciences Inc. and on October 1, 2014 we consolidated our outstanding common shares (the “Common Shares”) on the basis of one post-consolidation Common Share for each twelve pre-consolidation Common Shares.
The address of the Company’s head and registered office is 5955 Airport Road Suite #228, Mississauga, Ontario, Canada, L4V 1R9 and our phone number is (647) 479-9828. Our corporate website is www.aptose.com. The contents of the website and items accessible through the website are specifically not incorporated in this AIF by reference.
Aptose has three subsidiaries: Aptose Biosciences U.S. Inc. (“Aptose USA”), a company incorporated under the laws of Delaware, USA, Aptose Suisse GmbH (“Aptose Suisse”) a company incorporated under the laws of the canton of Zug, Switzerland and NuChem Pharmaceuticals Inc. (“NuChem”), a company incorporated under the laws of Ontario, Canada. Aptose owns 100% of the issued and outstanding voting share capital of Aptose USA and Aptose Suisse and 80% of the issued and outstanding voting share capital of NuChem.
| 3 |
| III. | THE COMPANY |
Aptose is a science-driven biotechnology company advancing highly differentiated agents to treat unmet medical needs in life-threatening cancers, such as acute myeloid leukemia (“AML”), high-risk myelodysplastic syndromes (“MDS”) and other hematologic malignancies. Based on insights into the genetic and epigenetic profiles of certain cancers and patient populations, Aptose is building a pipeline of novel and targeted oncology therapies directed at dysregulated processes and signaling pathways in cancer cells, and this strategy is intended to optimize efficacy and quality of life by minimizing the cytotoxic side effects associated with conventional therapies. Our product pipeline includes cancer drug candidates that exert potent activity as stand-alone agents and that enhance the activities of other anticancer agents without causing overlapping toxicities. Indeed, we believe our targeted products can emerge as first-in-class or best-in-class agents that deliver single agent benefit and may serve as part of a combination therapeutic strategy for specific populations of cancer patients.
We believe the future of cancer treatment and management lies in the prospective selection and treatment of patients having malignancies that are genetically or epigenetically predisposed to response based on a drug’s unique mechanism of action. We are of the view that many drugs currently approved for the treatment and management of cancer are not selective for the specific genetic alterations (targets) that cause the patient’s tumor and hence lead to significant toxicities due to off-target effects. Aptose’s strategy is to develop agents that target underlying disease-promoting mutations or altered pathways within a patient population, and we intend to apply this strategy across several therapeutic indications in oncology, including hematologic malignancies and solid tumor indications.
Aptose has one clinical-stage program, one late-preclinical program, and a third program that is discovery-stage and partnered with another company. CG026806 (“CG’806”), Aptose’s pan-FLT3 / pan-Bruton’s tyrosine kinase (“BTK”) inhibitor, is currently in late preclinical development and moving toward investigational new drug (“IND”) submission. Development of CG’806 is intended for the treatment of patients with relapsed / refractory AML and patients having certain B-cell malignancies. APTO-253 is Aptose’s second program and at the Phase 1b clinical stage for the treatment of patients with relapsed / refractory blood cancers, including AML and high-risk MDS under an IND allowed by the United States Food and Drug Administration (“FDA”) to evaluate APTO-253 as a therapeutic agent dosed on a weekly administration schedule for the treatment of certain hematologic malignancies. The APTO-253 program is currently on clinical hold and awaiting a new clinical batch of drug product to be manufactured and released.
Aptose’s leadership team comprises accomplished industry, financial and clinical research professionals who are dedicated to building a comprehensive anticancer drug pipeline and clinical development programs focused on targeted therapeutics directed against dysregulated oncogenic processes in patients with life-threatening hematologic malignancies.
The common shares of the Company are currently listed on the NASDAQ Capital Market (“NASDAQ”) under the symbol “APTO” and on the Toronto Stock Exchange (“TSX”) under the symbol “APS”.
Products
As noted above, Aptose is committed to the development of anticancer drugs that target aberrant oncologic signaling that underlie a particular life-threatening malignancy. This targeted approach is intended to impact the disease-causing events in cancer cells without affecting normal processes within cells. Such an approach requires that we first identify critical underlying oncogenic mechanisms in cancer cells and then develop a therapeutic that selectively impacts such oncogenic mechanisms. As a multi-kinase pan-FLT3 / BTK inhibitor, CG’806 targets multiple critical pathways that overlap to lead to the proliferation of cancer cells, including the B-cell receptor signaling pathway and FLT3 receptor pathways that converge at various points in the signaling cascade. Further, Aptose created the APTO-253 small molecule targeted drug that inhibits expression of the c-Myc oncogene and is under development as a novel therapy for AML and the related MDS.
| 4 |
The following table sets forth various product conditions in our pipeline and their respective stages of development.
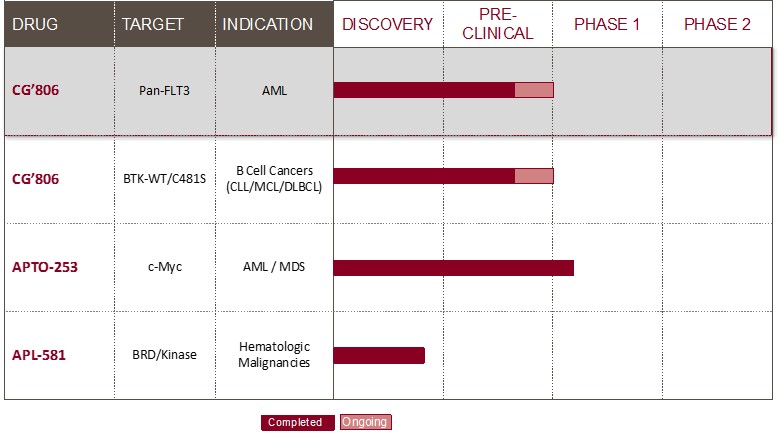
CG’806
Overview
In June 2016, we announced a definitive agreement with South Korean company CrystalGenomics, Inc. (“CG”), granting us an exclusive option to research, develop and commercialize CG’806 in all countries of the world except the Republic of Korea and China, for all fields of use. CG’806 is a highly potent, orally bioavailable non-covalent small molecule being developed for AML and certain B cell malignancies because of its actions as a pan-FLT3/pan-BTK inhibitor. We paid $1.0 million to CG to acquire the option. Should we elect to exercise the option, upon exercise, we would pay an additional $2.0 million in cash or combination of cash and common shares, and would receive full development and commercial rights for the program in all territories outside of the Republic of Korea and China. The option fee is due on the earlier of (i) filing of an IND application with the FDA, (ii) first dosage of a human in a clinical trial or (iii) or June 2018.
CG’806 exhibits a picomolar IC50 toward the FMS-like tyrosine kinase 3 (FLT3) with the Internal Tandem Duplication (“FLT3-ITD”), potency against the wild type FLT3 and a host of mutant forms of FLT3, as well as single-digit nanomolar IC50’s against BTK and its C481S mutant (“BTK-C481S”). Consequently, CG’806 is characterized as a pan-FLT3/pan-BTK inhibitor. Further, CG’806 suppresses a small group of other relevant oncogenic kinases/pathways (including CSF1R, Aurora kinases (“AURK”), TRK, and the AKT and ERK pathways) that are operative in AML and certain B cell malignancies, but does not inhibit the TEC, EGFR and ErbB2/4 kinases that are responsible for safety concerns with certain other kinase inhibitors.
| 5 |
As a potent inhibitor of FLT3-ITD, CG’806 may become an effective therapy in a high-risk subset of AML patients. This is because the FLT3-ITD mutation occurs in approximately 30% of patients with AML and is associated with a poor prognosis. In murine xenograft studies of human AML (FLT3-ITD), CG’806 administered orally once daily for 14 days resulted in tumor elimination without measurable toxicity. Importantly, CG’806 targets other oncogenic kinases which may also be operative in FLT3-ITD AML, thereby potentially allowing the agent to become an important therapeutic option for a broader group of this difficult-to-treat AML patient population. The findings that CG’806 targets all forms of FLT3 and several other key oncogenic pathways, and that CG’806 was well tolerated from a safety perspective during efficacy studies, suggest that CG’806 may also have applicability in treating patients, particularly those over the age of 65, who cannot tolerate other therapies.
Separate from the AML and FLT3 story, overexpression of the BTK enzyme can drive oncogenic signaling of certain B cell malignancies, such as chronic lymphocytic leukemia (“CLL”), mantle cell lymphoma (“MCL”), diffuse large cell B cell lymphoma (“DLBCL”) and others. Therapy of these patients with covalent, irreversible BTK inhibitors, such as ibrutinib, that target the active site Cysteine (“Cys”) residue of BTK can be beneficial in many patients. However, therapy with covalent BTK inhibitors can select for BTK with a C481S mutation, thereby conferring resistance to covalent BTK inhibitors. Furthermore, approximately half of CLL patients have discontinued treatment with ibrutinib after 3.4 years of therapy. Discontinuation of ibrutinib is due to the development of drug resistance (in particular, patients have malignancies that developed the BTK-C481S mutation), or due to refractory disease (patient tumors did not respond to ibrutinib) or intolerance (side effects led to discontinuation of ibrutinib), according to a study performed at The Ohio State University. The C481S mutation is observed in 5-10% of the patients, while 40-45% of the patients were intolerant or refractory to ibrutinib. As a non-covalent, reversible inhibitor of BTK, CG’806 does not rely on the Cysteine 481 residue (“C481”) for inhibition of the BTK enzyme. Indeed, recent X-ray crystallographic studies (with wild type and C481S BTK) demonstrated that CG’806 binds productively to the BTK active site in a manner that is indifferent to the presence or absence of mutations at the 481 residue. Moreover, in vitro studies demonstrated that CG’806 kills B cell malignancy cell lines on average approximately 1500 times more potently than ibrutinib, and CG’806 demonstrated a high degree of safely in animal efficacy studies. Consequently, patients who are resistant, refractory or intolerant to ibrutinib or other commercially approved or development-stage BTK inhibitors with B cell malignancies may continue to be sensitive to CG’806 therapy. This is particularly true since CG’806 inhibits the wild type and mutant forms of BTK, as well as other kinases/pathways that drive the survival and proliferation of B cell malignancies
Role of BTK in B-cell signaling
BTK, a member of the TEC family kinase, is an essential element of B-cell receptor (“BCR”) signaling, which is required for B-cell maturation, survival and proliferation. It is an upstream activator of multiple pro-survival / anti-apoptotic pathways, including the NF-KB, mTOR-AKT and ERK pathways. BTK is overexpressed in malignant cells from patients with various B-cell malignancies, such as CLL, MCL, AML, and DLBCL. Disruption of BCR signaling via inhibition of BTK, has been shown to lead to clinical remissions in these patients.
| 6 |
CG’806 as a Non-covalent, Reversible Kinase Inhibitor
Binding studies of CG’806 have confirmed non-covalent, reversible inhibition of BTK, FLT3-ITD and Aurora Kinase A. Commercially-approved, covalent BTK inhibitors possess a Michael acceptor to react with C481 in BTK and irreversibly inactivate the BTK enzyme. In contrast, CG’806 does not require reactivity with the C481 residue for inhibition of the BTK enzyme, thereby allowing CG’806 to inhibit the wild type and C481 mutant form of the BTK enzyme.
Preclinical In Vitro Evaluation of CG’806
CG’806 is a potent inhibitor of BTK and FLT3 wild types, as well as the BTK C481S and FLT3-ITD mutants, which represent major sources of therapy relapse or are negative prognostic signals in patients. In enzymatic assays, CG’806 has demonstrated potency against the BTK C481S mutant with an IC50 of 2.5 nM. CG’806 also has potent activity against the FLT-ITD mutation, occurring in 30-35% of AML patients, with an IC50 against the purified enzyme of 0.8 nM (800pM). Likewise, CG’806 exerts low nM IC50 values against the FLT3 enzyme having various mutations in the tyrosine kinase domain (TKD) and the Gatekeeper region. Similarly, CG’806 demonstrated picomolar potency against Aurora A (IC50 0.4 nM). Notably, CG’806 is a potent inhibitor of interleukin-2-inducible T-cell kinase (“ITK”), at approximately 4 nM. ITK is speculated to play a role in suppressing activated T-cell function, hence inhibition of ITK alleviates this suppression, and provides for a potential immunomodulatory anti-tumor mechanism. Finally, CG’806 does not exhibit any inhibition of epidermal growth factor receptor (“EGFR”), TEC or ErbB2/4 kinases. Inhibition of one or more of these kinases has been speculated to contribute to the toxicity observed from the commercially approved BTK inhibitor.
BTK is overexpressed in the blast cells of approximately 80% of AML patients as compared to normal peripheral blood mononuclear cells (PBMCs) in healthy subjects. Researchers have shown that BTK inhibition attenuates the proliferation and survival of FLT3-ITD primary AML blasts and AML cell lines, as well as inhibits the downstream activation of FLT3-ITD-dependent Myc and STAT5 kinases. We believe that CG’806 is the only drug in development that inhibits both FLT3-ITD and BTK pathways reported to synergize to drive the proliferation and survival of AML.
CG’806 Xenograft Studies
In vivo subcutaneous AML tumor models of anti-cancer efficacy revealed CG’806 induced rapid and sustained tumor eradication (Figure 1). CG’806 was administered orally once daily, for 14 days. Moreover, CG’806 exhibited the sustained tumor elimination post therapy, while demonstrating no impact to murine body weight, no impacts to hematology cell counts or visible organ toxicities – necropsy and clinical pathology findings did not reveal any abnormal observations. A maximum tolerated dose has not yet been identified with murine xenograft studies, having been performed up to 450 mg/kg orally for 14 days (CG preliminary toxicity data).
| 7 |
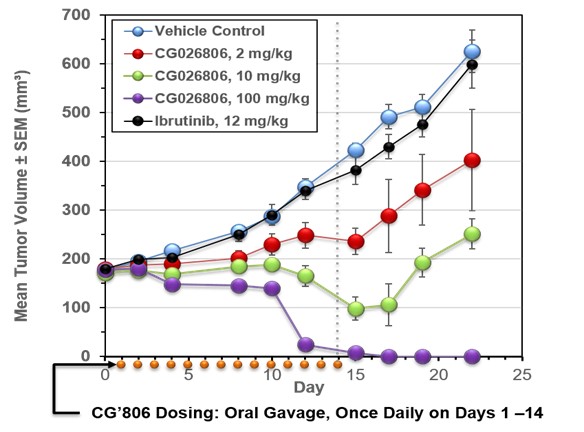
Figure 1. Efficacy of CG’806 in MV4-11 xenograft model.
MV4-11 tumor bearing mice were administered an oral suspension once daily for 14 days of CG’806 at 2 mg/kg (blue line), 10 mg/kg (green line) or 100 mg/kg (red line), Ibrutinib, 12 mg/kg (turquoise line), or vehicle (Control; black line) with 7-day post-treatment follow-up. Tumor volumes and body weights were measured 3 times weekly.
CG’806 Intellectual Property
A Patent Cooperation Treaty (PCT) application providing composition of matter and use protection for CG’806 was filed in late 2013, with a potential expiry in 2033 before extension opportunities, across all major geographies.
CG’806 Manufacturing and Preclinical Progress
We have invested significant time, effort and capital to create a scalable chemical synthetic route for the manufacture of CG’806 drug substance, to develop an oral formulation for clinical development, and to study the actions of CG’806 in various preclinical biological pathway studies. Our efforts to develop the scalable chemical synthetic route have taken longer than anticipated and thus pushed the timeline for the IND submission and initiation of the first-in-human Phase I clinical trial further into the future than we had originally anticipated. We now have solved the synthetic route, can scale the manufacture of API, and now have manufactured and delivered a batch of API which was used for Dose Range Finding Studies that were performed and completed in early January, 2018. Currently we are manufacturing a multi-kg batch of GLP grade API (drug substance) for use in GLP toxicology studies. We also reported that we selected the oral formulation that we intend to take into the GLP toxicology studies and the first-in-human clinical trials. In addition, R&D funds are being utilized to support exploratory formulation studies in an ongoing effort to craft superior formulations for CG’806. Provided we are able to manufacture CG’806 for both the non-clinical (GLP) studies and clinical trial, complete the non-clinical studies, and receive a favorable approval from the FDA on our IND submission and continue on the anticipated timeline, we expect to initiate a first-in-human Phase I clinical trial by late 2018. The total direct costs of such activities and to reach the submission of the IND are currently expected to range between US$3 million and US$4.5 million. However any interruptions or additional studies in these activities could cause a delay in the anticipated commencement of the Phase I trial. Greater granularity on the timing of the IND submission and clinical trial will be provided in the coming months. CG’806 is being developed with the intent to deliver the agent as an oral therapeutic and to develop it in parallel for AML and for appropriate B cell malignancies (likely CLL). As clinical trials are lengthy, complex, costly, and uncertain processes, an estimate of the future costs is not reasonable at this time.
| 8 |
Developments in 2017
On January 23, 2017, we announced that we would prioritize our resources toward the development of CG’806 and temporarily delay clinical activities with APTO-253, in order to elucidate the cause of recent manufacturing setbacks related to the intravenous formulation of APTO-253, with the intention of restoring the molecule to a state supporting clinical development and partnering. Although we have two compelling cancer drugs, resources could support the full development activities of only one at this time. Recent advances with CG’806 elevated this agent as having the best risk-reward profile to pursue with those resources. Such data established CG’806 as a well-differentiated pan-FLT3 inhibitor that demonstrates tumor eradication in the absence of toxicity in AML xenograft models, and it is on track for development as a therapy for certain AML patients. In addition, CG’806 is a potent non-covalent inhibitor of proliferation among certain BTK-driven B-cell derived cancer cells. The encouraging properties of CG’806, including its potency against well-established targets in diseases of severe medical need, warrant expeditious advancement and prioritization of resources toward this molecule.
On May 7, 2017, we presented preclinical data for our pan-FLT3/pan-BTK inhibitor CG’806 at the 2017 American Association for Cancer Research (AACR) Conference for Hematologic Malignancies: Translating Discoveries to Novel Therapies in Boston, MA. Two separate presentations highlighting CG’806 were presented. In one presentation, our scientists, with researchers from the Knight Cancer Institute at Oregon Health & Science University (OHSU), presented data relating to the potency of CG’806 against samples derived from patients with various hematologic malignancies. In a separate presentation, our scientists, with researchers from the MD Anderson Cancer Center, presented data demonstrating CG’806’s potent activity against AML cells harboring wild type or specific mutant forms of FLT3.
In September 2017 the USPTO informed us that the patent has been awarded. The patent claims numerous compounds, including the CG’806 compound, pharmaceutical compositions comprising the CG’806 compound, and methods of treating various diseases caused by abnormal or uncontrolled activation of protein kinases.
On December 11, 2017 at the American Society of Hematology Annual Meeting, we presented with the OHSU Knight Cancer Institute preclinical data demonstrating that CG’806, a pan-FLT3/pan-BTK inhibitor, has broad and potent drug activity against AML, CLL and other hematologic disease subtypes. We also announced the presentation of preclinical data from research led by The University of Texas MD Anderson Cancer Center demonstrating that CG’806 exerts a profound anti-leukemia effect in human and murine leukemia cell lines harboring FLT-3 ITD mutations, mutations that are usually associated with very poor prognoses in leukemia patients. In addition, CG’806 induces apoptosis, or programmed cell death, in AML patient samples by multiple mechanisms and is able to overcome resistance that is seen with other FLT3 inhibitors. The data were highlighted in poster presentations on December 10 and 11, 2017 at the American Society of Hematology Annual Meeting.
| 9 |
On December 26, 2017, we announced that the FDA has granted orphan drug designation to CG’806 for the treatment of patients with AML. Orphan drug designation is granted by the FDA to encourage companies to develop therapies for the treatment of diseases that affect fewer than 200,000 individuals in the United States. Orphan drug status provides research and development tax credits, an opportunity to obtain grant funding, exemption from FDA application fees and other benefits. If CG’806 is approved to treat AML, the orphan drug designation provides Aptose with seven years of marketing exclusivity.
On March 15, 2018, we announced two abstracts related to the mechanistic properties of CG’806 in AML cells and in B cell malignancy cells have been accepted for poster presentations at the upcoming 2018 Annual Meeting of the American Association for Cancer Research (AACR).
APTO-253
Overview
APTO-253, the Company’s second program, is a novel small molecule therapeutic agent that inhibits expression of the c-Myc oncogene, leading to cell cycle arrest and programmed cell death (apoptosis) in human-derived solid tumor and hematologic cancer cells, without causing general myelosuppression of the healthy bone marrow. The c-Myc oncogene is overexpressed in hematologic cancers, including AML. C-Myc is a transcription factor that regulates cell growth, proliferation, differentiation and apoptosis, and overexpression amplifies new sets of genes to promote oncogenesis. APTO-253 dramatically down-regulates expression of the c-Myc oncogene in AML cells and depletes those cells of the c-Myc oncoprotein, leading to apoptotic cell death in AML cells. Thus APTO-253 may serve as safe and effective c-Myc inhibitor for AML that combines well with other agents and does not impact the normal bone marrow.
APTO-253 was being evaluated by us in a Phase Ib clinical trial in patients with relapsed / refractory hematologic malignancies, particularly AML and MDS before being placed on clinical hold by the FDA in November 2015. If and when the APTO-253 clinical trial is re-initiated, upon completion of the dose-escalation stage of the study and determination of the appropriate dose, the plan would be to enroll additional AML patients for a disease-specific single-agent expansion cohorts. For future development, upon selection of a lead hematologic indication from this Phase Ib study, combination of APTO-253 with a standard therapy would be considered.
As previously disclosed, the Phase Ib trial was placed on clinical hold in order to solve a chemistry-based formulation issue, and the chemistry of the API and the formulation had undergone minor modifications to deliver a stable and soluble drug product for return to the clinical setting. In December 2016, we announced that we had successfully manufactured multiple non-GMP batches of a new drug product formulation for APTO-253, including a batch that had been stable and soluble for over six months. However, the 40L batch that was the intended clinical supply encountered an unanticipated mishap during the filling process that compromised the stability of that batch of drug product. On January 23, 2017, we announced that the root cause and corrective action studies would take longer than originally expected and that we would temporarily delay clinical activities with APTO-253 in order to elucidate the cause of manufacturing setback, with the intention of restoring the molecule to a state supporting clinical development and partnering. Formal root cause analyses studies have now been completed and have identified the reason for the drug product stability failure, and we have established a corrective and prevention action plan for the manufacture of future batches of drug product. Given these findings, we have manufactured a new GMP clinical supply of drug product and are in the process of performing studies required to demonstrate the fitness of the drug product for clinical usage, and then we plan to present the findings to the FDA in the second quarter of 2018 with the hope of having the clinical hold removed by the end of the second quarter of 2018 and returning APTO-253 to the clinical trial soon thereafter. The total direct costs of such activities to reach the presentation of the findings to the FDA are currently expected to range between US$1 million and US$1.5 million. Investors are cautioned that there can be no assurance that the FDA will remove the clinical hold.
| 10 |
In the event the clinical hold is removed by the FDA, based on our current estimates and the information available to us at this time, we expect to complete the clinical drug product manufacture, initiate studies to investigate additional drug delivery methods for APTO-253 and to initiate additional non-clinical studies for solid tumor and hematologic development. As preparing, submitting, and advancing applications for regulatory approval, developing drugs and drug product and clinical trials are sometimes complex, costly, and time consuming processes, an estimate of the future costs is not reasonable at this time.
Solid Tumors
In January 2011, Aptose announced the first patient enrolment in a Phase I dose-escalation study for APTO-253 in patients with advanced or metastatic solid tumors who are unresponsive to conventional therapy or for which no effective therapy is available. The study was initially being conducted at Memorial Sloan-Kettering Cancer Center in New York. Objectives of the study included determination or characterization of the safety profile, maximum tolerated dose, and antitumor activity of APTO-253, as well as pharmacokinetics and a recommended Phase II dose for subsequent clinical trials.
In June 2012, MD Anderson Cancer Center in Houston was added as a second site under the direction of Dr. Jennifer Wheler as the principal investigator. In addition, Aptose announced that the study had successfully completed the accelerated drug dose escalation stage (Stage 1), with further escalation under way in the non-accelerated dose escalation stage (Stage 2) for the purpose of determining the maximal tolerated dose level and recommended Phase II dose. The addition of a second site expanded patient availability for enrollment.
In January 2013, Aptose announced that Phase I clinical study of APTO-253 has successfully escalated to the target dose level based on predicted and observed clinical effects without limitation by toxicity. The success of this study allowed Aptose to initiate a biomarker clinical investigation to further explore the effects of the drug at relevant doses determined in the clinical trial.
In April 2013, Aptose announced that studies demonstrated the antitumor activity of APTO-253 in animal models of human NSCLC with a dose-response effect in NSCLC.
In July 2013, Aptose announced the results of the Phase 1 clinical trial of APTO-253. In this first-in-man dose-escalation clinical study, APTO-253 demonstrated a favorable safety profile, as well as encouraging signs of antitumor activity. The design of this trial consisted of APTO-253 as a single agent in patients with advanced solid tumors resistant to multiple standard therapies. The study enrolled 27 patients, all of which had failed a median of four prior chemotherapies. Although this was primarily a dose-escalation safety study, efficacy and pharmacokinetics were also explored.
The clinical trial enrolled patients at seven dose levels ranging from 20 to 229 mg/m2. Of the 27 patients enrolled, 17 were evaluable for efficacy. Of these 17 patients, seven (41%) achieved stable disease by Response Evaluation Criteria In Solid Tumors (“RECIST”). This included patients with colorectal, lung, appendiceal, liver and uterine cancers. Dose related activity was demonstrated at the higher dose levels (176 and 229 mg/m2). At these two highest dose levels, four of five evaluable patients (80%) achieved sustained stable disease by RECIST ranging from 5.6 months to 8 months, representative of disease control. Of these, a patient with non-small cell lung cancer at the highest dose level additionally demonstrated non-index tumor shrinkage.
| 11 |
The safety assessment indicated that APTO-253 was well tolerated at all dose levels tested in this trial. The dose escalation was not limited by toxicity. The most common adverse event was Grade 1 or 2 fatigue seen in three patients. There was one Grade 3 toxicity, asymptomatic low blood phosphate level that was reversible by supplementation with phosphates. The pharmacokinetic profile was consistent with the predictive profile seen preclinically, and the elimination profile and half-life in patients were suggestive of a very rapid distribution phase and prolonged retention.
Multi-Targeting Bromodomain Program
In November 2015, Aptose entered into a definitive agreement with Moffitt Cancer Center (“Moffitt”) for exclusive global rights to potent, multi-targeting, single-agent inhibitors for the treatment of hematologic and solid tumor cancers. These small molecule agents are inhibitors of the Bromodomain and Extra-Terminal motif (“BET”) protein family members, which simultaneously target specific kinase enzymes. The molecules developed by Moffitt exhibited potency against the BET family members and specific oncogenic kinases which, when inhibited, are synergistic with BET inhibition. Under the agreement, Aptose would gain access to the drug candidates developed by Moffitt and the underlying intellectual property covering the chemical modifications enabling potent bromodomain (“BRD”) inhibition on the chemical backbone of a kinase inhibitor.
In January 2017, Aptose terminated the collaboration with Moffitt for the development of the dual BRD4 / JAK2 inhibitor program.
Multi-Targeting Epigenetic Program
In November 2015, Aptose announced an exclusive drug discovery partnership with Laxai Avanti Life Sciences (“LALS”) for their expertise in next generation epigenetic-based therapies. Under the agreement, LALS was to be responsible for developing multiple clinical candidates, including optimizing candidates that exert dual BRD4 / kinase inhibitory activity. Based on available resources, Aptose halted further investment in the collaboration with LALS in late 2016. However, the program delivered novel intellectual property and hit molecules for further optimization. As a consequence, Aptose may choose to out-license the program.
On March 7, 2018, we entered into an exclusive global license agreement with Ohm Oncology (“OHM”), an affiliate of LALS that was formed in 2016 to advance the clinical development of compelling molecules derived from the LALS initiative, for the development, manufacture and commercialization of APL-581, as well as related molecules from Aptose’s dual bromodomain and extra-terminal domain motif (BET) protein and kinase inhibitor program. Under the agreement, Aptose will retain reacquisition rights to certain molecules, while OHM/LALS will have the rights to develop and sublicense all other molecules. Aptose will receive a nominal upfront cash payment and is eligible to receive up to $125 million of additional payments based on the achievement of certain development, regulatory and sales milestones, as well as significant royalties on future sales generated from the program, if any.
Clinical Indications for Aptose Programs
Acute Myeloid Leukemia
AML is a rapidly progressing cancer of the blood and bone marrow characterized by the uncontrolled proliferation of dysfunctional myeloblasts that do not mature into healthy blood cells. It is the most common form of acute leukemia in adults. The American Cancer Society estimates [there will be] approximately 19,520 new cases of AML and approximately 10,670 deaths from AML in the United States in 2018. [Standard induction therapy with chemotherapy is successful in many AML patients, but the majority of these patients will relapse with treatment refractory disease. The average age of a patient with AML is 67 years. Approximately 48% patients less than age 60, and 34% of patients greater than or equal to age 60, with residual disease after induction therapy will achieve a remission, as reported by Datamonitor Healthcare.
| 12 |
Myelodysplastic Syndromes
MDS are a group of blood and bone marrow disorders. In MDS, stem cells do not mature normally, and the number of blasts (immature cells) and dysplastic (abnormally developed) cells increases. Also, the number of healthy mature cells decreases, meaning there are fewer normal red blood cells, white blood cells, and platelets. The numbers of blood cells are often called blood cell counts. Because of the decrease in healthy cells, people with MDS often have anemia (a lowered blood cell count), and may have neutropenia (a low white blood cell count) and thrombocytopenia (a low platelet count). Also, the chromosomes (long strands of genes) in the bone marrow cells may be abnormal. According to the American Cancer Society, there are approximately 13,000 new cases of MDS annually in the United States. Additionally, Datamonitor Healthcare reports median survival in higher risk MDS patients may range between five months and two years. There are several subtypes of MDS, and some subtypes of MDS may eventually turn into AML.
Specialized Skill and Knowledge
The business of the Company requires personnel with specialized skills and knowledge in oncology. Researchers must be able to design and implement studies to assess the efficacy of anticancer drugs. Specialized knowledge and skills relating to chemistry and formulation process development are also needed. Such knowledge and skills are needed to develop product specific analytical methods and formulation processes. The Company’s business also requires clinical and regulatory expertise and knowledge. The Company has trained scientists and personnel with broad experience in these fields.
Competitive Conditions
The biotechnology and pharmaceutical industries are characterized by rapidly evolving technology and intense competition. There are numerous companies in these industries that are focusing their efforts on activities similar to ours. Some of these are companies with established positions in the pharmaceutical industry and may have substantially more financial and technical resources, more extensive research and development capabilities, and greater marketing, distribution, production and human resources than Aptose. In addition, we face competition from other companies for opportunities to enter into partnerships with biotechnology and pharmaceutical companies and academic institutions.
Competition with our potential products may include chemotherapeutic agents, monoclonal antibodies, antisense therapies, small molecules, immunotherapies, vaccines and other biologics with novel mechanisms of action. These drugs may kill cancer cells indiscriminately, or through a targeted approach, and some have the potential to be used in non-cancer indications. We also expect that we will experience competition from established and emerging pharmaceutical and biotechnology companies that have other forms of treatment for the cancers that we target, including drugs currently in development for the treatment of cancer that employ a number of novel approaches for attacking these cancer targets. Cancer is a complex disease with more than 100 indications requiring drugs for treatment. The drugs in competition with our potential drugs have specific targets for attacking the disease, targets which are not necessarily the same as ours. These competitive drugs, however, could potentially also be used together in combination therapies with our drugs to manage the disease. Other factors that could render our potential products less competitive may include the stage of development, where competitors’ products may achieve earlier commercialization, as well as superior patent protection, better safety profiles, or a preferred cost-benefit profile.
| 13 |
Components
Standard raw materials, component parts, and products required by the Company in pursuing its activities are supplied from reputable companies active in the biotechnology industry. Pricing is predictable as there are many alternatives of such supplies that are readily available.
Intangible Properties
We believe that our issued patents and pending applications are important in establishing and maintaining a competitive position with respect to our products and technology.
As of March 27, 2018, we are the owner of record of 6 issued U.S. patents, which together provide coverage for the APTO-253 compound, it’s pharmaceutical composition and methods of treating various cancers with APTO-253, including solid tumors and leukemia. The APTO-253 ccomposition of matter patent expires in 2028 in the United States and 2026 in other countries. We also hold 17 international (non-U.S.) patents which together provide coverage for APTO-253, three of which are issued European patents, validated in at least eight countries in Europe. Our patents also include several compounds that are similar to APTO-253, which provide protection from competitors seeking to develop anticancer products that are related in chemical structure to APTO-253.
On September 12, 2017, we announced that United States Patent and Trademark Office (“USPTO”) has issued a patent (number 9,758,508) entitled “2,3-dihydro-isoindole-1-on derivative as BTK kinase suppressant, and pharmaceutical composition including same”. The patent claims numerous compounds, including the CG’806 compound, pharmaceutical compositions comprising the CG’806 compound, and methods of treating various diseases. The patent is expected to provide protection until December of 2033.
Environmental Protection
The Company’s research and development activities involve the controlled use of hazardous and radioactive materials and, accordingly, the Company is subject to federal, provincial and local laws and regulations governing the use, manufacture, storage, handling and disposal of such materials and certain waste products. To the knowledge of the Company, compliance with such environmental laws and regulations does not and will not have any significant impact on its capital spending, profits or competitive position within the normal course of its operating activities. There can be no assurance, however, that the Company will not be required to incur significant costs to comply with environmental laws and regulations in the future or that its operations, business or assets will not be materially adversely affected by current or future environmental laws or regulations.
Employees
As at December 31, 2017, we employed 18 full-time persons and 2 part-time persons in research and drug development and administration activities. Four of our employees hold Ph.D.’s and numerous others hold degrees and designations such as MSc, BSc, CPA (CA), CPA (California) and MBA. To encourage a focus on achieving long-term performance, employees and members of the board of directors of the Company (the “Board”) have the ability to acquire an ownership interest in the Company through Aptose’s share option and alternate compensation plans.
| 14 |
None of our employees are unionized, and we consider our relations with our employees to be good.
Government Regulation
Overview
Our overall regulatory strategy is to work with the appropriate government departments which regulate the use and sale of therapeutic drug products. This includes Health Canada in Canada, the FDA in the United States, the European Medicines Agency in Europe, and other local regulatory agencies with oversight of preclinical studies, clinical trials and marketing of therapeutic products. Where possible, we intend to take advantage of opportunities for accelerated development of drugs designed to treat rare and serious or life-threatening diseases. We also intend to pursue priority evaluation of any application for marketing approval filed in Canada, the United States or the European Union and to file additional drug applications in other markets where commercial opportunities exist. We may not be able to pursue these opportunities successfully.
Regulation(s) by government authorities in Canada, the United States, and the European Union are significant factors in guiding our current research and drug development activities. To clinically test, manufacture and market drug products for therapeutic use, we must be in compliance with guidance and regulations established by the regulatory agencies in the countries in which we currently operate or intend to operate.
The laws of most of these countries require the licensing of manufacturing facilities, carefully controlled research and the extensive testing of products. Biotechnology companies must establish the safety and efficacy of their new products in clinical trials; they must establish and comply with current GMP(s) for the manufacturing of the product and control over marketing activities before being allowed to market a product. The safety and efficacy of a new drug must be shown through human clinical trials of the drug carried out in accordance with the guidance and regulations established by local and federal regulatory agencies.
The process of completing clinical trials and obtaining regulatory approval for a new drug takes a number of years and requires the expenditure of substantial resources. Once a new drug or product license application is submitted, regulatory agencies may not review the application in a timely manner and may not approve the product. Even after a New Drug Application (“NDA”) submission has occurred and/or approval has been obtained, further studies, including post-marketing studies, may be required to provide additional data on efficacy and safety necessary to confirm the approved indication or to gain approval for the use of the new drug as a treatment for clinical indications other than those for which the new drug was initially tested. Also, regulatory agencies require post-marketing surveillance programs to monitor a new drug’s side effects, safety and long term effects of the product. A serious safety or effectiveness problem involving an approved new drug may result in a regulatory agency mandating a withdrawal of the new drug from the market and possible civil action. It is possible that we could encounter such difficulties or excessive costs in our efforts to secure necessary approvals, which could delay or prevent us from manufacturing or marketing our products.
In addition to the regulatory product approval framework, biotechnology companies, including Aptose, are subject to regulation under local, provincial, state and federal law, including requirements regarding occupational safety, laboratory practices, environmental protection and hazardous substance control, and may be subject to other present and future local, provincial, state, federal and foreign regulation, including possible future regulation of the biotechnology industry.
| 15 |
Regulation in Canada
In Canada, the manufacture and sale of new drugs are controlled by Health Canada. New drugs must pass through a number of testing stages, including pre-clinical testing and human clinical trials. Pre-clinical testing involves testing the new drug’s chemistry, pharmacology and toxicology in vitro and in vivo. Successful results (that is, potentially valuable pharmacological activity combined with an acceptable low level of toxicity) enable the developer of the new drug to file a clinical trial application to begin clinical trials involving humans.
To study a drug in Canadian patients, a clinical trial application submission must be filed with Health Canada. The clinical trial application submission must contain specified information, including the results of the pre-clinical tests completed at the time of the submission and any available information regarding use of the drug in humans. In addition, since the method of manufacture may affect the efficacy and safety of a new drug, information on manufacturing methods and standards and the stability of the drug substance and dosage form must be presented. Production methods and quality control procedures must be in place to ensure an acceptably pure product, essentially free of contamination, and to ensure uniformity with respect to all quality aspects.
In addition, all federally regulated trials must be approved and monitored by an independent committee of doctors, scientists, advocates and others to ensure safety and ethical standards. These committees are called Institutional Review Boards (“IRBs”) or Ethics Review Boards (“ERBs”). The review boards study and approve all study-related documents before a clinical trial begins and also carefully monitor data to detect benefit or harm, and validity of results.
Provided Health Canada does not reject a clinical trial application submission and IRB or ERB approval has been obtained, clinical trials can begin. Clinical trials for product candidates in Canada, as in the United States, are generally carried out in three phases. Phase I involves studies to evaluate toxicity and ideal dose levels in healthy humans. The new drug is administered to human patients who have met the clinical trial entry criteria to determine pharmacokinetics, human tolerance and prevalence of any adverse side effects. Phases II and III involve therapeutic studies. In Phase II, efficacy, dosage, side effects and safety are established in a small number of patients who have the disease or disorder that the new drug is intended to treat. In Phase III, there are controlled clinical trials in which the new drug is administered to a large number of patients who are likely to receive benefit from the new drug. In Phase III, the effectiveness of the new drug in patients is compared to that of standard accepted methods of treatment in order to provide sufficient data for the statistical proof of safety and efficacy for the new drug.
If clinical studies establish that a new drug has value, the manufacturer submits a new drug submission application to Health Canada for marketing approval. The new drug submission contains all information known about the new drug, including the results of pre-clinical testing and clinical trials. Information about a substance contained in new drug submission includes its proper name, its chemical name, and details on its method of manufacturing and purification, and its biological, pharmacological and toxicological properties. The new drug submission also provides information about the dosage form of the new drug, including a quantitative listing of all ingredients used in its formulation, its method of manufacture, manufacturing facility information, packaging and labelling, the results of stability tests, and its diagnostic or therapeutic claims and side effects, as well as details of the clinical trials to support the safety and efficacy of the new drug. Furthermore, for biological products, an on-site evaluation is completed to assess the production process and manufacturing facility. It is required prior to the issuance of a notice of compliance. All aspects of the new drug submission are critically reviewed by Health Canada. If a new drug submission is found satisfactory, a notice of compliance is issued permitting the new drug to be sold for the approved use. In Canada, an establishment license must be obtained prior to marketing the product.
| 16 |
Health Canada has a policy of priority evaluation of new drug submissions for all drugs intended for serious or life-threatening diseases for which no drug product has received regulatory approval in Canada and for which there is reasonable scientific evidence to indicate that the proposed new drug is safe and may provide effective treatment.
The monitoring of a new drug does not cease once it is on the market. For example, a manufacturer of a new drug must report any new information received concerning serious side effects, as well as the failure of the new drug to produce desired effects. If Health Canada determines it to be in the interest of public health, a notice of compliance for a new drug may be suspended and the new drug may be removed from the market.
A post surveillance program involves clinical trials conducted after a drug is marketed (referred to as Phase IV studies in the United States) and is an important source of information on as yet undetected adverse outcomes, especially in populations that may not have been involved in the premarketing trials (e.g., children, the elderly, pregnant women) and the drug’s long-term morbidity and mortality profile. Regulatory authorities may require companies to conduct Phase IV studies as a condition of market approval. Companies often conduct post-marketing studies in the absence of a regulatory mandate.
An exception to the foregoing requirements relating to the manufacture and sale of a new drug is the limited authorization that may be available in respect of the sale of new drugs for emergency treatment. Under the special access program, Health Canada may authorize the sale of a quantity of a new drug for human use to a specific practitioner for the emergency treatment of a patient under the practitioner’s care. Prior to authorization, the practitioner must supply Health Canada with information concerning the medical emergency for which the new drug is required, such data as is in the possession of the practitioner with respect to the use, safety and efficacy of the new drug, the names of the institutions at which the new drug is to be used and such other information as may be requested by Health Canada. In addition, the practitioner must agree to report to both the drug manufacturer and Health Canada the results of the new drug’s use in the medical emergency, including information concerning adverse reactions, and must account to Health Canada for all quantities of the new drug made available.
The Canadian regulatory approval requirements for new drugs outlined above are similar to those of other major pharmaceutical markets. While the testing carried out in Canada is often acceptable for the purposes of regulatory submissions in other countries, individual regulatory authorities may request supplementary testing during their assessment of any submission. Therefore, the clinical testing conducted under Health Canada authorization or the approval of regulatory authorities of other countries may not be accepted by regulatory authorities outside Canada or other countries.
Regulation in the United States
In the United States, the FDA controls and investigates the investigation, manufacturing, and sale of new drugs. New drugs require FDA approval of a NDA prior to commercial sale. In the case of certain biological products, a Biological License Application (“BLA”) must be obtained prior to marketing and batch releasing. As in Canada, to obtain marketing approval, data from adequate and well-controlled human clinical trials, demonstrating to the FDA’s satisfaction a new drug’s safety and effectiveness for its intended use, are required. Data are generated in studies conducted pursuant to an IND submission, similar to that required for a clinical trial application in Canada. Clinical trials with human subjects are characterized as Phase I, Phase II and Phase III trials or a combination thereof. In a marketing application, the manufacturer must also demonstrate the identity, potency, quality and purity of the active ingredients of the new drug involved, and the stability of those ingredients. Further, the manufacturing facilities, equipment, processes and quality controls for the new drug must comply with the FDA’s current [Good Manufacturing Practice] regulations for drugs or biological products both in a pre-licensing inspection before product licensing and in subsequent periodic inspections after licensing. An establishment license grants the sponsor permission to fabricate, package, label, distribute, import, wholesale or test of the newly approved drug.
| 17 |
Federally regulated trials must be approved and monitored by an independent committee of doctors, scientists, advocates and others to ensure safety and ethical standards. These committees are called IRBs or ERBs. The review boards study and approve all study-related documents before a clinical trial begins and also carefully monitor data to detect benefit or harm, and validity of results.
The above describes briefly what is necessary for a new drug to be approved for marketing in North America. The European Medicines Agency and Japanese Pharmaceuticals and Medical Devices Agency are also important regulatory authorities in drug development. Together with the FDA, they are the three International Conference on Harmonization parties which oversee the three largest markets for drug sales.
Financings
In October 2017, we entered into a Common Shares Purchase Agreement (the “Purchase Agreement”) with Aspire Capital Fund, LLC (“Aspire Capital”) to sell up to US $15.5 million of common shares to Aspire Capital. Under the terms of the Purchase Agreement, Aspire Capital has made an initial purchase of 357,143 common shares at a price of $1.40 per share, representing gross proceeds of approximately $500,000 ($324,000 net of share issue costs). Under the terms of the Purchase Agreement, Aspire Capital has committed to purchase up to an aggregate of $15.0 million of our common shares, at our request from time to time during a 30-month period beginning on the effective date of a registration statement related to the transaction and at prices based on the market price at the time of each sale. Under terms of the Purchase Agreement, we also issued 321,429 common shares to Aspire Capital as consideration for Aspire Capital entering into the Purchase Agreement. Subsequent to the year end, we issued an additional 3.2 million common shares under the Purchase agreement for gross proceeds of approximately $8.9 million.
We intend to use this equity arrangement as an additional option to assist us in achieving our capital objectives. The equity line provides us with the opportunity to regularly raise capital at prevailing market prices, at our sole discretion providing us with the ability to better manage our cash resources.
| IV. | Risk Factors and Uncertainties |
Investing in our securities involves a high degree of risk. Before making an investment decision with respect to our Common Shares, you should carefully consider the following risk factors. Additional risks not currently known by us or that we consider immaterial at the present time may also impair our business, financial condition, prospects or results of operations. If any of the following risks occur, our business, financial condition, prospects or results of operations would likely be materially adversely affected. In that case, the trading price of our Common Shares could decline and you may lose all or part of the money you paid to buy our Common Shares. The risks set out below are not the only risks and uncertainties we currently face; other risks may arise in the future.
Risks Related to our Business
We are an early stage development company.
We are at an early stage of development. In the past five years, none of our potential products has obtained regulatory approval for commercial use and sale in any country and as such, no significant revenues have resulted from product sales. Significant additional investment will be necessary to complete the development of any of our product candidates. Preclinical and clinical trial work must be completed before our potential products could be ready for use within the markets that we have identified. We may fail to develop any products, obtain regulatory approvals, enter clinical trials or commercialize any products. We do not know whether any of our potential product development efforts will prove to be effective, meet applicable regulatory standards, obtain the requisite regulatory approvals, be capable of being manufactured at a reasonable cost or be accepted in the marketplace. We also do not know whether sales, license fees or related royalties will allow us to recoup any investment we make in the commercialization of our products.
| 18 |
The product candidates we are currently developing are not expected to be commercially viable for at least the next several years and we may encounter unforeseen difficulties or delays in commercializing our product candidates. In addition, our potential products may not be effective or may cause undesirable side effects.
Our product candidates require significant funding to reach regulatory approval assuming positive clinical results. For example, our product candidate APTO-253 began enrolment in a Phase I clinical trial in patients with relapsed or refractory hematologic malignancies and was placed on clinical hold by the FDA following a voluntary suspension of dosing by us. We are currently working with the FDA to have such hold lifted, but significant additional funding or a partnership will be necessary to complete a restarted Phase I clinical and, if required, Phase II or Phase III clinical trials. Such funding for our product candidates may be difficult, or impossible to raise in the public or private markets or through partnerships. If funding or partnerships are not readily attainable, the development of our product candidates may be significantly delayed or stopped altogether. The announcement of a delay or discontinuation of development would likely have a negative impact on our share price.
We need to raise additional capital.
We have an ongoing need to raise additional capital. To obtain the necessary capital, we must rely on some or all of the following: additional share issues, debt issuances (including promissory notes), collaboration agreements or corporate partnerships and grants and tax credits to provide full or partial funding for our activities. Additional funding may not be available on terms that are acceptable to us or in amounts that will enable us to carry out our business plan.
Our need for capital may require us to:
| • | engage in equity financings that could result in significant dilution to existing investors; |
| • | delay or reduce the scope of or eliminate one or more of our development programs; |
| • | obtain funds through arrangements with collaborators or others that may require us to relinquish rights to technologies, product candidates or products that we would otherwise seek to develop or commercialize ourselves; |
| • | license rights to technologies, product candidates or products on terms that are less favourable to us than might otherwise be available; |
| • | considerably reduce operations; or |
| • | cease our operations. |
| 19 |
We have a history of operating losses. We expect to incur net losses and we may never achieve or maintain profitability.
We have not been profitable since our inception in 1986. We reported net losses of $11.7 million in the fiscal year ended December 31, 2017, $14.2 million in the fiscal year ended December 31, 2016, $11.7 million in the fiscal year ended December 31, 2015, and as of December 31, 2017, we had an accumulated deficit of $247 million.
We have not generated any significant revenue to date and it is possible that we will never have sufficient product sales revenue (if any) to achieve profitability. We expect to continue to incur losses for at least the next several years as we or our collaborators and licensees pursue clinical trials and research and development efforts. To become profitable, we, either alone or with our collaborators and licensees, must successfully develop, manufacture and market our current product candidates APTO-253 or CG’806 as well as continue to identify, develop, manufacture and market new product candidates. It is possible that we will never have significant product sales revenue or receive royalties on our licensed product candidates. If funding is insufficient at any time in the future, we may not be able to develop or commercialize our products, take advantage of business opportunities or respond to competitive pressures.
We currently do not earn any revenues from our drug candidates and are therefore considered to be in the development stage. The continuation of our research and development activities and the commercialization of the targeted therapeutic products are dependent upon our ability to successfully finance and complete our research and development programs through a combination of equity financing and payments from strategic partners. We have no current sources of significant payments from strategic partners.
Clinical trials are long, expensive and uncertain processes and the FDA or Health Canada may ultimately not approve any of our product candidates. We may never develop any commercial drugs or other products that generate revenues.
In the past five years, none of our product candidates has received regulatory approval for commercial use and sale in North America. We cannot market a pharmaceutical product in any jurisdiction until it has completed thorough preclinical testing and clinical trials in addition to that jurisdiction’s extensive regulatory approval process. Approval in one country does not assure approval in another country. In general, significant research and development and clinical studies are required to demonstrate the safety and effectiveness of our product candidates before we can submit any regulatory applications.
Clinical trials are long, expensive and uncertain processes. Clinical trials may not be commenced or completed on schedule and the FDA or Health Canada or any other regulatory body may not ultimately approve our product candidates for commercial sale. The clinical trials of any of our drug candidates could be unsuccessful, which would prevent us from advancing, commercializing or partnering the drug.
Even if the results of our preclinical studies or clinical trials are initially positive, it is possible that we will obtain different results in the later stages of drug development or that results seen in clinical trials will not continue with longer term treatment. Positive results in Phase I clinical trials may not be repeated in larger Phase II or Phase III clinical trials.
Our preclinical studies and clinical trials may not generate positive results that will allow us to move towards the commercial use and sale of our product candidates. Furthermore, negative preclinical or clinical trial results may cause our business, financial condition, or results of operations to be materially adversely affected. For example, our Phase Ib clinical trial of APTO-253 in patients with AML was placed on clinical hold by the FDA in November 2015 and since that time the Company has encountered manufacturing setbacks which have further delayed the return of APTO-253 to the clinic. There can be no assurance that the clinical hold will be lifted by the FDA, that the Company will have the resources, or that we will decide, to continue the development of APTO-253. Even if the Phase Ib of APTO-253 is continued, there is a long development path ahead that will take many years to complete and is prone to the risks of failure or delays inherent in drug development. Likewise, our CG’806 product candidate has not yet entered clinical trials and it is expected to undergo many years of testing and regulatory examinations prior to any potential regulatory approvals.
| 20 |
Preparing, submitting and advancing applications for regulatory approval is complex, expensive and time intensive and entails significant uncertainty. A commitment of substantial resources to conduct time-consuming research, preclinical studies and clinical trials is required if we are to complete development of our products.
Clinical trials of our products require that we identify and enroll a large number of patients with the illness under investigation. We may not be able to enroll a sufficient number of appropriate patients to complete our clinical trials in a timely manner, particularly in smaller indications and indications where there is significant competition for patients. If we experience difficulty in enrolling a sufficient number of patients to conduct our clinical trials, we may need to delay or terminate ongoing clinical trials and will not accomplish objectives material to our success. Delays in planned patient enrolment or lower than anticipated event rates in our current clinical trials or future clinical trials also may result in increased costs, program delays, or both.
In addition, unacceptable toxicities or adverse side effects may occur at any time in the course of preclinical studies or human clinical trials or, if any product candidates are successfully developed and approved for marketing, during commercial use of any approved products. The appearance of any unacceptable toxicities or adverse side effects could interrupt, limit, delay or abort the development of any of our product candidates or, if previously approved, necessitate their withdrawal from the market. Furthermore, disease resistance or other unforeseen factors may limit the effectiveness of our potential products.
Our failure to develop safe, commercially viable drugs would substantially impair our ability to generate revenues and sustain our operations and would materially harm our business and adversely affect our share price.
We may not achieve our projected development goals in the time frames we announce and expect.
We set goals for, and make public statements regarding, the expected timing of the accomplishment of objectives material to our success, such as the submission of IND, the commencement and completion of clinical trials and the expected costs to develop our product candidates. The actual timing and costs of these events can vary dramatically due to factors within and beyond our control, such as delays or failures in our IND submissions or clinical trials, issues related to the manufacturing of drug supply, uncertainties inherent in the regulatory approval process, market conditions and interest by partners in our product candidates among other things. We may not make regulatory submissions or receive regulatory approvals as planned; our clinical trials may not be completed; or we may not secure partnerships for any of our product candidates. Any failure to achieve one or more of these milestones as planned would have a material adverse effect on our business, financial condition and results of operations.
| 21 |
Delays in clinical testing could result in delays in commercializing our product candidates and our business may be substantially harmed.
We cannot predict whether any clinical trials will begin as planned, will need to be restructured or will be completed on schedule, or at all. Our product development costs will increase if we experience delays in clinical testing. Significant clinical trial delays could shorten any periods during which we may have the exclusive right to commercialize our product candidates or allow our competitors to bring products to market before us, which would impair our ability to successfully commercialize our product candidates and may harm our financial condition, results of operations and prospects. The recommencement and completion of clinical trials for our products, including the APTO-253 phase I clinical trial and the IND submission and phase I clinical trial for CG’806, may be delayed for a number of reasons, including delays related, but not limited, to:
| • | failure by regulatory authorities to grant permission to proceed or placing the clinical trial on hold; |
| • | patients failing to enroll or remain in our trials at the rate we expect; |
| • | suspension or termination of clinical trials by regulators for many reasons, including concerns about patient safety or failure of our contract manufacturers to comply with cGMP requirements; |
| • | any changes to our manufacturing process that may be necessary or desired; |
| • | delays or failure to obtain GMP-grade clinical supply from contract manufacturers of our products necessary to conduct clinical trials; |
| • | product candidates demonstrating a lack of safety or efficacy during clinical trials; |
| • | patients choosing an alternative treatment for the indications for which we are developing any of our product candidates or participating in competing clinical trials; |
| • | patients failing to complete clinical trials due to dissatisfaction with the treatment, side effects or other reasons; |
| • | reports of clinical testing on similar technologies and products raising safety and/or efficacy concerns; |
| • | competing clinical trials and scheduling conflicts with participating clinicians; |
| • | clinical investigators not performing our clinical trials on their anticipated schedule, dropping out of a trial, or employing methods not consistent with the clinical trial protocol, regulatory requirements or other third parties not performing data collection and analysis in a timely or accurate manner; |
| • | failure of our contract research organizations, or CROs, to satisfy their contractual duties or meet expected deadlines; |
| • | inspections of clinical trial sites by regulatory authorities or IRBs, or ethics committees finding regulatory violations that require us to undertake corrective action, resulting in suspension or termination of one or more sites or the imposition of a clinical hold on the entire study; |
| 22 |
| • | one or more IRBs or ethics committees rejecting, suspending or terminating the study at an investigational site, precluding enrollment of additional subjects, or withdrawing its approval of the trial; or |
| • | failure to reach agreement on acceptable terms with prospective clinical trial sites. |
Our product development costs will increase if we experience delays in testing or approval or if we need to perform more or larger clinical trials than planned. Additionally, changes in regulatory requirements and policies may occur, and we may need to amend study protocols to reflect these changes. Amendments may require us to resubmit our study protocols to regulatory authorities or IRBs or ethics committees for re-examination, which may impact the cost, timing or successful completion of that trial. Delays or increased product development costs may have a material adverse effect on our business, financial condition and prospects.
We rely on contract manufacturers over whom we have limited control. If we are subject to quality, cost or delivery issues with the preclinical and clinical grade materials supplied by contract manufacturers, our business operations could suffer significant harm.
We rely on contract manufacturing organizations (“CMOs”), to manufacture our product candidates for some preclinical studies and clinical trials. We rely on CMOs for manufacturing, filling, packaging, storing and shipping of drug product in compliance with cGMP regulations applicable to our products. The FDA ensures the quality of drug products by carefully monitoring drug manufacturers’ compliance with cGMP regulations. The cGMP regulations for drugs contain minimum requirements for the methods, facilities and controls used in manufacturing, processing and packing of a drug product.
We contracted with multiple CMOs for the manufacture of APTO-253 and CG’806 to supply drug supply and then drug product for our clinical trials. The synthesis of CG’806 drug supply is challenging from a scale-up synthetic chemistry perspective. The formulation and manufacture of APTO-253 is a complex process with many variables involved. We pre-qualified CMOs to have the capacity, the systems and the experience to supply CG’806 and APTO-253 for our clinical trials. We have qualified the manufacturing facilities and the FDA has also performed site audits for our selected CMOs. In spite of the efforts to prequalify CMOs, delays and errors may occur, and any such manufacturing failures, delays or compliance issues could cause delays in the completion of our clinical trial programs.
There can be no assurances that CMOs will be able to meet our timetable and requirements. We have contracted with alternate suppliers in the event our current CMOs are unable to scale up production, or if our current CMOs otherwise experience any other significant problems in the manufacture of CG’806 and APTO-253. However, it is possible that all third-party manufacturing sources may experience failure or delays and may demand commercially unreasonable terms, which may lead to further delays in the development of our product candidates. Further, contract manufacturers must operate in compliance with cGMP and failure to do so could result in, among other things, the disruption of product supplies. Our dependence upon third parties for the manufacture of our products may adversely affect our profit margins and our ability to develop and deliver products on a timely and competitive basis.
If we have difficulty enrolling patients in clinical trials, the completion of the trials may be delayed or cancelled.
As our product candidates advance from preclinical testing to clinical testing, and then through progressively larger and more complex clinical trials, we will need to enroll an increasing number of patients that meet our eligibility criteria. There is significant competition for recruiting cancer patients in clinical trials, and we may be unable to enroll the patients we need to complete clinical trials on a timely basis or at all. Certain factors that affect enrollment of patients onto our clinical trials are impacted by external forces that may be beyond our control. Such factors include, but are not limited to, the following:
| 23 |
| • | size and nature of the patient population; |
| • | eligibility and exclusion criteria for the trial; |
| • | design of the study protocol; |
| • | competition with other companies for clinical sites or patients; |
| • | the perceived risks and benefits of the product candidate under study; |
| • | the patient referral practices of physicians; and |
| • | the number, availability, location and accessibility of clinical trial sites. |
If we are unable to successfully develop companion diagnostics for our therapeutic product candidates, or experience significant delays in doing so, we may not achieve marketing approval or realize the full commercial potential of our therapeutic product candidates.
We plan to develop companion diagnostics for our therapeutic product candidates. We expect that, at least in some cases, regulatory authorities may require the development and regulatory approval of a companion diagnostic as a condition to approving our therapeutic product candidates. We have limited experience and capabilities in developing or commercializing diagnostics and plan to rely in large part on third parties to perform these functions. We do not currently have any agreement in place with any third party to develop or commercialize companion diagnostics for any of our therapeutic product candidates.
Companion diagnostics are subject to regulation by the FDA, Health Canada and comparable foreign regulatory authorities as medical devices and may require separate regulatory approval or clearance prior to commercialization. If we, or any third parties that we engage to assist us, are unable to successfully develop companion diagnostics for our therapeutic product candidates, or experience delays in doing so, our business may be substantially harmed.
We rely and will continue to rely on third parties to conduct and monitor many of our preclinical studies and our clinical trials, and their failure to perform as required could cause substantial harm to our business.
We rely and will continue to rely on third parties to conduct a significant portion of our preclinical and clinical development activities. Preclinical activities include in vivo studies providing access to specific disease models, pharmacology and toxicology studies, and assay development. Clinical development activities include trial design, regulatory submissions, clinical patient recruitment, clinical trial monitoring, clinical data management and analysis, safety monitoring and project management, contract manufacturing and quality assurance. If there is any dispute or disruption in our relationship with third parties, or if they are unable to provide quality services in a timely manner and at a feasible cost, our active development programs will face delays. Further, if any of these third parties fails to perform as we expect or if their work fails to meet regulatory requirements, our testing could be delayed, cancelled or rendered ineffective.
| 24 |
We heavily rely on the capabilities and experience of our key executives and scientists and the loss of any of them could affect our ability to develop our products.
The loss of Dr. William G. Rice, our Chairman, President and Chief Executive Officer, or other key members of our staff, including Gregory Chow, our Senior Vice President and Chief Financial Officer, could harm us. We have employment agreements with Dr. Rice and Mr. Chow, although such employment agreements do not guarantee their retention. We also depend on our scientific and clinical collaborators and advisors, all of whom have outside commitments that may limit their availability to us. In addition, we believe that our future success will depend in large part upon our ability to attract and retain highly skilled scientific, managerial, medical, clinical and regulatory personnel, particularly as we expand our activities and seek regulatory approvals for clinical trials. We routinely enter into consulting agreements with our scientific and clinical collaborators and advisors, key opinion leaders and academic partners in the ordinary course of our business. We also enter into contractual agreements with physicians and institutions who will recruit patients into our clinical trials on our behalf in the ordinary course of our business. Notwithstanding these arrangements, we face significant competition for these types of personnel from other companies, research and academic institutions, government entities and other organizations. We cannot predict our success in hiring or retaining the personnel we require for continued growth. The loss of the services of any of our executive officers or other key personnel could potentially harm our business, operating results or financial condition.
Our employees may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could have a material adverse effect on our business.
We are exposed to the risk of employee fraud or other misconduct. Misconduct by employees could include failures to comply with FDA/Health Canada regulations, provide accurate information to the FDA/Health Canada, comply with manufacturing standards we have established, comply with federal, state and provincial health-care fraud and abuse laws and regulations, report financial information or data accurately or disclose unauthorized activities to us. In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Employee misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a substantial impact on our business and results of operations, including the imposition of substantial fines or other sanctions.
We may expand our business through the acquisition of companies or businesses or by entering into collaborations or by in-licensing product candidates, each of which could disrupt our business and harm our financial condition.
We may in the future seek to expand our pipeline and capabilities by acquiring one or more companies or businesses, entering into collaborations or in-licensing one or more product candidates. For example, in June 2016, we entered into a definitive agreement with CG, granting Aptose an exclusive option to research, develop and commercialize CG’806 in all countries of the world except Korea and China, for all fields of use.
Acquisitions, collaborations and in-licenses involve numerous risks, including, but not limited to:
| • | substantial cash expenditures; |
| 25 |
| • | technology development risks; |
| • | potentially dilutive issuances of equity securities; |
| • | incurrence of debt and contingent liabilities, some of which may be difficult or impossible to identify at the time of acquisition; |
| • | difficulties in assimilating the operations of the acquired companies; |
| • | potential disputes regarding contingent consideration; |
| • | diverting our management’s attention away from other business concerns; |
| • | entering markets in which we have limited or no direct experience; |
| • | potential loss of our key employees or key employees of the acquired companies or businesses; and |
| • | failure of the in-licenses agents or technologies to deliver the desired activities or functions. |
We have experience in entering collaborations and in-licensing product candidates, however, we cannot provide assurance that any acquisition, collaboration or in-license will result in short-term or long-term benefits to us. We may incorrectly judge the value or worth of an acquired company or business or in-licensed product candidate. In addition, our future success would depend in part on our ability to manage the rapid growth associated with some of these acquisitions, collaborations and in-licenses. We cannot assure you that we would be able to successfully combine our business with that of acquired businesses, manage a collaboration or integrate in-licensed product candidates. Furthermore, the development or expansion of our business may require a substantial capital investment by us.
Negative results from clinical trials or studies of others and adverse safety events involving the targets of our products may have an adverse impact on our future commercialization efforts.
From time to time, studies or clinical trials on various aspects of biopharmaceutical products are conducted by academic researchers, competitors or others. The results of these studies or trials, when published, may have a significant effect on the market for the biopharmaceutical product that is the subject of the study. The publication of negative results of studies or clinical trials or adverse safety events related to our product candidates, or the therapeutic areas in which our product candidates compete, could adversely affect our share price and our ability to finance future development of our product candidates, and our business and financial results could be materially and adversely affected.
The design or our execution of clinical trials may not support regulatory approval.
The design or execution of a clinical trial can determine whether its results will support regulatory approval and flaws in the design or execution of a clinical trial may not become apparent until the clinical trial is well advanced. In some instances, there can be significant variability in safety or efficacy results between different trials of the same product candidate due to numerous factors, including changes in trial protocols, differences in size and type of the patient populations, adherence to the dosing regimen and other trial protocols and the rate of dropout among clinical trial participants. We do not know whether any Phase II, Phase III or other clinical trials that we may conduct will demonstrate consistent or adequate efficacy and safety to obtain regulatory approval to market our product candidates.
| 26 |
Further, the FDA, Health Canada and comparable foreign regulatory authorities have substantial discretion in the approval process and in determining when or whether regulatory approval will be obtained for any of our product candidates. Our product candidates may not be approved even if they achieve their primary endpoints in future Phase 3 clinical trials or registration trials. The FDA, Health Canada or other regulatory authorities may disagree with our trial design and the Company’s interpretation of data from preclinical studies and clinical trials. In addition, any of these regulatory authorities may change requirements for the approval of a product candidate even after reviewing and providing comments or advice on a protocol for a pivotal Phase 3 clinical trial that has the potential to result in FDA, Health Canada or other agencies’ approval. In addition, any of these regulatory authorities may also approve a product candidate for fewer or more limited indications than the Company requests or may grant approval contingent on the performance of costly post-marketing clinical trials. The FDA, Health Canada or other regulatory authorities may not approve the labeling claims that we believe would be necessary or desirable for the successful commercialization of our product candidates.
As a result of intense competition and technological change in the biotechnical and pharmaceutical industries, the marketplace may not accept our products or product candidates, and we may not be able to compete successfully against other companies in our industry and achieve profitability.
Many of our competitors have:
| • | drug products that have already been approved or are in development, and operate large, well-funded research and development programs in the biotechnical and pharmaceutical fields; |
| • | substantially greater financial, technical and management resources, stronger intellectual property positions and greater manufacturing, marketing and sales capabilities, areas in which we have limited or no experience; and |
| • | significantly greater experience than we do in undertaking preclinical testing and clinical trials of new or improved pharmaceutical products and obtaining required regulatory approvals. |
Consequently, our competitors may obtain FDA, Health Canada and other regulatory approvals for product candidates sooner and may be more successful in manufacturing and marketing their products than we or our collaborators are.
Our competitors’ existing and future products, therapies and technological approaches will compete directly with the products we seek to develop. Current and prospective competing products may be more effective than our existing and future products insofar as they may provide greater therapeutic benefits for a specific problem or may offer easier delivery or comparable performance at a lower cost.
Any product candidate that we develop and that obtains regulatory approval must then compete for market acceptance and market share. Our products may not gain market acceptance among physicians, patients, healthcare payers, insurers, the medical community and other stakeholders. The degree of market acceptance of our product candidates, if approved for commercial sale, will depend on a number of factors, including:
| • | efficacy and potential advantages compared to alternative treatments; |
| • | the ability to offer its product candidates for sale at competitive prices; |
| • | convenience and ease of administration compared to alternative treatments; |
| 27 |
| • | the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies; |
| • | the strength of marketing and distribution support; |
| • | sufficient third party coverage or reimbursement; and |
| • | the prevalence and severity of any side effects. |
Further, any products we develop may become obsolete before we recover any expenses we incurred in connection with the development of these products. As a result, we may never achieve profitability.
Risks Related to our Intellectual Property
We may be unable to obtain patents to protect our technologies from other companies with competitive products, and patents of other companies could prevent us from manufacturing, developing or marketing our products.
Patent protection
The patent positions of pharmaceutical and biotechnology companies are uncertain and involve complex legal and factual questions. The USPTO and many other patent offices in the world have not established a consistent policy regarding the breadth of claims that they will allow in biotechnology patents.
Our pending patent applications may not result in issued patents and our issued patents may not be held valid and enforceable if challenged. Competitors may be able to circumvent any such issued patents by adoption of a competitive, though non-infringing product or process. Interpretation and evaluation of pharmaceutical or biotechnology patent claims present complex and often novel legal and factual questions. Our business could be adversely affected by increased competition in the event that any patent granted to it is held to be invalid or unenforceable or is inadequate in scope to protect our operations.
Allowable patentable subject matter and the scope of patent protection obtainable may differ between jurisdictions. If a patent office allows broad claims, the number and cost of patent interference proceedings in the United States, or analogous proceedings in other jurisdictions and the risk of infringement litigation may increase. If it allows narrow claims, the risk of infringement may decrease, but the value of our rights under our patents, licenses and patent applications may also decrease.
The scope of the claims in a patent application can be significantly modified during prosecution before the patent is issued. Consequently, we cannot know whether our pending applications will result in the issuance of patents or, if any patents are issued, whether they will provide us with significant proprietary protection or will be circumvented, invalidated or found to be unenforceable.
Publication of discoveries in scientific or patent literature often lags behind actual discoveries. Patent applications filed in the United States generally will be published 18 months after the filing date unless the applicant certifies that the invention will not be the subject of a foreign patent application. In many other jurisdictions, such as Canada, patent applications are published 18 months from the priority date. We may not be aware of such literature. Accordingly, we cannot be certain that the named inventors of our products and processes were the first to invent that product or process or that we were the first to pursue patent coverage for our inventions.
| 28 |
In addition, United States patent laws may change which could prevent or limit us from filing patent applications or patent claims in the United States to protect our products and technologies or limit the exclusivity periods that are available to patent holders for United States patents. For example, the Leahy-Smith America Invents Act, (the “Leahy-Smith Act”) was signed into law in 2011 and includes a number of significant changes to United States patent law. These include changes to transition from a “first-to-invent” system to a “first-to-file” system and to the way issued patents are challenged. These changes may favour larger and more established companies that have more resources to devote to patent application filing and prosecution. It is not clear what, if any, impact the Leahy-Smith Act will ultimately have on the cost of prosecuting our patent applications in the United States, our ability to obtain patents in the United States based on our discoveries and our ability to enforce or defend our United States issued patents.
Until such time, if ever, that further patents are issued to us, we will rely upon the law of trade secrets to the extent possible given the publication requirements under international patent treaty laws and/or requirements under foreign patent laws to protect our technology and our products incorporating the technology. In this regard, we have adopted certain confidentiality procedures. These include: limiting access to confidential information to certain key personnel; requiring all directors, officers, employees and consultants and others who may have access to our intellectual property to enter into confidentiality agreements which prohibit the use of or disclosure of confidential information to third parties; and implementing physical security measures designed to restrict access to such confidential information and products. Our ability to maintain the confidentiality of our technology is crucial to our ultimate possible commercial success. The procedures adopted by us to protect the confidentiality of our technology may not be effective, third parties may gain access to our trade secrets or our trade secrets or those of our collaborators may be independently discovered by others. Our collaborators, employees and consultants and other parties may not comply with the terms of their agreements with us, and we might be unable to adequately enforce our rights or obtain adequate compensation for the damages caused by unauthorized disclosure or use of our trade secrets or know how. Further, by seeking patent protection in various countries, it is inevitable that a substantial portion of our technology will become available to our competitors, through publication of such patent applications.
Enforcement of intellectual property rights
Protection of the rights revealed in published patent applications can be complex, costly and uncertain. Our commercial success depends in part on our ability to maintain and enforce our proprietary rights. If third parties engage in activities that infringe our proprietary rights, our management’s focus will be diverted and we may incur significant costs in asserting our rights. We may not be successful in asserting our proprietary rights, which could result in our patents being held invalid or a court holding that the third party is not infringing, either of which would harm our competitive position.
Others may design around our patented technology. We may have to participate in interference proceedings declared by the United States Patent and Trademark Office, European opposition proceedings, or other analogous proceedings in other parts of the world to determine priority of invention and the validity of patent rights granted or applied for, which could result in substantial cost and delay, even if the eventual outcome is favourable to us. Our pending patent applications, even if issued, may not be held valid or enforceable.
Our products and product candidates may infringe the intellectual property rights of others, or others may infringe on our intellectual property rights which could increase our costs.
Our success also depends on avoiding infringement of the proprietary technologies of others. In particular, there may be certain issued patents and patent applications claiming subject matter which we or our collaborators may be required to license in order to research, develop or commercialize APTO-253 or CG’806. In addition, third parties may assert infringement or other intellectual property claims against us. An adverse outcome in these proceedings could subject us to significant liabilities to third-parties, require disputed rights to be licensed from third-parties or require us to cease or modify our use of the technology. If we are required to license third-party technology, a license under such patents and patent applications may not be available on acceptable terms or at all. Further, we may incur substantial costs defending ourselves in lawsuits against charges of patent infringement or other unlawful use of another’s proprietary technology. We may also need to bring claims against others who we believe are infringing our rights in order to become or remain competitive and successful. Any such claims can be time consuming and expensive to pursue.
| 29 |
We may incur substantial cost in defending our intellectual property.
While we believe that our products and technology do not infringe proprietary rights of others, third parties may assert infringement claims in the future and such claims could be successful. Even if challenges are unsuccessful, we could incur substantial costs in defending ourselves against patent infringement claims brought by others or in prosecuting suits against others. In addition, others may obtain patents that we would need to license, which may not be available to us on reasonable terms. Whether we are able to obtain a necessary license would depend on the terms offered, the degree of risk of infringement and the need for the patent.
If product liability, clinical trial liability or environmental liability claims are brought against us or we are unable to obtain or maintain product liability, clinical trial or environmental liability insurance, we may incur substantial liabilities that could reduce our financial resources.
The clinical testing and commercial use of pharmaceutical products involves significant exposure to product liability, clinical trial liability, environmental liability and other risks that are inherent in the testing, manufacturing and marketing of our products. These liabilities, if realized, could have a material adverse effect on the Company’s business, results of operations and financial condition.
We have obtained limited product liability insurance coverage for our clinical trials on humans; however, our insurance coverage may be insufficient to protect us against all product liability damages. Regardless of merit or eventual outcome, liability claims may result in decreased demand for a future product, injury to reputation, withdrawal of clinical trial volunteers, loss of revenue, costs of litigation, distraction of management and substantial monetary awards to plaintiffs. Additionally, if we are required to pay a product liability claim, we may not have sufficient financial resources to complete development or commercialization of any of our product candidates and our business and results of operations will be adversely affected. In general, insurance will not protect us against some of our own actions, such as negligence.
As the Company’s development activities progress towards the commercialization of product candidates, our liability coverage may not be adequate, and the Company may not be able to obtain adequate product liability insurance coverage at a reasonable cost, if at all. Even if the Company obtains product liability insurance, its financial position may be materially adversely affected by a product liability claim. A product liability claim could also significantly harm the Company’s reputation and delay market acceptance of its product candidates. Additionally, product recalls may be issued at the direction of the FDA, other government agencies or other companies having regulatory control for pharmaceutical sales. If a product recall occurs in the future, such a recall could adversely affect our business, financial condition or reputation.
| 30 |
If we fail to comply with environmental, health and safety laws and regulations, we could become subject to fines or penalties or incur costs that could have a material adverse effect on the success of our business.
We are subject to numerous environmental, health and safety laws and regulations, including those governing laboratory procedures and the handling, use, storage, treatment and disposal of hazardous materials and wastes. Our operations involve the use of hazardous and flammable materials, including chemicals and radioactive and biological materials. Our operations also produce hazardous waste products. We generally contract with third parties for the disposal of these materials and wastes. We cannot eliminate the risk of contamination or injury from these materials. In the event of contamination or injury resulting from our use of hazardous materials, we could be held liable for any resulting damages, and any liability could exceed our resources. We also could incur significant costs associated with civil or criminal fines and penalties.
Although we maintain workers’ compensation insurance to cover us for costs and expenses we may incur due to injuries to our employees resulting from the use of hazardous materials, this insurance may not provide adequate coverage against potential liabilities. We do not maintain insurance for environmental liability or toxic tort claims that may be asserted against us in connection with our storage or disposal of biological, hazardous or radioactive materials.
In addition, we may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations. These current or future laws and regulations may impair our research, development or production efforts. Failure to comply with these laws and regulations also may result in substantial fines, penalties or other sanctions.
We may be unable to obtain partnerships for our product candidates, which could curtail future development and negatively affect our share price. In addition, our partners might not satisfy their contractual responsibilities or devote sufficient resources to our partnership.
Our strategy for the research, development and commercialization of our products requires entering into various arrangements with corporate collaborators, licensors, licensees and others, and our commercial success is dependent upon these outside parties performing their respective contractual responsibilities. The amount and timing of resources that such third parties will devote to these activities may not be within our control. These third parties may not perform their obligations as expected and our collaborators may not devote adequate resources to our programs. In addition, we could become involved in disputes with our collaborators, which could result in a delay or termination of the related development programs or result in litigation. We intend to seek additional collaborative arrangements to develop and commercialize some of our products. We may not be able to negotiate collaborative arrangements on favourable terms, or at all, in the future, and our current or future collaborative arrangements may not be successful.
If we cannot negotiate collaboration, license or partnering agreements, we may never achieve profitability and we may not be able to continue to develop our product candidates. Commencing Phase I Phase II and Phase III clinical trials for CG’806 and continuing Phase Ib, and commencing Phase II and Phase III clinical trials for APTO-253 would require significant amounts of funding and such funding may not be available to us.
Exchange rate risk
We may be exposed to fluctuations of the United States dollar against certain other currencies because we hold most of our cash and cash equivalents in United States dollars, while we incur some of our expenses in foreign currencies, primarily the Canadian dollar. Fluctuations in the value of currencies could cause us to incur currency exchange losses. We do not currently employ a hedging strategy against exchange rate risk. We cannot assert with any assurance that we will not suffer losses as a result of unfavorable fluctuations in the exchange rates between the United States dollar, the Canadian dollar and other currencies.
| 31 |
Extensive Government Regulation
Government regulation is a significant factor in the development, production and marketing of the Company’s products. Research and development, testing, manufacture, marketing and sales of pharmaceutical products or related products are subject to extensive regulatory oversight, often in multiple jurisdictions, which may cause significant additional costs and/or delays in bringing products to market, and in turn, may cause significant losses to investors. The regulations applicable to the Company’s product candidates may change. Even if granted, regulatory approvals may include significant limitations on the uses for which products can be marketed or may be conditioned on the conduct of post-marketing surveillance studies. Failure to comply with applicable regulatory requirements can, among other things, result in warning letters, the imposition of civil penalties or other monetary payments, delay in approving or refusal to approve a product candidate, suspension or withdrawal of regulatory approval, product recall or seizure, operating restrictions, interruptions of clinical trials or manufacturing, injunctions or criminal prosecution. In addition, regulatory agencies many not approve the labeling claims that are necessary or desirable for the successful commercialization of the Company’s product candidates.
Requirements for regulatory approval vary widely from country to country. Whether or not approved in Canada or the United States, regulatory authorities in other countries must approve a product prior to the commencement of marketing the product in those countries. The time required to obtain any such approval may be longer or shorter than in Canada or the United States. Approved drugs, as well as their manufacturers, are subject to continuing and ongoing review, and discovery of problems with these products or the failure to adhere to manufacturing or quality control requirements may result in regulatory restrictions being imposed.
Risks Related to Our Common Shares
Our share price has been and is likely to continue to be volatile and an investment in our Common Shares could suffer a decline in value.
You should consider an investment in our Common Shares as risky and invest only if you can withstand a significant loss and wide fluctuations in the market value of your investment. The market price of our Common Shares has been highly volatile and is likely to continue to be volatile. This leads to a heightened risk of securities litigation pertaining to such volatility. Factors affecting our Common Share price include but are not limited to:
| • | our ability to continue as a going concern; |
| • | our ability to raise additional capital; |
| • | the progress of our pre-clinical and clinical trials; |
| • | our ability to obtain partners and collaborators to assist with the future development of our products; |
| • | general market conditions; |
| 32 |
| • | announcements of technological innovations or new product candidates by us, our collaborators or our competitors; |
| • | published reports by securities analysts; |
| • | developments in patent or other intellectual property rights; |
| • | the cash and investments held by us and our ability to secure future financing; |
| • | public concern as to the safety and efficacy of drugs that we and our competitors develop; |
| • | shareholder interest in our Common Shares; and |
| • | low liquidity in the daily trading volume of our Common Shares. |
Future sales of our Common Shares by us or by our existing shareholders could cause our share price to fall.
The issuance of Common Shares by us could result in significant dilution in the equity interest of existing shareholders and adversely affect the market price of our Common Shares. Sales by existing shareholders of a large number of our Common Shares in the public market and the issuance of Common Shares in connection with strategic alliances, or the perception that such additional sales could occur, could cause the market price of our Common Shares to decline and have an undesirable impact on our ability to raise capital.
We are susceptible to stress in the global economy and therefore, our business may be affected by the current and future global financial condition.
If the increased level of volatility and market turmoil that have marked recent years continue, our operations, business, financial condition and the trading price of our Common Shares could be materially adversely affected. Furthermore, general economic conditions may have a great impact on us, including our ability to raise capital, our commercialization opportunities and our ability to establish and maintain arrangements with others for research, manufacturing, product development and sales.
An active trading market in our Common Shares may not be sustained.
Our Common Shares are listed for trading on the NASDAQ and the TSX. However, an active trading market in our Common Shares on the stock exchanges may not be sustained and we may not be able to maintain our listings.
Other Risks
It may be difficult for non-Canadian investors to obtain and enforce judgments against us because of our Canadian incorporation and presence.
We are a corporation existing under the laws of Canada. Some of our directors and officers, and many of the experts named in this AIF and the documents incorporated by reference into this AIF, are residents of Canada, and all or a substantial portion of their assets, and a substantial portion of our assets, are located outside the United States. Consequently, although we have appointed an agent for service of process in the United States, it may be difficult for holders of our shares who reside in the United States to effect service within the United States upon our directors and officers and experts who are not residents of the United States. It may also be difficult for holders of our shares who reside in the United States to realize in the United States upon judgments of courts of the United States predicated upon our civil liability and the civil liability of our directors, officers and experts under the United States federal securities laws. Investors should not assume that Canadian courts (i) would enforce judgments of United States courts obtained in actions against us or our directors, officers or experts predicated upon the civil liability provisions of the United States federal securities laws or the securities or “blue sky” laws of any state within the United States or (ii) would enforce, in original actions, liabilities against us or our directors, officers or experts predicated upon the United States federal securities laws or any such state securities or “blue sky” laws. In addition, we have been advised by our Canadian counsel that in normal circumstances, only civil judgments and not other rights arising from United States securities legislation are enforceable in Canada and that the protections afforded by Canadian securities laws may not be available to investors in the United States.
| 33 |
We are likely a “passive foreign investment company” which may have adverse United States federal income tax consequences for United States shareholders.
United States investors in our Common Shares should be aware that the Company believes it was classified as a passive foreign investment company (“PFIC”) during the tax year ended December 31, 2017, and based on the nature of our business, the projected composition of our gross income and the projected composition and estimated fair market value of our assets, the Company expects to be a PFIC for the current tax year ending December 31, 2018 and may be a PFIC in subsequent tax years. If the Company is a PFIC for any year during a United States shareholder’s holding period, then such United States shareholder generally will be required to treat any gain realized upon a disposition of Common Shares, or any so-called “excess distribution” received on its Common Shares, as ordinary income, and to pay an interest charge on a portion of such gain or distributions, unless the shareholder makes a timely and effective “qualified electing fund” election (“QEF election”) or a “mark-to-market” election with respect to the Common Shares. A United States shareholder who makes a QEF election generally must report on a current basis its share of the Company’s net capital gain and ordinary earnings for any year in which the Company is a PFIC, whether or not the Company distributes any amounts to its shareholders. However, United States shareholders should be aware that we do not intend to satisfy record keeping requirements that apply to a qualified electing fund, and we do not intend to supply United States shareholders with information that such United States shareholders require to report under the QEF election rules, in the event that we are a PFIC and a United States shareholder wishes to make a QEF election. Thus, United States shareholders should assume that they will not be able to make a QEF election with respect to their Common Shares. A United States shareholder who makes the mark-to-market election generally must include as ordinary income each year the excess of the fair market value of the Common Shares over the taxpayer’s basis therein. Each United States shareholder should consult its own tax advisor regarding the United States federal, United States local, and foreign tax consequences of the PFIC rules and the acquisition, ownership, and disposition of our Common Shares.
We are an “emerging growth company,” and we cannot be certain if the reduced reporting requirements applicable to emerging growth companies will make our common shares less attractive to investors.
We are an “emerging growth company,” as defined in the Jumpstart Our Business Startups Act of 2012 (United States), or the JOBS Act. For as long as we continue to be an emerging growth company, we may take advantage of exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies, including not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002 (United States) (the “SOX”), reduced disclosure obligations regarding executive compensation in our periodic reports and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and shareholder approval of any golden parachute payments not previously approved.
| 34 |
We will cease to be an emerging growth company upon the earliest of:
| • | the last day of the fiscal year during which we have total annual gross revenues of $1,000,000,000 (as such amount is indexed for inflation every five years by the United States Securities Exchange Commission (the “SEC”) or more; |
| • | the last day of our fiscal year following the fifth anniversary of the completion of our first sale of common equity securities pursuant to an effective registration statement under the Securities Act (United States) which will be in September 2020; |
| • | the date on which we have, during the previous three-year period, issued more than $1,000,000,000 in non- convertible debt; or |
| • | the date on which we are deemed to be a “large accelerated filer”, as defined in Rule 12b–2 of the Exchange Act (United States) (the “Exchange Act”), which would occur if the market value of our ordinary shares that are held by non-affiliates exceeds $700,000,000 as of the last day of our most recently-completed second fiscal quarter. |
We cannot predict if investors will find our common shares less attractive because we may rely on these exemptions. If some investors find our Common Shares less attractive as a result, there may be a less active trading market for our Common Shares and our share price may be more volatile.
Any failure to maintain an effective system of internal controls may result in material misstatements of our consolidated financial statements or cause us to fail to meet our reporting obligations or fail to prevent fraud; and in that case, our shareholders could lose confidence in our financial reporting, which would harm our business and could negatively impact the price of our common shares.
Section 404(a) of the SOX requires that our management assess and report annually on the effectiveness of our internal controls over financial reporting and identify any material weaknesses in our internal controls over financial reporting. Although Section 404(b) of the SOX requires our independent registered public accounting firm to issue an annual report that addresses the effectiveness of our internal controls over financial reporting, we have opted to rely on the exemptions provided to us by virtue of being a foreign private issuer and an emerging growth company, and consequently will not be required to comply with SEC rules that implement Section 404(b) of SOX until we lose our emerging growth company status.
Effective internal controls are necessary for us to provide reliable financial reports and prevent fraud. If we fail to maintain an effective system of internal controls, we might not be able to report our financial results accurately or prevent fraud; and in that case, our shareholders could lose confidence in our financial reporting, which would harm our business and could negatively impact the price of our common shares. While we believe that we have sufficient personnel and review procedures to allow us to maintain an effective system of internal controls, we cannot assure you that we will not experience potential material weaknesses in our internal control. Even if we conclude that our internal control over financial reporting provides reasonable assurance regarding the reliability of financial reporting and the preparation of consolidated financial statements for external purposes in accordance with International Financial Reporting Standards, as issued by the International Accounting Standards Board, because of its inherent limitations, internal control over financial reporting may not prevent or detect fraud or misstatements. Failure to implement required new or improved controls, or difficulties encountered in their implementation, could harm our results of operations or cause us to fail to meet our future reporting obligations.
If we fail to timely achieve and maintain the adequacy of our internal control over financial reporting, we may not be able to produce reliable financial reports or help prevent fraud. Our failure to achieve and maintain effective internal control over financial reporting could prevent us from complying with our reporting obligations on a timely basis, which could result in the loss of investor confidence in the reliability of our consolidated financial statements, harm our business and negatively impact the trading price of our common shares.
| 35 |
As a foreign private issuer, we are not subject to certain United States securities law disclosure requirements that apply to a domestic United States issuer, which may limit the information which would be publicly available to our shareholders.
As a foreign private issuer, we are exempt from certain rules under the Exchange Act that impose disclosure requirements as well as procedural requirements for proxy solicitations under Section 14 of the Exchange Act. In addition, our officers, directors and principal shareholders are exempt from the reporting and “short-swing” profit recovery provisions of Section 16 of the Exchange Act. Moreover, we are not required to file periodic reports and financial statements with the SEC as frequently or as promptly as a company that files as a domestic issuer whose securities are registered under the Exchange Act, nor are we generally required to comply with the SEC’s Regulation Faire Disclosure, which restricts the selective disclosure of material non-public information. For as long as we are a “foreign private issuer” we intend to file our annual financial statements on Form 20-F and furnish our quarterly updates on Form 6-K to the SEC for so long as we are subject to the reporting requirements of Section 13(g) or 15(d) of the Exchange Act. However, the information we file or furnish is not the same as the information that is required in annual and quarterly reports on Form 10-K or Form 10-Q for United States domestic issuers. Accordingly, there may be less information publicly available concerning us than there is for a company that files as a domestic issuer.
Data security incidents and privacy breaches could result in important remediation costs, increased cyber security costs, litigation and reputational harm.
Cyber security incidents can result from deliberate attacks or unintentional events. Cyber-attacks and security breaches could include unauthorized attempts to access, disable, improperly modify or degrade the Company’s information, systems and networks, the introduction of computer viruses and other malicious codes and fraudulent “phishing” emails that seek to misappropriate data and information or install malware onto users’ computers. Cyber-attacks in particular vary in technique and sources, are persistent, frequently change and are increasingly more targeted and difficult to detect and prevent against. Our network security and data recovery measures and those of third parties with which we contract, many not be adequate to protect again cyber-attacks.
Disruptions due to cyber security incidents could adversely affect Aptose’s business. In particular, a cyber security incident could result in the loss or corruption of data from Aptose’s research and development activities, including clinical trials, which may cause significant delays to some or all of the Company’s clinical programs. Also, the Company’s trade secrets, including unpatented know-how, technology and other proprietary information could be disclosed to competitors further to a breach, which would harm the Company’s business and competitive position. We expect that risks and exposures related to cyber security attacks will remain high for the foreseeable future due to the rapidly evolving nature and sophistication of these threats. While we have invested in the protection of data and information technology, there can be no assurance that our efforts to implement adequate security measures would be sufficient to protect the Company against cyber-attacks.
| 36 |
We must successfully upgrade and maintain our information technology systems.
We rely on various information technology systems to manage our operations. There are inherent costs and risks associated with maintaining, modifying and/or changing these systems and implementing new systems, including potential disruption of our internal control structure, substantial capital expenditures, additional administration and operating expenses, retention of sufficiently skilled personnel to implement and operate its systems, demands on management time and other risks and costs of delays or difficulties in transitioning to new systems or of integrating new systems into our current systems. In addition, our information technology system implementations may not result in productivity improvements at a level that outweighs the costs of implementation, or at all. The implementation of new information technology systems may also cause disruptions in our business operations and have an adverse effect on our business, prospects, financial condition and operating results.
| V. | DIVIDENDS |
The Company has not declared or paid any dividends on its Common Shares to date. The payment of dividends in the future will be dependent on the Company’s earnings, financial condition and such other factors as the Board considers appropriate. However, the Company’s current policy is to reinvest future earnings in order to finance its growth and the development of its business. As a result, the Company does not intend to pay dividends in the foreseeable future.
| VI. | DESCRIPTION OF CAPITAL STRUCTURE |
We are authorized to issue an unlimited number of Common Shares. Our shareholders have no rights to share in our profits, are subject to no redemption or sinking fund provisions, have no liability for further capital calls and are not subject to any discrimination due to number of Common Shares owned.
Each holder of record of Common Shares, without par value, is entitled to one vote for each share held on all matters properly submitted to the shareholders for their vote, except matters which are required to be voted on as a particular class or series of stock. Cumulative voting for directors is not permitted.
Holders of outstanding Common Shares are entitled to those dividends declared by the board of directors out of legally available funds. By not more than 50 days nor less than seven days in advance of a dividend, the Board may establish a record date for the determination of the persons entitled to such dividend. In the event of liquidation, dissolution or winding up our affairs, holders of Common Shares are entitled to receive, pro rata, our net assets available after provision has been made for the preferential rights of the holders of preferred stock, including any surplus available after such event of liquidation, dissolution or winding up of the affairs of the Company. Holders of outstanding Common Shares have no pre-emptive, conversion or redemption rights. All of the issued and outstanding Common Shares are, and all unissued Common Shares, when offered and sold will be, duly authorized, validly issued, fully paid and non-assessable. To the extent that additional Common Shares may be issued in the future, the relative interests of the then existing shareholders may be diluted. There were 27,502,053 Common Shares issued and outstanding at December 31, 2017.
| VII. | Market For Securities |
Trading Price and Volume
The Common Shares are currently listed and posted for trading on the TSX and are traded under the symbol “APS” and NASDAQ under the symbol “APTO”.
| 37 |
The following table sets forth the reported high and low trade prices in Canadian dollars, the average volume of trading, and the cumulative volume of trading of the Common Shares as reported by the TSX for the periods indicated below:
| Price Range |
Total Cumulative Volume | ||
| High ($) | Low ($) | ||
| January 2017 | 1.93 | 1.23 | 3,213,034 |
| February 2017 | 1.66 | 1.27 | 1,176,580 |
| March 2017 | 1.82 | 1.35 | 924,968 |
| April 2017 | 1.38 | 1.05 | 1,264,036 |
| May 2017 | 1.79 | 1.16 | 1,901,623 |
| June 2017 | 2.20 | 1.36 | 1,009,177 |
| July 2017 | 2.19 | 1.63 | 1,070,017 |
| August 2017 | 2.20 | 1.69 | 592,046 |
| September 2017 | 2.12 | 1.69 | 1,250,418 |
| October 2017 | 2.07 | 1.64 | 595,242 |
| November 2017 | 2.92 | 1.95 | 1,484,171 |
| December 2017 | 3.00 | 2.17 | 1,048,398 |
The following table sets forth the reported high and low trade prices in Canadian dollars, the average volume of trading, and the cumulative volume of trading of the Common Shares as reported by NASDAQ for the periods indicated below:
| Price Range |
Total Cumulative Volume | ||
| High ($) | Low ($) | ||
| January 2017 | 2.07 | 0.91 | 9,878,125 |
| February 2017 | 1.29 | 0.98 | 7,822,362 |
| March 2017 | 1.37 | 1.01 | 9,984,969 |
| April 2017 | 1.05 | 0.78 | 4,648,218 |
| May 2017 | 1.32 | 0.86 | 12,405,311 |
| June 2017 | 1.70 | 1.00 | 10,651,964 |
| July 2017 | 1.75 | 1.25 | 8,193,357 |
| August 2017 | 1.75 | 1.36 | 5,057,911 |
| September 2017 | 1.75 | 1.38 | 7,204,650 |
| October 2017 | 1.61 | 1.30 | 6,255,650 |
| November 2017 | 2.30 | 1.50 | 8,801,894 |
| December 2017 | 2.58 | 1.68 | 15,383,437 |
Prior Sales
The only securities of Aptose that are outstanding but not listed or quoted on a marketplace are stock options and stock-based awards.
| 38 |
During the most recently completed financial year, we granted the following securities pursuant to our stock incentive plan: (i) on March 28, 2017, we granted (A) options to purchase an aggregate of 480,000 Common Shares at a price of Cdn$1.52 per Common Share, and (B) an aggregate of 150,000 restricted stock units which fully vested on June 28, 2017; (ii) on June 6, 2017, we granted options to purchase an aggregate of 191,250 Common Shares at a price of US$1.03 per Common Share; (iii) on June 6, 2017, we granted options to purchase an aggregate of 56,250 Common Shares at a price of Cdn$1.38 per Common Share; (iv) on August 8, 2017, we granted options to purchase an aggregate of 32,500 Common Shares at a price of US$1.69 per Common Share; (v) on August 8, 2017, we granted options to purchase an aggregate of 20,000 Common Shares at a price of Cdn$2.04 per Common Share; (vi) on November 14, 2017, we granted options to purchase an aggregate of 8,000 Common Shares at a price of US$2.05 per Common Share; and (vii) on December 4, 2017, we granted options to purchase an aggregate of 38,500 Common Shares at a price of US$2.01 per Common Share.
| VIII. | Directors And Officers |
Directors
As at March 27, 2018, as a group, the Company’s directors and executive officers beneficially owned, directly or indirectly, or exercised control of over an aggregate of 306,782 Common Shares representing 1% of the issued and outstanding Common Shares as at such date. The information as to the number of Common Shares beneficially owned or over which control is exercised, not being within the knowledge of the Company, has been furnished by SEDI and confirmed with each director or executive officer, as the case may be, individually as of March 27, 2018.
We have an Audit Committee, a Corporate Governance and Nominating Committee and a Compensation Committee, the members of each such committee are shown below.
The following table sets forth the name, province or state and country of residence of each director of the Company and states the respective positions and offices held with the Company, their principal occupations during the last five years and the periods during which each director has served as a director of the Company. Each director holds office until the next annual meeting of shareholders or until his successor is duly elected, unless prior thereto the director resigns or the director’s office becomes vacant by reason of death or other cause.
|
Name and Municipality of Residence
|
Position Held with the Company | Principal Occupation during Past Five Years | Director Since |
|
Dr. Denis Burger(1)(2) (Oregon, United States)
|
Lead Director |
Chief Scientific Officer of Cytodyn Inc. (biotechnology company) (2015 to present)
President, Yamhill Valley Vineyards, Inc. (vineyards) (1983 to present)
Founding Chairman, Director, Trinity Biotech plc. (biotechnology company) (1992 to 1995 Chairman, 1995 to present as Director) |
September 2007 |
| 39 |
|
Dr. Bradley Thompson(1)(2)(3) (Alberta, Canada)
|
Director |
Chief Executive Officer, Kickshaw Ventures Ltd. (venture capital) (December 2016 to present)
Chief Executive Officer and Chief Technology Officer, Wyvern Pharmaceuticals Inc. (biotechnology company) (December 2016 to present)
President and Chief Executive Officer, Oncolytics Biotech Inc. (biotechnology company) (1999 to November 2016)
|
June 2013 |
|
Dr. Mark Vincent(3) (Ontario, Canada)
|
Director |
Physician; consultant medical oncologist, London Regional Cancer Program of Cancer Care Ontario (cancer care facility) (1990 to present)
Professor of Oncology, Western University of Ontario (university) (2008 to present)
|
September 2007 |
|
Warren Whitehead(1) (Ontario, Canada)
|
Director |
Financial Consultant (self-employed) (2015 to present)
Serves on several Boards of Directors (2009 to present)
Chief Financial Officer, ProMIS Neurosciences (formerly Amorfix Life Sciences) (biotechnology company) (2013 to 2015)
|
April 2011 |
|
Dr. William G. Rice (California, United States)
|
Chairman |
Chairman, President and Chief Executive Officer of the Company (2013 to present)
Chairman, President and Chief Executive Officer, Cylene Pharmaceuticals Inc. (biotechnology company) (2003 to 2013)
|
October 2013 |
|
Dr. Erich Platzer(2) Switzerland
|
Director |
Serves on several Boards of Directors (2003 to present)
Investment Advisor, HMB Partners AG (healthcare investment firm) (2003 to 2015)
|
December 2014 |
| (1) | Member of the Audit Committee |
| (2) | Member of the Compensation Committee |
| (3) | Member of the Corporate Governance and Nominating Committee |
| 40 |
Biographies
Dr. Denis Burger: Dr. Burger currently is the Chief Scientific Officer and member of the board of directors of Cytodyn Inc., a biotechnology company as well as Chairman of AMES Devices, a medical device company. Dr. Burger co-founded Trinity Biotech plc, based in Dublin, Ireland, in June 1992 and acted as Chairman from 1992 to 1995 and now serves on the board of directors of the company. Dr. Burger was the past Chairman, Chief Executive Officer and a director of AVI Biopharma Inc., an Oregon based biotechnology company, from 1992 to March 2007. Dr. Burger is also a partner in Sovereign Ventures, a healthcare consulting and funding firm based in Portland, Oregon. Dr. Burger received his MSc and Ph.D. in Microbiology and Immunology from the University of Arizona.
Dr. Erich Platzer: Dr. Platzer is a board certified physician in internal medicine, hematology and medical oncology. Previously, Dr. Platzer served as the business director of oncology, as well as the global strategic marketing and therapeutic area head of oncology at Roche, Basel. He was also the medical director in oncology and global development project leader and was responsible for various strategic corporate partnerships. Dr. Platzer is a director of Swiss Business Angel Groups, StartAngels and BioBAC, and has served as a pharmaceutical industry expert on the board of directors of multiple biotech companies in both the United States and Europe such as Probiodrug, AOT, Léman Micro Devices, Credentis, and Viroblock. Dr. Platzer co-founded HBM Healthcare Investments (formerly HBM BioVentures) a global leader in healthcare investing. He has over 12 years of experience in academic medicine and research and was a key member of the team at MSKCC that purified human G-CSF in 1983 (recombinant form: Neupogen®). He earned his M.D. from the Medical School and the Institute of Clinical Immunology and Rheumatology of the University of Erlangen, where he also received his “Dr. med. habil.” (M.D., Ph.D.).
Dr. William G. Rice: Dr. Rice joined Aptose as Chairman and Chief Executive Officer in October 2013. Prior to joining Aptose, Dr. Rice served as the President, Chief Executive Officer and Chairman of the board of Cylene Pharmaceuticals, Inc., a private biotechnology company (“Cylene”). Prior to Cylene, Dr. Rice was the founder, President, Chief Executive Officer and Director of Achillion Pharmaceuticals, Inc. He also served as Senior Scientist and Head of the Drug Mechanism Laboratory at the National Cancer Institute-Frederick Cancer Research and Development Center, and served as a faculty member in the division of Pediatric Hematology and Oncology at Emory University School of Medicine. Dr. Rice received his Ph.D. from Emory University Department of Biochemistry. He continues to serve as the Chairman of the board of Cylene and is a member of the board of directors of Oncolytics Biotech Inc.
Dr. Bradley Thompson: Dr. Thompson is an experienced biotechnology professional who is Chief Executive Officer of Kickshaw Ventures Ltd. and Chief Executive Officer and Chief Technology Officer of Wyvern Pharmaceuticals Inc. since December 2016. Prior to his role with Kickshaw and Wyvern Pharmaceuticals, Dr. Thompson was Chairman of the Board, President and Chief Executive Officer of Oncolytics Biotech Inc. from April 1999 to November 2016 and Chief Executive Officer of Synsorb Biotech from 1994 to 1999. He received his Ph.D. from the University of Western Ontario in the Department of Microbiology and Immunology.
Dr. Mark Vincent: Dr. Mark Vincent is a Professor of Oncology at the University of Western Ontario and a staff medical oncologist at the London Regional Cancer Program, where he has been since 1990. Dr. Vincent is also the co-founder and Chief Executive Officer of Sarissa, Inc. since 2000.
Mr. Warren Whitehead: Mr. Whitehead is a CPA (CMA) who has held senior financial management positions in several biotechnology and pharmaceutical companies. Most recently Mr. Whitehead was the Chief Financial Officer of ProMIS Neurosciences Inc. (formerly Amorfix Life Sciences Ltd.). Prior to this, he served as Chief Financial Officer of ARIUS Research Inc., providing financial guidance and leadership during the acquisition of ARIUS by Roche in 2008. Prior to ARIUS, Mr. Whitehead was Chief Financial Officer at Labopharm Inc., where he completed a series of public equity financings and a listing on NASDAQ. He is currently the Chairman of the board of directors of PlantForm Corporation.
| 41 |
Executive Officers
The following table sets forth the name, province or state and country of residence of the other non-director executive officers:
| Name and Municipality of Residence | Position held with the Company | Principal Occupation during Past Five Years |
|
Dr. William Rice (California, United States)
|
Chairman, President and Chief Executive Officer |
Chairman, President and Chief Executive Officer of the Company (2013 to present)
Chairman, President and Chief Executive Officer, Cylene Pharmaceuticals Inc. (biotechnology company) (2003 to 2013)
|
|
Gregory Chow (California, United States)
|
Senior Vice President and Chief Financial Officer |
Chief Financial Officer of the Company
Former Managing Director, Director of Private Placements at Wedbush Securities
Former Director in the Private Placements/Equity Capital Markets Group at RBC Capital Markets
Former Senior Auditor at BDO Seidman, LLP
|
Gregory Chow: Mr. Chow joined Aptose as Chief Financial Officer in December 2013. Previously, Mr. Chow served as Managing Director, Director of Private Placements at Wedbush Securities, where he led the private placement capital activities within the Life Sciences Investment Banking Group. Prior to joining Wedbush, he was a Director in the Private Placements / Equity Capital Markets Group at RBC Capital Markets, where he led life science private capital activities. Previously, he led the Private Capital Group at Wells Fargo Securities and was a Senior Auditor at BDO Seidman, LLP in their Century City, CA office. Mr. Chow is a Certified Public Accountant (inactive) in the State of California. Mr. Chow received his MBA in Finance from The Wharton School at the University of Pennsylvania, and his BA in Business Economics with an emphasis in Accounting from the University of California, Santa Barbara.
Shareholding, Cease Trade Orders, Bankruptcies, Penalties or Sanctions
Except as disclosed below and to the knowledge of the Company, none of the current executive officers or directors of the Company or shareholders holding a sufficient number of securities of the Company to affect materially the control thereof is, or within 10 years before the date hereof, has been:
| a. | a director, chief executive officer or chief financial officer of any corporation (including the Company) that: |
(i) was subject to an order that was issued while the proposed director was acting in the capacity as director, chief executive officer or chief financial officer, or
(ii) was subject to an order that was issued after the proposed director ceased to be a director, chief executive officer or chief financial officer and which resulted from an event that occurred while that person was acting in the capacity as director, chief executive officer or chief financial officer.
| 42 |
| b. | a director or executive officer of any corporation (including the Company) that, while that person was acting in that capacity, or within a year of that person ceasing to act in that capacity, became bankrupt, made a proposal under any legislation relating to bankruptcy or insolvency or was subject to or instituted any proceedings, arrangement or compromise with creditors or had a receiver, receiver manager or trustee appointed to hold its assets; or |
| c. | has become bankrupt, made a proposal under any legislation relating to bankruptcy or insolvency, or become subject to or instituted any proceedings, arrangement or compromises with creditors, or had a receiver, manager or trustee appointed to hold the assets of the proposed director. |
For the purposes of (a) above, “order” means a cease trade order, an order similar to a cease trade order or an order that denied the relevant Company access to any exemption under securities legislation, in each case that was in effect for a period of more than 30 consecutive days.
Except as disclosed below and to the knowledge of the Company, none of the current executive officers or directors of the Company has been subject to:
| a. | any penalties or sanctions imposed by a court relating to securities legislation or by a securities regulatory authority or has entered into a settlement agreement with a securities regulatory authority; or |
| b. | any other penalties or sanctions imposed by a court or regulatory body that would likely be considered important to a reasonable security holder in deciding whether to vote for a proposed director. |
Conflicts of Interest
There are no existing or potential material conflicts of interest between the Company or its subsidiary and any director or officer of the Company or its subsidiary.
| IX. | AUDIT COMMITTEE |
The charter of our Audit Committee is attached as Schedule A. The current members of the Audit Committee are Bradley Thompson, Denis Burger and Warren Whitehead. Mr. Warren Whitehead is the Chairman of the Audit Committee and has been considered to be the Financial Expert. Pursuant to Canadian securities laws, the Board has determined that Messrs. Thompson, Burger and Whitehead are financially literate as all have experience in reviewing and analyzing the financial reports and ascertaining the financial position of a corporation. Mr. Burger, in his previous position as Chairman and Chief Executive Officer of AVI Biopharma, is educated and experienced in reading and analyzing financial statements. Mr. Burger has also served on the audit committee of three other publicly listed biotechnology companies. Dr. Thompson has experience reading and interpreting financial statements through his former role as Chairman and CEO of a publicly listed biotechnology company as well as through his extensive experience serving on various company boards. Mr. Whitehead is a CPA (CMA) and has served as the Chief Financial Officer of Arius Research Inc. and Labopharm Inc. Additionally, we believe Mr. Thompson, Mr. Whitehead and Mr. Burger qualify as “independent” as that term is defined in the relevant securities laws relating to the composition of the audit committee.
| 43 |
Audit Committee Mandate
The Audit Committee’s mandate is to assist the Board in fulfilling its oversight responsibilities. In particular, the Audit Committee:
| · | serves as an independent and objective party to monitor the integrity of our financial reporting process and systems of internal controls regarding finance, accounting, and legal compliance, including the review of our consolidated financial statements, MD&A and annual and interim results; |
| · | identifies and monitors the management of the principal risks that could impact our financial reporting; |
| · | monitors the independence and performance of our independent auditors, including the pre-approval of all audit fees and all permitted non-audit services; |
| · | provides an avenue of communication among the independent auditors, management, and the Board; and |
| · | encourages continuous improvement of, and foster adherence to, our policies, procedures and practices at all levels. |
The Audit Committee is also responsible for implementing and overseeing our whistle-blowing procedures.
Ethical Business Conduct
We have adopted a code of ethics which applies to all of our officers, directors, employees and consultants. A copy of the code of ethics is available on our website at www.aptose.com or, without charge, upon written request from our Senior Vice President and chief Financial Officer at our offices located at 5955 Airport Road, Suite #228, Mississauga, Ontario L4V 1R9, Canada.
Pre-Approval Policies and Procedures
The Audit Committee of our Board has, pursuant to the Audit Committee charter, adopted specific responsibilities and duties regarding the provision of services by our external auditor, currently KPMG LLP. KPMG LLP is independent of the Company in accordance with the Rules of Professional Conduct of the Institute of Chartered Professional Accountants of Ontario.
Our charter requires Audit Committee pre-approval of all permitted audit, audit-related and tax services.
Subject to the charter, the Audit Committee may establish fee thresholds for a group of pre-approved services. The Audit Committee then recommends to the Board approval of the fees and other significant compensation to be paid to the independent auditors.
External Auditor Service Fees
The following table summarizes the Audit, Audit Related, Tax Related and Other Fees (excluding expenses and taxes) billed by the Company’s auditor, KPMG LLP to the Company and its subsidiaries for the two most recently completed fiscal years.
| 44 |
| Fees | December 31, 2017 | December 31, 2016 |
| Audit Fees (1) | CA$ 125,000 | CA$ 116,000 |
| Audit Related Fees (2) | CA$ 200,400 | CA$ 211,400 |
| Tax Fees (3) | $- | CA$ 62,150 |
| All Other Fees (4) | $- | $- |
| Total Fees | CA$ 338,400 | CA$ 399,310 |
| (1) | Audit Fees consist of the aggregate fees billed by the external auditor of the Company for audit services. |
| (2) | Audited Related Fees consist of the aggregate fees billed for assurance and related services that are reasonably related to the performance of the audit or review of the issuer’s financial statements and are not reported under “Audit Fees” above and include the provision of comfort letters and consents, the consultation concerning financial accounting and reporting of specific issues and the review of documents filed with regulatory authorities. |
| (3) | Tax Fees include fees billed for tax compliance, tax advice and tax planning services, including the preparation of original tax returns and claims for refund; tax consultations, such as assistance and representation in connection with tax audits and appeals, tax advice related to mergers and acquisitions, and requests for rulings or technical advice from taxing authorities; tax planning services; and consultation and planning services. |
| (4) | All Other Fees include the aggregate fees billed for products and services provided by the auditors, other than the services reported above. |
| X. | Legal Proceedings and Regulatory Actions |
The Company is not a party to any legal proceeding, and its property is not and was not the subject of any material legal proceeding, during the year ended December 31, 2017. The Company is not aware of any legal proceeding outstanding, threatened or pending as of the date hereof by or against the Company.
The Company is not and was not subject to, during the year ended December 31, 2017: (i) penalties or sanctions imposed by a court relating to Canadian securities legislation or by a Canadian securities legislation or by a Canadian securities regulatory authority; (ii) any other penalties or sanctions imposed by a court or regulatory body that would likely be considered important to a reasonable investor in making an investment decision; and (iii) settlement agreements entered into with a court relating to Canadian securities legislation or with a Canadian securities regulatory authority.
| XI. | Interest of Management and Others in Material Transactions |
There are no material interests, direct or indirect, of directors, executive officers, any shareholder who beneficially owns, directly or indirectly, more than 10% of the outstanding Common Shares, or any known associates or affiliates of such persons, in any transaction within the last three years or in any proposed transaction which has materially affected or would materially affect the Company.
| XII. | Transfer Agent and Registrar |
The registrar and transfer agent for the Common Shares is Computershare Investor Services Inc. at its principal office in the City of Toronto.
| 45 |
| XIII. | Material Contracts |
The following are the material contracts, other than contracts entered into in the ordinary course of business, that the Company has entered into since January 1, 2017 or prior thereto but which are still in effect:
| (i) | exclusive global license agreement with OHM that provides OHM with the rights for the development, manufacture and commercialization of APL-581, as well as related molecules from Aptose’s dual bromodomain and extra-terminal domain motif (BET) protein and kinase inhibitor program; |
| (ii) | Purchase Agreement with Aspire Capital Fund, LLC which provides that, upon the terms and subject to the conditions and limitations set forth therein, Aspire Capital is committed to purchase up to an aggregate of $15,500,000 of Common Shares; |
| (iii) | a registration rights agreement entered into among the Company and Aspire Capital dated as of October 27, 2017 pursuant to which the Company provides certain registration rights under the Securities Act of 1933 (United States), as amended, and the rules and regulations thereunder; and |
| (iv) | a definitive agreement entered into among the Company and CG, dated June 1, 2016, granting Aptose an exclusive option to research, develop and commercialize CG’806 in all countries of the world except Korea and China, for all fields of use. |
A copy of these contracts can be found under the profile of the Company on SEDAR at www.sedar.com.
| XIV. | Interest of Experts |
KPMG LLP, the auditor of the Company, is the only person, company or partnership which is named as having prepared or certified a statement, report or valuation described, included or referred to in a filing made by the Company during or relating to the Company’s most recently completed financial year and whose profession or business gives authority to a statement, report or valuation made. The partners and associates of KPMG LLP do not hold any of the issued and outstanding Common Shares.
| XV. | Additional Information |
Additional information, including directors’ and officers’ remuneration and indebtedness, principal holders of our securities, options and to purchase securities and interests of insiders in material transactions, if any, is contained in the Management Information Circular of the Company dated April 18, 2017 prepared in connection with the Company’s most recent annual shareholders’ meeting and is available on SEDAR at www.sedar.com. Additional financial information, including the Company’s audited financial statements and management’s discussion and analysis of financial condition and results of operations, is available on SEDAR at www.sedar.com.
All requests for the above-mentioned documents must be addressed to the Senior Vice President and Chief Financial Officer of Aptose Biosciences Inc., #5955 Airport Road, Suite #228, Mississauga, Ontario L4V 1R9, or by fax at (905) 234-2120.
| 46 |
Schedule A
MANDATE OF THE AUDIT COMMITTEE[1]
| 1. | Purpose |
The primary purposes of the Audit Committee (the “Committee”) of the Board shall be to act on behalf of the Board, in fulfilling the Board’s oversight responsibilities with respect to the Company’s corporate accounting and financial reporting processes, the systems of internal control over financial reporting, and audits of financial statements, as well as the quality and integrity of the Company’s financial statements and reports and the qualifications, independence and performance of the registered public accounting firm or firms engaged as the Company’s independent outside auditors for the purpose of preparing or issuing an audit report or performing other audit, review or attest services (the “Auditors”). The Committee shall also provide oversight assistance in connection with the Company’s legal, regulatory and ethical compliance programs as established by management and the Board. The operation of the Committee shall be subject to the constating documents of the Company as in effect from time to time and applicable law.
The policy of the Committee, in discharging these obligations, shall be to maintain and foster an open avenue of communication among the Committee, the Auditors and the Company’s financial management.
The members of the Committee are not full-time employees of the Company and may or may not be accountants or auditors by profession or experts in the fields of accounting or auditing and, in any event, do not serve in such capacity. Consequently, it is not the duty of the Committee to conduct audits or to determine that the Company’s financial statements and disclosures are complete and accurate and are in accordance with generally accepted accounting principles and applicable rules and regulations. These are the responsibilities of management and the external auditors.
| 2. | Composition |
The Committee shall be comprised of a minimum three directors as determined by the Board. Each of the members of the Committee shall satisfy the independence and financial literacy requirements of any applicable securities laws, securities regulatory authorities and stock exchanges, including without limitation, requirements set out in, or by, National Instrument 52-110 Audit Committee, the Nasdaq Stock Market (“Nasdaq”), the Toronto Stock Exchange (the “TSX”) and the United Stated Securities and Exchange Commission, as in effect from time to time. At least one member shall satisfy the applicable Nasdaq financial sophistication requirements as in effect from time to time.
Committee members shall be appointed by the Board. Members of the Committee shall serve until their resignation or removal. The Board may fill vacancies on the Committee by a majority vote of the authorized numbers of Directors, but may remove Committee members only with the approval of a majority of the independent Directors then serving on the full Board. The Board shall designate a Committee member as the Chair of the Committee on an annual basis, or if the Board does not do so, the Committee members shall appoint a Committee member as Chair by a majority vote of the authorized number of Committee members.
_______________________
[1] As revised and adopted by the Board on March 3, 2015.
| 3. | Meetings, Reports and Resources of the Committee |
(a) Meetings. In discharging its responsibilities, the Committee shall meet as often as it determines necessary or advisable, but not less frequently than quarterly. The Committee may also hold special meetings or act by unanimous written consent as the Committee may decide. The meetings may be in person or telephone. The Chair shall prepare and/or approve an agenda in advance of each meeting. The Committee shall appoint a secretary to be the secretary of each meeting of the Committee to keep written minutes of the meeting and deliberations and will ensure that such minutes are included in the Company’s minute book. The Chair of the Committee shall report at the next regularly scheduled Board meeting following the applicable Committee meeting.
(b) Critical Reporting. The Committee shall report to the Board with respect to material issues that arise regarding the quality or integrity of the Company’s financial statements, the Company’s compliance with legal or regulatory requirements, the performance or independence of the Auditors or such other matters as the Committee deems appropriate from time to time or whenever it shall be called upon to do so. The Committee may invite any person to attend part or all of a meeting of the Committee.
(c) Procedures. The Committee may establish its own procedures, including the formation and delegation of authority to subcommittees, in a manner not inconsistent with this charter, the articles or applicable laws or regulations. The Chair or a majority of the Committee members may call meetings of the Committee. A majority of the members of the Committee constitute a quorum for the transaction of Committee business, and the vote of a majority of the Committee members present at the meeting at which a quorum is present shall be the act of the Committee. The Committee shall review, discuss and assess its own performance at least annually. The Committee shall also periodically review (at least annually) and assess the adequacy of this charter, including the Committee’s role and responsibilities as outlined in this charter, and shall recommend any proposed changes to the Board of its consideration. The Committee may meet in separate sessions with the Auditors, as appropriate, and management and other directors to discuss any matters that the Committee, the Auditors or management believe should be discussed privately with the Committee.
(d) Reports. The Committee shall prepare the report required by the rules of the Securities and Exchange Commission (the “SEC”) to be included in the Company’s annual proxy statement (if the Company is required to file an annual proxy statement pursuant to SEC rules), as well as any other report required of the Committee under applicable laws.
(e) Committee Access and Resources. The Committee shall have authority to appoint, determine compensation for, and at the Company’s expense, retain and oversee the Auditors subject to applicable law and regulations including Section 10A(m)(2) of the United States Securities Exchange Act of 1934, as amended, and the rules thereunder and otherwise to fulfill its responsibilities under this charter. The Committee is at all times authorized to have direct, independent and confidential access to the Company’s other directors, management and personnel to carry out the Committee’s purposes. The Committee is also authorized to retain and terminate at the Company’s expense, independent counsel or other advisers selected by the Committee for matters related to the Committee’s purposes.
| 4. | Authority and Responsibilities |
The Committee shall oversee the Company’s financial reporting process on behalf of the Board, and shall have direct responsibility for the oversight of the work of the Auditors and any other registered public accounting firm engaged for the purpose of performing other review or attest services for the Company. The Auditors and each such other registered public accounting firm shall report directly and be accountable to the Committee. The Committee’s functions and procedures should remain flexible to address most effectively changing circumstances. To implement the Committee’s purpose and policy, the Committee shall be charged with the following functions and processes with the understanding, however, that the Committee may supplement or (except as otherwise required by applicable laws or rules) deviate from these activities as appropriate under the circumstances:
| 2 |
(a) Evaluation and Retention of Auditors. To evaluate the performance of the Auditors, including the lead partner, to assess their qualifications (including their internal quality-control procedures and any material issues raised by that firm’s most recent internal quality-control review or any investigations by regulatory authorities) and to determine whether to recommend to the Board the retention or to termination of the engagement of the existing Auditors or the appointment or engagement of a different independent registered public accounting firm.
(b) Communication Prior to Engagement. Prior to engagement of any prospective Auditors, to review a written disclosure by the prospective Auditors of all relationships between the prospective Auditors, or their affiliates, and the Company, or persons in financial oversight roles at the Company, that may reasonably be thought to bear on independence, and to discuss with the prospective Auditors the potential effects of such relationships on the independence of the prospective Auditors, consistent with applicable laws, regulations and accounting rules.
(c) Approval of Audit Engagements. To determine and recommend to the Board the engagement of the Auditors, prior to commencement of such engagement, to perform all proposed audit, review and attest services, including the scope of and plans for the audit, the adequacy of staffing, to determine and recommend to the Board the compensation to be paid, at the Company’s expense, to the Auditors and the negotiation and execution, on behalf of the Company, of the Auditors’ engagement letters.
(d) Approval of Non-Audit Services. To determine and approve engagements of the Auditors, prior to commencement of such engagements (unless in compliance with exceptions available under applicable laws and rules related to immaterial aggregate amounts of services), to perform any proposed permissible non-audit services, including the scope of the service and the compensation to be paid therefor, at the Company’s expense, which approval may be pursuant to preapproval policies and procedures established by the Committee consistent with applicable laws and rules, including the delegation of preapproval authority to one or more Committee members so long as any such preapproval decisions are presented to the full Committee at the next scheduled meeting.
(e) Audit Partner Rotation. To monitor the rotation of the partners of the Auditors on the Company’s audit engagement team as required by applicable laws and rules and to consider periodically and, if deemed appropriate, adopt a policy regarding rotation of auditing firms.
(f) Auditor Independence. At least annually, consistent with applicable rules and regulations, to receive and review written disclosures from the Auditors delineating all relationships between the Auditors, or their affiliates, and the Company, or persons in financial oversight roles at the Company, that may reasonably be thought to bear on independence and a letter from the Auditors affirming their independence, to consider and discuss with the Auditors any potential effects of any such relationships on the independence of the Auditors as well as any compensation or services that could affect the Auditors’ objectivity and independence, and to assess and otherwise take appropriate action to oversee the independence of the Auditors.
(g) Former Employees of Auditor. To consider and, if deemed appropriate, adopt clear policies regarding Committee preapproval of employment by the Company of individuals employed or formerly employed by the Auditors and engaged on the Company’s account.
| 3 |
(h) Audited Financial Statement Review. To review, upon completion of the audit, the financial statements proposed to be included in the Company’s public disclosure documents, including financial news releases, management’s discussion and analysis, registration statements, annual reports, including on Form 10-K or Form 20-F, as applicable, to be filed on SEDAR and/or with the SEC, management’s discussion and analysis and to recommend whether or not such financial statements and other materials should be approved by the Board for disclosure.
(i) Annual Audit Results. To review with management and the Auditors, the results of the annual audit, including the Auditors’ assessment of the quality, not just acceptability, of the Company’s accounting principles and practices, the Auditors’ views about qualitative aspects of the Company’s significant accounting practices, the reasonableness of significant judgments and estimates (including material changes in estimates), all known and likely misstatements identified during the audit (other than those the Auditors believe to be trivial), the adequacy of the disclosures in the financial statements and any other matters required to be communicated to the Committee by the Auditors under the standards of the applicable accounting rules.
(j) Auditor Communications. At least annually, to discuss with the Auditors the matters required to be discussed by applicable law, regulations and accounting rules.
(k) Quarterly Results. To review and discuss with management and the Auditors, as appropriate, the results of the Auditors’ review of the Company’s quarterly financial statements and approve such quarterly financial statements, prior to public disclosure of quarterly financial information or filing of any required disclosure with any securities regulatory authority, including the filing with the SEC of the Company’s Quarterly Report on Form 10-Q (if required by SEC rules), and to discuss with the Auditors any other matters required to be communicated to the Committee by the Auditors under generally accepted auditing standards, as appropriate.
(l) Management’s Discussion and Analysis. To review and discuss with management and the Auditors, as appropriate, the Company’s disclosures contained under the caption “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in its periodic reports to be filed with the SEC and the disclosure in the “Management’s Discussion and Analysis” to be filed with applicable securities regulatory authorities in Canada.
(m) Press Releases. To review and discuss with management and the Auditors, as appropriate, earnings press releases, and press releases containing information relating to material developments as well as the substance of financial information, information relating to material developments and earnings guidance provided to analysts and ratings agencies, which discussions may be general discussions of the type of information to be disclosed or the type of presentation to be made.
(n) Accounting Principles and Policies. To review with management and the Auditors, as appropriate, significant issues that arise regarding accounting principles and financial statement presentation, including critical accounting policies and practices, alternative accounting policies available under international financial reporting standards, in Canada, and generally accepted accounting principles, in the United States, related to material items discussed with management, the potential impact on the Company’s financial statements of off-balance sheet structures and any other significant reporting issues and judgments, significant regulatory, legal and accounting initiatives or developments that may have a material impact on the Company’s financial statements, compliance programs and policies if, in the judgment of the Committee, such review is necessary or appropriate. To approve, if appropriate, major changes to the Company’s accounting principles and practices as suggested by the independent auditors or management and assure that the reasoning is described in determining the appropriateness of changes in accounting principles and disclosures.
| 4 |
(o) Risk Assessment and Management. To review and discuss with management and, as appropriate, the Auditors the Company’s guidelines and policies with respect to risk assessment and risk management, including the Company’s major financial risk exposures and the steps taken by management to monitor and control these exposures; and to review and discuss with management insurance programs, including director and officer insurance, product liability insurance and general liability insurance (but excluding compensation and benefits-related insurance).
(p) Management Cooperation with Audit. To evaluate the cooperation received by the Auditors during their audit examination, including a review with the Auditors of any significant difficulties encountered during the audit or any restrictions on the scope of their activities or access to required records, data and information and, whether or not resolved, significant disagreements with management and management’s response, if any.
(q) Management Letters. To review and discuss with the Auditors and, if appropriate, management, any management or internal control letter issued or, to the extent practicable, proposed to be issued by the Auditors and management’s response, if any, to such letter, as well as any additional material written communications between the Auditors and management.
(r) National Office Communications. To review and discuss with the Auditors, as appropriate, communications between the audit team and the Auditors’ national office with respect to accounting or auditing issues presented by the engagement.
(s) Disagreements Between Auditors and Management. To review with management and the Auditors, or any other registered public accounting firm engaged to perform review or attest services, any conflicts or disagreements between management and the Auditors, or such other accounting firm, whether or not resolved, regarding financial reporting, accounting practices or policies or other matters, that individually or in the aggregate could be significant to the Company’s financial statements or the Auditors’ report, and to resolve any conflicts or disagreements regarding financial reporting.
(t) Internal Control Over Financial Reporting. To confer with management and the Auditors, as appropriate, regarding the scope, adequacy and effectiveness of internal control over financial reporting including significant deficiencies or material weaknesses identified by the Company’s Auditors. To review with the management and the Auditors any fraud, whether or not material, that includes management or other employees who have any significant role in the Company’s internal control over financial reporting and any significant changes in internal controls or other factors that could significantly affect internal controls, including any corrective actions in regard to significant deficiencies or material weaknesses.
(u) Correspondence with Regulators. To consider and review with management, the Auditors, outside counsel, as appropriate, and any special counsel, separate accounting firm or other consultants and advisors as the Committee deems appropriate, any correspondence with regulators or governmental agencies and any published reports that raise material issues regarding the Company’s financial statements or accounting policies.
(v) Complaint Procedures. To establish procedures, when and as required by applicable laws and rules, for the receipt, retention and treatment of complaints received by the Company regarding accounting, internal accounting controls or auditing matters and the confidential and anonymous submission by employees of concerns regarding questionable accounting or auditing matters, and to establish such procedures as the Committee may deem appropriate for the receipt, retention and treatment of complaints received by the Company with respect to any other matters that may be directed to the Committee for review and assessment.
| 5 |
(w) Ethical Compliance; Compliance with Legal and Regulatory Requirements. To review the results of management’s efforts to monitor compliance with the Company’s programs and policies designed to ensure adherence to applicable laws and rules, as well as to its Code of Business Conduct and Ethics, as amended from time to time, and regarding legal matters and compliance with legal and regulatory requirements that may have a material effect on the Company’s business, financial statements or compliance policies, including any material reports or inquiries from regulatory or governmental agencies. To review with the Company’s counsel, on at least an annual basis, any legal matters that could have a significant impact on the organization’s financial statements and the Company’s compliance with applicable laws and regulations.
(x) Related-Party Transactions. To review and provide oversight of related-party transactions, as required by applicable securities laws and the rules and regulations of applicable securities regulatory authorities and stock exchanges, in accordance with the Company’s Disclosure and Insider Trading Policy and Code of Business Conduct and Ethics.
(y) Engagement of Registered Public Accounting Firms. To determine and recommend to the Board for approval the engagement of any registered public accounting firm (in addition to the Auditors), prior to commencement of such engagement, to perform any other review or attest service, including the recommendation to the Board of the compensation to be paid, at the Company’s expense, to such firm and the negotiation and execution, on behalf of the Company, of such firm’s engagement letter. To discharge such Auditors when circumstances warrant.
(z) Investment Policy. To review, on a periodic basis, as appropriate, the Company’s investment policy and recommend to the Board any changes to the investment policy.
(aa) Investigations. To investigate any matter brought to the attention of the Committee within the scope of its duties if, in the judgment of the Committee, such investigation is necessary or appropriate.
(bb) Hiring Policies of Auditors. To review and approve the Company’s hiring policies with respect to partners, employees and former partners and employees of the current and former Auditors of the Company.
(cc) Disclosure. To describe in the Company’s annual information form the Committee’s composition and responsibilities and how they were discharged.
The approval of this Audit Committee Charter shall be construed as delegation of authority to the Audit Committee with respect to the responsibilities set forth herein.
5