If this is an annual or transition report, indicate by check mark
if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934.
Yes [ ] No [X]
Indicate by check mark whether the registrant: (1) has filed all
reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or such shorter period that
the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Yes [X] No [ ]
Indicate by check mark whether the registrant is a large
accelerated filer, an accelerated filer, or a non-accelerated filer. See definition of “accelerated filer and large accelerated filer” in
Rule 12b-2 of the Exchange Act.
Large accelerated filer [ ] Accelerated
filer [X] Non-accelerated filer [ ]
Indicate by check mark which financial statement item the
registrant has elected to follow.
Item 17 [X] Item 18 [
]
If this is an annual report, indicate by check mark whether the
registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
Yes [ ] No [X]
TABLE OF CONTENTS
| Page |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
PART I |
|||||||||||
ITEM
1. |
IDENTITY OF DIRECTORS, SENIOR MANAGEMENT AND ADVISORS |
2 | |||||||||
ITEM
2. |
OFFER STATISTICS AND EXPECTED TIMETABLE |
2 | |||||||||
ITEM
3. |
KEY
INFORMATION |
2 | |||||||||
ITEM
4. |
INFORMATION ON THE COMPANY |
14 | |||||||||
ITEM
4A. |
UNRESOLVED STAFF COMMENTS |
30 | |||||||||
ITEM
5. |
OPERATING AND FINANCIAL REVIEW AND PROSPECTS |
30 | |||||||||
ITEM
6. |
DIRECTORS, SENIOR MANAGEMENT AND EMPLOYEES |
41 | |||||||||
ITEM
7. |
MAJOR SHAREHOLDERS AND RELATED PARTY TRANSACTIONS |
51 | |||||||||
ITEM
8. |
FINANCIAL INFORMATION |
52 | |||||||||
ITEM
9. |
THE
OFFER AND LISTING |
52 | |||||||||
ITEM
10. |
ADDITIONAL INFORMATION |
53 | |||||||||
ITEM
11. |
QUALITATIVE AND QUANTITATIVE DISCLOSURES ABOUT MARKET RISK |
63 | |||||||||
ITEM
12. |
DESCRIPTION OF SECURITIES OTHER THAN EQUITY SECURITIES |
64 | |||||||||
PART II |
|||||||||||
ITEM
13. |
DEFAULTS, DIVIDENDS, ARREARAGES AND DELINQUENCIES |
64 | |||||||||
ITEM
14. |
MATERIAL MODIFICATIONS TO THE RIGHTS OF SECURITY HOLDERS AND USE OF PROCEEDS |
64 | |||||||||
ITEM
15. |
CONTROLS AND PROCEDURES |
64 | |||||||||
ITEM
16. |
[RESERVED] |
65 | |||||||||
ITEM
16A. |
AUDIT COMMITTEE FINANCIAL EXPERT |
65 | |||||||||
ITEM
16B. |
CODE
OF ETHICS |
65 | |||||||||
ITEM
16C. |
PRINCIPAL ACCOUNTANT FEES AND SERVICES |
65 | |||||||||
ITEM
16D. |
EXEMPTIONS FROM THE LISTING STANDARDS FOR AUDIT COMMITTEES |
65 | |||||||||
ITEM
16E. |
PURCHASES OF EQUITY SECURITIES BY THE ISSUER AND AFFILIATED PURCHASERS |
66 | |||||||||
PART III |
|||||||||||
ITEM
17. |
FINANCIAL STATEMENTS |
66 | |||||||||
ITEM
18. |
FINANCIAL STATEMENTS |
66 | |||||||||
ITEM
19. |
EXHIBITS |
66 | |||||||||
General
In this Annual Report on Form 20-F, all
references to “Lorus”, the “Company”, “we”, “us” and “our” each refers to Lorus Therapeutics
Inc. and its subsidiaries. References to this “Form 20-F” and this “Annual Report” mean references to this Annual Report on Form
20-F for the year ended May 31, 2006.
We use the Canadian dollar as our
reporting currency. All references in this Annual Report to “dollars” or “$” are expressed in Canadian dollars, unless otherwise
indicated. See also “Item 3. Key Information” for more detailed currency and conversion information. Our consolidated financial statements
which form part of the annual report are presented in Canadian dollars and are prepared in accordance with accounting principles generally accepted in
Canada (“Canadian GAAP”) which differ in certain respects from accounting principles generally accepted in the United States (“U.S.
GAAP”). The differences between Canadian GAAP and U.S. GAAP, as they relate to our business, are explained in the notes to our consolidated
financial statements.
Special note regarding
forward-looking statements
This annual report on Form 20-F may
contain forward-looking statements within the meaning of Canadian and U.S. securities laws. Such statements include, but are not limited to, statements
relating to:
• |
our expectations regarding future financings; |
• |
our plans to conduct clinical trials; |
• |
our expectations regarding the progress and the successful and timely completion of the various stages of our drug discovery, preclinical and clinical studies and the regulatory approval process; |
• |
our plans to obtain partners to assist in the further development of our product candidates; and |
• |
our expectations with respect to existing and future corporate alliances and licensing transactions with third parties, and the receipt and timing of any payments to be made by us or to us in respect of such arrangements, and |
the Company’s plans, objectives, expectations and
intentions and other statements including words such as “anticipate”, “contemplate”, “continue”, “believe”,
“plan”, “estimate”, “expect”, “intend”, “will”, “should”, “may”, and other
similar expressions.
Such statements reflect our current
views with respect to future events and are subject to risks and uncertainties and are necessarily based upon a number of estimates and assumptions
that, while considered reasonable by us are inherently subject to significant business, economic, competitive, political and social uncertainties and
contingencies. Many factors could cause our actual results, performance or achievements to be materially different from any future results,
performance, or achievements that may be expressed or implied by such forward-looking statements, including, among others:
• |
our ability to obtain the substantial capital required to fund research and operations; |
• |
our lack of product revenues and history of operating losses; |
• |
our early stage of development, particularly the inherent risks and uncertainties associated with (i) developing new drug candidates generally, (ii) demonstrating the safety and efficacy of these drug candidates in clinical studies in humans, and (iii) obtaining regulatory approval to commercialize these drug candidates; |
• |
our drug candidates require time-consuming and costly preclinical and clinical testing and regulatory approvals before commercialization; |
• |
clinical studies and regulatory approvals of our drug candidates are subject to delays, and may not be completed or granted on expected timetables, if at all, and such delays may increase our costs and could delay our ability to generate revenue; |
• |
the regulatory approval process; |
• |
the progress of our clinical trials; |
• |
our ability to find and enter into agreements with potential partners; |
• |
our ability to attract and retain key personnel; |
• |
our ability to obtain patent protection and protect our intellectual property rights; |
1
• |
our ability to protect our intellectual property rights and to not infringe on the intellectual property rights of others; |
• |
our ability to comply with applicable governmental regulations and standards; |
• |
development or commercialization of similar products by our competitors, many of which are more established and have greater financial resources than we do; |
• |
commercialization limitations imposed by intellectual property rights owned or controlled by third parties; |
• |
our business is subject to potential product liability and other claims; |
• |
our ability to maintain adequate insurance at acceptable costs; |
• |
further equity financing may substantially dilute the interests of our shareholders; |
• |
changing market conditions; and |
• |
other risks detailed from time-to-time in our ongoing quarterly filings, annual information forms, annual reports and annual filings with Canadian securities regulators and the United States Securities and Exchange Commission, and those which are discussed under Item 3.D. “Risk Factors”. |
Should one or more of these risks or
uncertainties materialize, or should the assumptions set out in the section entitled “Risk Factors” underlying those forward-looking
statements prove incorrect, actual results may vary materially from those described herein. These forward-looking statements are made as of the date of
this annual report on Form 20-F or, in the case of documents incorporated by reference herein, as of the date of such documents, and we do not intend,
and do not assume any obligation, to update these forward-looking statements, except as required by law. We cannot assure you that such statements will
prove to be accurate as actual results and future events could differ materially from those anticipated in such statements. Investors are cautioned
that forward-looking statements are not guarantees of future performance and accordingly investors are cautioned not to put undue reliance on
forward-looking statements due to the inherent uncertainty therein.
PART I
Item
1. |
Identity of Directors, Senior Management and Advisors |
Not applicable.
Item
2. |
Offer Statistics and Expected Timetable |
Not applicable.
Item
3. |
Key Information |
A. |
Selected Financial Data |
The following tables present our
selected consolidated financial data. You should read these tables in conjunction with our audited consolidated financial statements and accompanying
notes included in Item 17 of this annual report on Form 20-F and the “Management’s Discussion and Analysis of Financial Condition and Results
of Operations” included in Item 5 of this annual report on Form 20-F.
The financial data as at May 31, 2006,
2005, 2004, 2003 and 2002 and for the years ended May 31, 2006, 2005, 2004, 2003 and 2002 have been derived from, and are qualified in their entirety
by reference to, our audited consolidated financial statements, which have been prepared in accordance with Canadian Generally Accepted Accounting
Principles (Canadian GAAP) and reconciled to United States Generally Accepted Accounting Principles (US GAAP) in note 17 to the consolidated financial
statements.
The following table presents a summary
of our consolidated statement of operations derived from our audited financial statements for the years ended May 31, 2006, 2005, 2004, 2003 and
2002.
2
Consolidated statements of operations data:
(In thousands, except per share data)
(In thousands, except per share data)
| |
Years Ended May 31, |
|
|||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
20061 |
|
20051 |
|
20041 |
|
20031 |
|
20021 |
|
Period From Inception2 |
||||||||||||||||
In accordance
with Canadian GAAP |
|||||||||||||||||||||||||||
Revenue |
$ | 26 | $ | 6 | $ | 608 | $ | 66 | $ | — | $ | 706 | |||||||||||||||
Research and
development |
$ | 10,237 | $ | 14,394 | $ | 26,785 | $ | 12,550 | $ | 8,659 | $ | 110,475 | |||||||||||||||
General and
administrative |
$ | 4,334 | $ | 5,348 | $ | 4,915 | $ | 4,290 | $ | 4,867 | $ | 47,475 | |||||||||||||||
Operating
expenses |
$ | 16,550 | $ | 21,782 | $ | 32,148 | $ | 17,855 | $ | 15,482 | $ | 173,610 | |||||||||||||||
Net
loss |
$ | 17,909 | $ | 22,062 | $ | 30,301 | $ | 16,634 | $ | 13,487 | $ | 164,552 | |||||||||||||||
Basic and
diluted loss per share |
$ | 0.10 | $ | 0.13 | $ | 0.18 | $ | 0.12 | $ | 0.09 | |||||||||||||||||
In accordance
with US GAAP |
|||||||||||||||||||||||||||
Net loss3 |
$ | 16,388 | $ | 20,298 | $ | 30,301 | $ | 16,634 | $ | 13,487 | |||||||||||||||||
Basic and
diluted loss per share |
$ | 0.09 | $ | 0.12 | $ | 0.18 | $ | 0.12 | $ | 0.09 | |||||||||||||||||
The following table presents a summary of our consolidated
balance sheet as at May 31, 2006, 2005, 2004, 2003 and 2002.
Consolidated balance sheet data:
| (In Thousands) |
|
As at May 31, |
|
||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
20061 |
|
20051 |
|
20041 |
|
20031 |
|
20021 |
||||||||||||||
In accordance
with Canadian GAAP |
|||||||||||||||||||||||
Cash and cash
equivalents |
$ | 2,692 | $ | 2,776 | $ | 1,071 | $ | 905 | $ | 1,165 | |||||||||||||
Short-term
investments |
$ | 5,627 | $ | 18,683 | $ | 25,657 | $ | 24,219 | $ | 36,657 | |||||||||||||
Prepaid expenses
and other assets |
$ | 515 | $ | 1,126 | $ | 1,697 | $ | 1,104 | $ | 1,195 | |||||||||||||
Total
assets |
$ | 11,461 | $ | 27,566 | $ | 34,424 | $ | 34,255 | $ | 47,572 | |||||||||||||
Total
debt |
$ | 14,017 | $ | 14,300 | $ | 5,825 | $ | 5,360 | $ | 3,432 | |||||||||||||
Total
shareholders’ equity |
$ | (2,556 | ) | $ | 13,266 | $ | 28,599 | $ | 28,895 | $ | 44,140 | ||||||||||||
Weighted average
number of common shares outstanding |
173,523 | 172,112 | 171,628 | 144,590 | 143,480 | ||||||||||||||||||
Dividends paid
on common shares |
— | — | — | — | — | ||||||||||||||||||
In accordance
with US GAAP3 |
|||||||||||||||||||||||
Total
assets |
$ | 11,625 | $ | 27,838 | $ | 34,424 | $ | 34,255 | $ | 47,572 | |||||||||||||
Total
debt |
$ | 17,277 | $ | 18,040 | $ | 5,825 | $ | 5,360 | $ | 3,432 | |||||||||||||
Total
shareholders’ equity |
$ | (5,652 | ) | $ | 9,798 | $ | 28,599 | $ | 28,895 | $ | 44,140 | ||||||||||||
| 1 |
Changes in accounting polices: | |
(a) |
Stock based compensation: Effective June 1, 2004, the Company adopted the fair value method of accounting for stock options granted to employees on or after June 1, 2002 as required by the amended CICA Handbook Section 3870, Stock-Based Compensation and Other Stock-Based Payments. The change was adopted retroactively without restatement as permitted under the revised section. |
3
Under the fair value method, the estimated fair value of stock
options granted is recognized over the service period, that is the applicable vesting period, as stock compensation expense and a credit to stock
options. When options granted on or after June 1, 2002 are exercised, the proceeds received and the related amounts in stock options are credited to
share capital. When options granted prior to June 1, 2002 are exercised, the proceeds are credited to share capital. The impact to the financial
statements arising from adoption of the fair value method was an increase to the deficit and stock option balances presented in shareholders equity of
$2.8 million at June 1, 2004. We recorded stock based compensation expense under Canadian GAAP of $1.2 million for the year ended May 31, 2006 (2005
– $1.5 million, 2004 – nil).
(b) Business combinations, goodwill and other intangibles:
Goodwill represents the excess of the purchase price over the fair value of net identifiable assets acquired in the GeneSense business combination, and
until June 1, 2002, was amortized on a straight-line basis over three years. In August 2001, the CICA issued Handbook Sections 1581, “Business
Combinations”, and 3062, “Goodwill and Other Intangible Assets”. The new standards required that the purchase method of accounting must
be used for business combinations and require that goodwill no longer be amortized but instead be tested for impairment at least annually. The
standards also specify criteria that intangible assets must meet to be recognized and reported apart from goodwill. The new standards were
substantially consistent with U.S. GAAP.
The Company adopted these new standards as of June 1, 2002 and
the Company discontinued amortization of all existing goodwill. The Company also evaluated existing intangible assets, including estimates of remaining
useful lives in accordance with the provisions of the standard.
In connection with Section 3062’s transitional goodwill
impairment evaluation, the Company assessed whether goodwill was impaired as of June 1, 2002. The Company completed the transitional goodwill
impairment assessment during the first quarter of 2003 and determined that no impairment existed at the date of adoption.
The Company recorded amortization of goodwill in the amount of
$1.5 million for the year ended May 31, 2002.
2 |
Period from inception September 5, 1986 to May 31, 2006 |
3 |
The significant differences between the line items under Canadian
GAAP and those as determined under U.S. GAAP arise primarily from: There were no significant differences between Canadian and US GAAP during the years ended May 2002, 2003 and 2004. |
2005:
Convertible debentures
Convertible debentures
Under Canadian GAAP, the conversion
option embedded in the convertible debentures is presented separately as a component of shareholders’ equity. Under U.S. GAAP, the embedded
conversion option is not subject to bifurcation and is thus presented as a liability along with the balance of the convertible debentures. Under U.S.
GAAP, Emerging Issues Task Force No.00-19 and APB Opinion No. 14, the fair value of warrants issued in connection with the convertible debentures
financing would be recorded as a reduction to the proceeds from the issuance of convertible debentures, with the offset to additional paid-in capital.
The warrants have been presented as a separate component of shareholders’ equity for Canadian GAAP purposes. Under U.S. GAAP the Company has
allocated the total proceeds received from the issuance of the convertible debentures to the debt and warrant portions based on their relative fair
values. The fair value of the purchase warrants has been determined based on an option-pricing model. The resulting allocation based on relative fair
values resulted in the allocation of $13.9 million to the debt instrument and $1.1 million to the purchase warrants. The financing fees totaling $1.1
million related to the issuance of the convertible debentures have been allocated pro rata between deferred financing charges of $964 thousand and
against the purchase warrants of $97 thousand. This allocation resulted in the net amount allocated to the warrants of $1.0 million. The financing
charges are being amortized over the five-year life of the convertible debentures agreement.
Each reporting period, the Company is
required to accrete the carrying value of the convertible debentures such that at maturity on October 6, 2009, the carrying value of the debentures
will be their face value of $15.0 million.
4
To date, the Company has recognized $97,000 in accretion expense. This accretion expense has increased the value of the convertible debenture from $13.9 million to $14.0 million at May 31, 2005.
Stock
options
Effective June 1, 2004, the Company
adopted the fair value based method of accounting for employee stock options granted on or after June 1, 2002, retroactively without restatement as
allowed under the transitional provisions of CICA Handbook Section 3870. As a result, the opening balances of deficit and stock options were increased
by $2.8 million at June 1, 2004. During 2005, the Company recorded stock compensation expense of $1.5 million in the consolidated statement of
operations, representing the amortization applicable to the current year at the estimated fair value of options granted since June 1, 2002; and an
offsetting adjustment to stock options of $1.5 million in the consolidated balance sheets. No similar adjustments are required under U.S. GAAP as the
Company has elected to continue measuring compensation expense, as permitted under SFAS No. 123, using the intrinsic value based method of accounting
for stock options. Under this method, compensation is the excess, if any, of the quoted market value of the stock at the date of the grant over the
amount an employee must pay to acquire the stock. Election of this method requires pro-forma disclosure of compensation expense as if the fair value
method has been applied for awards granted in fiscal periods after December 15, 1994.
The company grants Performance Based
Stock Options as a compensation tool. Under Canadian GAAP, the fair value treatment of these options is consistent with all other employee stock
options. Under US GAAP, the option is treated as a variable award and is revalued, using the intrinsic value method of accounting, at the end of each
reporting period until the final measurement date. Due to the decline in our share price during the year, there was no expense recorded for US GAAP
purposes.
2006:
Convertible debentures
Convertible debentures
Disclosure is consistent with
2005.
To date, the Company has recognized $407
thousand in accretion expense. This accretion expense has increased the value of the convertible debentures from $13.9 million to $14.3 million at May
31, 2006.
Stock
options
Effective June 1, 2004, the Company
adopted the fair value based method of accounting for employee stock options granted on or after June 1, 2002, retroactively without restatement as
allowed under the transitional provisions of CICA Handbook Section 3870. As a result, the opening balances of deficit and stock options were increased
by $2.8 million at June 1, 2004. During 2006, the Company recorded stock compensation expense in the consolidated financial statements, representing
the amortization applicable to the current year at the estimated fair value of stock options granted since June 1, 2002.
During 2006, the Company recorded stock
compensation expense of $1.2 million (2005 – $1.5 million) in the consolidated statement of operations, representing the amortization applicable
to the current year at the estimated fair value of options granted since June 1, 2002; and an offsetting adjustment to stock options of $1.2 million in
the consolidated balance sheets. No similar adjustments are required under U.S. GAAP as the Company has elected to continue measuring compensation
expense, as permitted under SFAS No. 123, using the intrinsic value based method of accounting for stock options. Under this method, compensation is
the excess, if any, of the quoted market value of the stock at the date of the grant over the amount an employee must pay to acquire the stock.
Election of this method requires pro-forma disclosure of compensation expense as if the fair value method has been applied for awards granted in fiscal
periods after December 15, 1994.
The Company grants performance based
stock options as a compensation tool. Under Canadian GAAP, the accounting treatment of these options is consistent with all other employee stock
options. Under US GAAP, the option is treated as a variable award and is revalued, using the intrinsic value method of accounting, at the end of each
reporting period until the final measurement date. At each reporting date, compensation cost is measured based on an estimate of the number of options
that will vest considering the performance criteria and the difference between the market price of the underlying stock and the exercise price at such
dates. The compensation cost is being recognized over the estimated performance period. For the year ended May 31, 2006 the Company recorded stock
based compensation expense of $20 thousand under U.S. GAAP for performance based options.
5
During 2006, employees of the Company
(excluding directors and officers) were given the opportunity to choose between keeping 100% of their existing options at the existing exercise price
and forfeiting 50% of the options held in exchange for having the remaining 50% of the exercise price of the options re-priced to $0.30 per share.
Employees holding 2,290,000 stock options opted for re-pricing their options, resulting in the amendment of the exercise price of 1,145,000 stock
options and the forfeiture of 1,145,000 stock options. Under Canadian GAAP the accounting treatment of these options requires that any incremental
value resulting from the amendment be determined and recognized over the remaining vesting period. Under US GAAP, the amended options are treated as a
variable award and are revalued, using the intrinsic value method of accounting at the end of each reporting period until the date the options are
exercised, forfeited or expired unexercised. The Company recorded stock-based compensation of $36 thousand under US GAAP related to these amended stock
options.
Refer to note 17 of the consolidated
financial statements in Item 17 for further details.
Exchange
Rate Information
We publish our consolidated financial statements in Canadian
(“CDN”) dollars. In this report, except where otherwise indicated, all amounts are stated in CDN dollars.
The following table sets out the exchange rates of CDN$ for 1 US$
for the following periods:
| Period |
Average Close |
High |
Low |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
October,
2006 |
1.1283 | 1.1415 | 1.1146 | |||||||||||
September,
2006 |
1.1165 | 1.1289 | 1.1035 | |||||||||||
August,
2006 |
1.1189 | 1.1373 | 1.104 | |||||||||||
July,
2006 |
1.1296 | 1.145 | 1.105 | |||||||||||
June,
2006 |
1.1139 | 1.129 | 1.0963 | |||||||||||
May,
2006 |
1.1093 | 1.1132 | 1.1055 | |||||||||||
Fiscal Year
Ended May 31, 2006 |
1.1701 | 1.246 | 1.0948 | |||||||||||
Fiscal Year
Ended May 31, 2005 |
1.2551 | 1.378 | 1.1746 | |||||||||||
Fiscal Year
Ended May 31, 2004 |
1.3423 | 1.418 | 1.2683 | |||||||||||
Fiscal Year
Ended May 31, 2003 |
1.5245 | 1.601 | 1.3438 | |||||||||||
Fiscal Year
Ended May 31, 2002 |
1.5697 | 1.618 | 1.5069 | |||||||||||
B. |
Capitalization and Indebtedness |
Not applicable.
C. |
Reasons for the Offer and Use of Proceeds |
Not applicable.
D. |
Risk Factors |
Before making an investment decision
with respect to our common shares, you should carefully consider the following risk factors, in addition to the other information included or
incorporated by reference in this annual report on Form 20-F. Additional risks not currently known by us or that we consider immaterial at the present
time may also impair our business, financial condition, prospects or results of operations. If any of the following risks occur, our business,
financial condition, prospects or results of operations would likely be affected. In that case, the trading price of our common shares could decline
and you may lose all or part of the money you paid to buy our common shares. The risks set out below are not the only we currently face; other risks
may arise in the future.
6
We have a history of operating losses. We expect to
incur net losses and we may never achieve or maintain profitability.
We have not been profitable since our
inception in 1986. We reported net losses of $17.9 million; $22.1 million and $30.3 million for the years ended May 31, 2006, 2005 and 2004,
respectively. As of May 31, 2006, we had an accumulated deficit of $164.5 million.
To date we have only generated nominal
revenues from the sale of Virulizin® in Mexico and we stopped selling Virulizin® in Mexico in July 2005. We have not generated any other
revenue from product sales to date and it is possible that we will never have sufficient product sales revenue to achieve profitability. We expect to
continue to incur losses for at least the next several years as we or our collaborators and licensees pursue clinical trials and research and
development efforts. To become profitable, we, either alone or with our collaborators and licensees, must successfully develop, manufacture and market
our current product candidates, particularly Virulizin® and GTI-2040, as well as continue to identify, develop, manufacture and market new product
candidates. It is possible that we will never have significant product sales revenue or receive significant royalties on our licensed product
candidates. If funding is insufficient at any time in the future, we may not be able to develop or commercialize our products, take advantage of
business opportunities or respond to competitive pressures.
Our current and anticipated operations,
particularly our product development, requires substantial capital. We expect that our existing cash and cash equivalents, along with the funds
available to us through the subscription agreements with High Tech and Technifund described elsewhere in this Annual Report, will sufficiently fund our
current and planned operations through at least the next twelve months. However, our future capital needs will depend on many factors, including the
extent to which we enter into collaboration agreements with respect to any of our proprietary product candidates, receive royalty and milestone
payments from our possible collaborators and make progress in our internally funded research and development activities.
Our capital requirements will also
depend on the magnitude and scope of these activities, our ability to maintain existing and establish new collaborations, the terms of those
collaborations, the success of our collaborators in developing and marketing products under their respective collaborations with us, the success of our
contract manufacturers in producing clinical and commercial supplies of our product candidates on a timely basis and in sufficient quantities to meet
our requirements, competing technological and market developments, the time and cost of obtaining regulatory approvals, the extent to which we choose
to commercialize our future products through our own sales and marketing capabilities, the cost of preparing, filing, prosecuting, maintaining and
enforcing patent and other rights and our success in acquiring and integrating complementary products, technologies or companies. We do not have
committed external sources of funding and we cannot assure you that we will be able to obtain additional funds on acceptable terms, if at all. If
adequate funds are not available, we may be required to:
• |
engage in equity financings that would be dilutive to current shareholders; |
• |
delay, reduce the scope of or eliminate one or more of our development programs; |
• |
obtain funds through arrangements with collaborators or others that may require us to relinquish rights to technologies, product candidates or products that we would otherwise seek to develop or commercialize ourselves; or |
• |
license rights to technologies, product candidates or products on terms that are less favorable to us than might otherwise be available. |
We may be unable to obtain partnerships for one or
more of our product candidates, which could curtail future development and negatively impact our share price.
Our product candidates require
significant funding to reach regulatory approval upon positive clinical results. Such funding, in particular for Virulizin®, will be very
difficult, or impossible to raise in the public markets. As such, the Company must obtain partnerships to continue the development of certain product
candidates. If such partnerships are not attainable, the development of these product candidates may be significantly delayed or stopped altogether.
The announcement of such delay or discontinuation of development may have a negative impact on our share price.
7
In addition, our strategy for the
research, development and commercialization of our products requires entering into various arrangements with corporate collaborators, licensers,
licensees and others, and our commercial success is dependent upon these outside parties performing their respective contractual responsibilities. The
amount and timing of resources that such third-parties will devote to these activities may not be within our control. We cannot assure you that such
parties will perform their obligations as expected. We also cannot assure you that our collaborators will devote adequate resources to our programs. In
addition, we could become involved in disputes with our collaborators, which could result in a delay or termination of the related development programs
or result in litigation. We intend to seek additional collaborative arrangements to develop and commercialize some of our products. We may not be able
to negotiate collaborative arrangements on favorable terms, or at all, in the future, or that our current or future collaborative arrangements will be
successful.
If we cannot negotiate collaboration,
licence or partnering agreements, we may never achieve profitability.
Clinical trials are long, expensive and uncertain
processes and Health Canada or the FDA may ultimately not approve any of our product candidates. We may never develop any commercial drugs or other
products that generate revenues.
None of our products has received
regulatory approval for commercial use and sale in North America. We cannot market a pharmaceutical product in any jurisdiction until it has completed
thorough preclinical testing and clinical trials in addition to that jurisdiction’s extensive regulatory approval process. In general, significant
research and development and clinical studies are required to demonstrate the safety and effectiveness of our products before we can submit any
regulatory applications.
Clinical trials are long, expensive and
uncertain processes. Clinical trials may not be commenced or completed on schedule, and Health Canada (“HC”) or the FDA may not ultimately
approve our product candidates for commercial sale. Further, even if the results of our preclinical studies or clinical trials are initially positive,
it is possible that we will obtain different results in the later stages of drug development or that results seen in clinical trials will not continue
with longer term treatment. Drugs in late stages of clinical development may fail to show the desired safety and efficacy traits despite having
progressed through initial clinical testing. For example, positive results in early Phase I or Phase II clinical trials may not be repeated in larger
Phase II or Phase III clinical trials. A number of companies in the pharmaceutical industry, including us, have suffered setbacks in advanced clinical
trials, even after promising results in earlier clinical trials. The results of our Phase III clinical trial of Virulizin® did not meet the
primary endpoint of the study despite promising preclinical and early stage clinical data. All of our potential drug candidates are prone to the risks
of failure inherent in drug development.
Preparing, submitting and advancing
applications for regulatory approval is complex, expensive and time intensive and entails significant uncertainty. The results of our completed
preclinical studies and clinical trials may not be indicative of future clinical trial results. A commitment of substantial resources to conduct
time-consuming research, preclinical studies and clinical trials will be required if we are to complete development of our products. Clinical trials of
our products require that we identify and enrol a large number of patients with the illness under investigation. We may not be able to enrol a
sufficient number of appropriate patients to complete our clinical trials in a timely manner particularly in smaller indications such as acute myeloid
leukemia. If we experience difficulty in enrolling a sufficient number of patients to conduct our clinical trials, we may need to delay or terminate
ongoing clinical trials and will not accomplish objectives material to our success that could affect the price of our common shares. Delays in planned
patient enrolment or lower than anticipated event rates in our current clinical trials or future clinical trials may result in increased costs, program
delays, or both.
In addition, unacceptable toxicities or
adverse side effects may occur at any time in the course of preclinical studies or human clinical trials or, if any products are successfully developed
and approved for marketing, during commercial use of any approved products. The appearance of any such unacceptable toxicities or adverse side effects
could interrupt, limit, delay or abort the development of any of our product candidates or, if previously approved, necessitate their withdrawal from
the market. Furthermore, disease resistance or other unforeseen factors may limit the effectiveness of our potential products.
The clinical trials of any of our drug
candidates could be unsuccessful, which would prevent us from advancing, commercializing or partnering the drug.
8
Even if we receive approval to market
any product from any regulatory authorities on the basis of successful clinical studies of that product, following the market introduction of that
product we or others may discover safety and efficacy problems not observed in the clinical studies. In this respect, as a condition to granting
approval to market any of our products or at any time after granting such approval, one or more regulatory authorities may require us to conduct
further studies (referred to as “Phase IV studies”) to determine the safety and efficacy of the product following market introduction. If
such problems arise, one or more regulatory authorities may withdraw the approval for that product or we may otherwise voluntarily withdraw the product
from the market.
Despite the time and resources expended
by us, regulatory approval of drug candidates is never guaranteed. If any of our development programs are not successfully completed in a timely
fashion, required regulatory approvals are not obtained in a timely fashion, or products for which approvals are obtained are not commercially
successful or are ultimately found to not be safe or effective, our business could be seriously harmed.
Our failure to develop safe,
commercially viable drugs would substantially impair our ability to generate revenues and sustain our operations and would materially harm our business
and adversely affect our share price. We may never achieve profitability.
As a result of intense
competition and technological change in the pharmaceutical industry, the marketplace may not accept our products or product candidates, and we may not
be able to compete successfully against other companies in our industry and achieve profitability.
Many of our competitors have drug
products that have already been approved or are in development, and operate large, well-funded research and development programs in these fields. Many
of our competitors have substantially greater financial and management resources, stronger intellectual property positions and greater manufacturing,
marketing and sales capabilities, areas in which we have limited or no experience. In addition, many of our competitors have significantly greater
experience than we do in undertaking preclinical testing and clinical trials of new or improved pharmaceutical products and obtaining required
regulatory approvals. Consequently, our competitors may obtain HC, FDA and other regulatory approvals for product candidates sooner and may be more
successful in manufacturing and marketing their products than we or our collaborators are. Existing and future products, therapies and technological
approaches will compete directly with the products we seek to develop. Current and prospective competing products may provide greater therapeutic
benefits for a specific problem or may offer easier delivery or comparable performance at a lower cost. Any product candidate that we develop and that
obtains regulatory approval must then compete for market acceptance and market share. Our product candidates may not gain market acceptance among
physicians, patients, healthcare payers and the medical community. Further, any products we develop may become obsolete before we recover any expenses
we incurred in connection with the development of these products. As a result, we may never achieve profitability.
If we fail to attract and retain key employees, the
development and commercialization of our products may be adversely affected.
We depend heavily on the principal
members of our scientific and management staff. If we lose any of these persons, our ability to develop products and become profitable could suffer.
The risk of being unable to retain key personnel may be increased by the fact that we have not executed long term employment contracts with our
employees, except for our senior executives. Our future success will also depend in large part on our ability to attract and retain other highly
qualified scientific and management personnel. We face competition for personnel from other companies, academic institutions, government entities and
other organizations.
We may be unable to obtain patents to protect our
technologies from other companies with competitive products, and patents of other companies could prevent us from manufacturing, developing or
marketing our products.
Patent
protection
The patent positions of pharmaceutical
and biotechnology companies are uncertain and involve complex legal and factual questions. The United States (U.S.) Patent and Trademark Office and
many other patent offices in the world have not established a consistent policy regarding the breadth of claims that it will allow in
biotechnology
9
patents. Further, allowable patentable subject matter and the scope of patent protection obtainable may differ as between jurisdictions. If a patent office allows broad claims, the number and cost of patent interference proceedings in the U.S. or analogous proceedings in other jurisdictions and the risk of infringement litigation may increase. If a patent office allows narrow claims, the risk of infringement may decrease, but the value of our rights under our patents, licenses and patent applications may also decrease. In addition, the scope of the claims in a patent application can be significantly modified during prosecution before the patent is issued. Consequently, we cannot know whether our pending applications will result in the issuance of patents or, if any patents are issued, whether they will provide us with significant proprietary protection or will be circumvented, invalidated or found to be unenforceable. Until recently, patent applications in the U.S. were maintained in secrecy until the patents issued, and publication of discoveries in scientific or patent literature often lags behind actual discoveries. Patent applications filed in the United States after November 2000 generally will be published 18 months after the filing date unless the applicant certifies that the invention will not be the subject of a foreign patent application. In many other jurisdictions, such as Canada, patent applications are published 18 months from the priority date. We cannot assure you that, even if published, we will be aware of all such literature. Accordingly, we cannot be certain that the named inventors of our products and processes were the first to invent that product or process or that we were the first to file or pursue patent coverage for our inventions.
Enforcement of intellectual
property rights
Our commercial success depends in part
on our ability to maintain and enforce our proprietary rights. If third-parties engage in activities that infringe our proprietary rights, our
management’s focus will be diverted and we may incur significant costs in asserting our rights. We may not be successful in asserting our
proprietary rights, which could result in our patents being held invalid or a court holding that the third-party is not infringing, either of which
would harm our competitive position. In addition, we cannot assure you that others will not design around our patented technology. Moreover, we may
have to participate in interference proceedings declared by the U.S. Patent and Trademark Office, European opposition proceedings, or other analogous
proceedings in other parts of the world to determine priority of invention and the validity of patent rights granted or applied for, which could result
in substantial cost and delay, even if the eventual outcome is favorable to us. We cannot assure you that our pending patent applications, if issued,
would be held valid or enforceable. Additionally, many of our foreign patent applications have been published as part of the patent prosecution process
in such countries.
Trademark
protection
Protection of the rights revealed in
published patent applications can be complex, costly and uncertain. In order to protect goodwill associated with our company and product names, we rely
on trademark protection for our marks. For example, we have registered the Virulizin® trademark with the U.S. Patent and Trademark Office. A
third-party may assert a claim that the Virulizin® mark is confusingly similar to its mark and such claims or the failure to timely register the
Virulizin® mark or objections by the FDA could force us to select a new name for Virulizin®, which could cause us to incur additional
expense.
Trade
secrets
We also rely on trade secrets, know-how
and confidentiality provisions in our agreements with our collaborators, employees and consultants to protect our intellectual property. However, these
and other parties may not comply with the terms of their agreements with us, and we might be unable to adequately enforce our rights against these
people or obtain adequate compensation for the damages caused by their unauthorized disclosure or use. Our trade secrets or those of our collaborators
may become known or may be independently discovered by others.
Our products and product candidates may infringe the
intellectual property rights of others, which could increase our costs.
Our success also depends on avoiding
infringement of the proprietary technologies of others. In particular, there may be certain issued patents and patent applications claiming subject
matter that we or our collaborators may be required to license in order to research, develop or commercialize at least some of our product candidates,
including Virulizin®, GTI-2040, GTI-2501 and small molecules. In addition, third-parties may assert infringement
10
or other intellectual property claims against us based on our patents or other intellectual property rights. An adverse outcome in these proceedings could subject us to significant liabilities to third-parties, require disputed rights to be licensed from third-parties or require us to cease or modify our use of the technology. If we are required to license such technology, we cannot assure you that a license under such patents and patent applications will be available on acceptable terms or at all. Further, we may incur substantial costs defending ourselves in lawsuits against charges of patent infringement or other unlawful use of another’s proprietary technology.
If product liability claims are brought against us or
we are unable to obtain or maintain product liability insurance, we may incur substantial liabilities that could reduce our financial
resources.
The clinical testing and commercial use
of pharmaceutical products involves significant exposure to product liability claims. We have obtained limited product liability insurance coverage for
our clinical trials on humans; however, our insurance coverage may be insufficient to protect us against all product liability damages. Further,
liability insurance coverage is becoming increasingly expensive and we might not be able to obtain or maintain product liability insurance in the
future on acceptable terms or in sufficient amounts to protect us against product liability damages. Regardless of merit or eventual outcome, liability
claims may result in decreased demand for a future product, injury to reputation, withdrawal of clinical trial volunteers, loss of revenue, costs of
litigation, distraction of management and substantial monetary awards to plaintiffs. Additionally, if we are required to pay a product liability claim,
we may not have sufficient financial resources to complete development or commercialization of any of our product candidates and our business and
results of operations will be adversely affected.
We have no manufacturing capabilities. We depend on
third parties, including a number of sole suppliers, for manufacturing and storage of our product candidates used in our clinical trials. Product
introductions may be delayed or suspended if the manufacture of our products is interrupted or discontinued.
We do not have manufacturing facilities
to produce supplies of Virulizin®, GTI-2040, GTI-2501, small molecule or any of our other product candidates to support clinical trials or
commercial launch of these products, if they are approved. We are dependent on third parties for manufacturing and storage of our product candidates.
If we are unable to contract for a sufficient supply of our product candidates on acceptable terms, or if we encounter delays or difficulties in the
manufacturing process or our relationships with our manufacturers, we may not have sufficient product to conduct or complete our clinical trials or
support preparations for the commercial launch of our product candidates, if approved.
Dependence on contract manufacturers
for commercial production involves a number of risks, many of which are outside our control. These risks include potential delays in transferring
technology, and the inability of our contract manufacturer to scale production on a timely basis, to manufacture commercial quantities at reasonable
costs, to comply with cGMP and to implement procedures that result in the production of drugs that meet our specifications and regulatory
requirements.
Our reliance on contract manufacturers
exposes us to additional risks, including:
• |
there may be delays in scale-up to quantities needed for clinical trials and commercial launch or failure to manufacture such quantities to our specifications, or to deliver such quantities on the dates we require; |
• |
our current and future manufacturers are subject to ongoing, periodic, unannounced inspection by the FDA and corresponding Canadian and international regulatory authorities for compliance with strictly enforced cGMP regulations and similar standards, and we do not have control over our contract manufacturers’ compliance with these regulations and standards; |
• |
our current and future manufacturers may not be able to comply with applicable regulatory requirements, which would prohibit them from manufacturing products for us; |
• |
if we need to change to other commercial manufacturing contractors, the FDA and comparable foreign regulators must approve these contractors prior to our use, which would require new testing and compliance inspections, and the new manufacturers would have to be educated in, or themselves develop substantially equivalent processes necessary for the production or our products; and |
11
• |
our manufacturers might not be able to fulfill our commercial needs, which would require us to seek new manufacturing arrangements and may result in substantial delays in meeting market demand. |
Any of these factors could cause us to
delay or suspend clinical trials, regulatory submission, required approvals or commercialization of our products under development, entail higher costs
and result in our being unable to effectively commercialize our products. We do not currently intend to manufacture any of our product candidates,
although we may choose to do so in the future. If we decide to manufacture our products, we would be subject to the regulatory risks and requirements
described above. We would also be subject to similar risks regarding delays or difficulties encountered in manufacturing our pharmaceutical products
and we would require additional facilities and substantial additional capital. We cannot assure you that we would be able to manufacture any of our
products successfully in accordance with regulatory requirements and in a cost effective manner.
Our operations involve hazardous materials and we
must comply with environmental laws and regulations, which can be expensive and restrict how we do business.
Our research and development activities
involve the controlled use of hazardous materials, radioactive compounds and other potentially dangerous chemicals and biological agents. Although we
believe our safety procedures for these materials comply with governmental standards, we cannot entirely eliminate the risk of accidental contamination
or injury from these materials. We currently have insurance, in amounts and on terms typical for companies in businesses that are similarly situated,
that could coverall or a portion of a damage claim arising from our use of hazardous and other materials. However, if an accident or environmental
discharge occurs, and we are held liable for any resulting damages, the associated liability could exceed our insurance coverage and our financial
resources.
We have limited sales, marketing and distribution
experience.
We have very limited experience in the
sales, marketing and distribution of pharmaceutical products. There can be no assurance that we will be able to establish sales, marketing, and
distribution capabilities or make arrangements with our collaborators, licensees or others to perform such activities or that such efforts will be
successful. If we decide to market any of our products directly, we must either acquire or internally develop a marketing and sales force with
technical expertise and with supporting distribution capabilities. The acquisition or development of a sales and distribution infrastructure would
require substantial resources, which may divert the attention of our management and key personnel and have a negative impact on our product development
efforts. If we contract with third-parties for the sales and marketing of our products, our revenues will be dependent on the efforts of these
third-parties, whose efforts may not be successful. If we fail to establish successful marketing and sales capabilities or to make arrangements with
third-parties, our business, financial condition and results of operations will be materially adversely affected.
Our interest income is subject to fluctuations of
interest rates in our investment portfolio.
Our investments are held to maturity
and have staggered maturities to minimize interest rate risk. There can be no assurance that interest income fluctuations will not have an adverse
impact on our financial condition. We maintain all our accounts in Canadian dollars, but a portion of our expenditures are in foreign currencies. We do
not currently engage in hedging our foreign currency requirements to reduce exchange rate risk.
Because of the uncertainty of pharmaceutical pricing,
reimbursement and healthcare reform measures, if any of our product candidates are approved for sale to the public, we may be unable to sell our
products profitably.
The availability of reimbursement by
governmental and other third-party payers affects the market for any pharmaceutical product. These third-party payers continually attempt to contain or
reduce the costs of healthcare. There have been a number of legislative and regulatory proposals to change the healthcare system and further proposals
are likely. Significant uncertainty exists with respect to the reimbursement status of newly approved healthcare products. In addition, third-party
payers are increasingly challenging the price and cost effectiveness of medical products and services. We might not be able to sell our products
profitably or recoup the value of our investment in product development if reimbursement is unavailable or limited in scope.
12
Risks Related To Our Common Shares and Convertible
Debentures
Our share price has been and may continue to be volatile and an investment in our common shares could suffer a decline in
value.
You should consider an investment in
our common shares as risky and invest only if you can withstand a significant loss and wide fluctuations in the market value of your investment. We
receive only limited attention by securities analysts and frequently experience an imbalance between supply and demand for our common shares. The
market price of our common shares has been highly volatile and is likely to continue to be volatile. Factors affecting our common share price
include:
• |
announcements concerning the results or clinical trials for our drug candidates; |
• |
the progress of our and our collaborators’ clinical trials, including our and our collaborators’ ability to produce clinical supplies of our product candidates on a timely basis and in sufficient quantities to meet our clinical trial requirements; |
• |
announcements of technological innovations or new product candidates by us, our collaborators or our competitors; |
• |
announcements concerning our competitors or the life sciences industry in general; |
• |
fluctuations in our operating results; |
• |
published reports by securities analysts; |
• |
developments in patent or other intellectual property rights; |
• |
publicity concerning discovery and development activities by our licensees; |
• |
the cash and short term investments held us and our ability to secure future financing; |
• |
public concern as to the safety and efficacy of drugs that we and our competitors develop; |
• |
governmental regulation and changes in medical and pharmaceutical product reimbursement policies; and |
• |
general market conditions. |
Future sales of our common shares by us or by our
existing shareholders could cause our share price to fall.
Additional equity financings or other
share issuances by us could adversely affect the market price of our common shares. Sales by existing shareholders of a large number of shares of our
common shares in the public market and the sale of shares issued in connection with strategic alliances, or the perception that such additional sales
could occur, could cause the market price of our common shares to drop.
Our cash flow may not be sufficient to cover interest
payments on our secured convertible debentures or to repay the debentures at maturity.
Our ability to make interest payments,
if required to be paid in cash, and to repay at maturity or refinance our prime rate +1% convertible debentures due in 2009 will depend on our ability
to generate sufficient cash or refinance them. We have never generated positive annual cash flow from our operating activities, and we may not generate
or sustain positive cash flows from operations in the future. Our ability to generate sufficient cash flow will depend on our ability, or the ability
of our strategic partners, to successfully develop and obtain regulatory approval for new products and to successfully market these products, as well
as the results of our research and development efforts and other factors, including general economic, financial, competitive, legislative and
regulatory conditions, many of which are outside of our control.
Conversion of our secured convertible debentures will
dilute the ownership interest of existing shareholders.
The conversion of some or all of the
convertible debentures will dilute the ownership interests of existing shareholders. Any sales in the public market of the common shares issuable upon
such conversion could adversely affect prevailing market prices of our common shares. In addition, the existence of the secured convertible debentures
may encourage short selling by market participants.
13
We may violate one or more of the operational
covenants related to our convertible debentures that could result in an event of default and the requirement for early payment of our convertible
debentures.
Our convertible debentures are subject
to certain operational covenants. In the event that one of those covenants is breached by us, an event of default could be declared requiring the
immediate payment of the face value of the debentures. This could result in our inability to pay and insolvency of the Company, a dilutive equity
financing in attempt to raise funds to repay the debentures, or a significant reduction in cash available for us to use towards the development of our
product candidates.
Item
4. |
Information on the Company |
A. |
History and development of the Company |
Lorus Therapeutics Inc. was
incorporated under the Business Corporations Act (Ontario) on September 5, 1986 under the name RML Medical Laboratories Inc. On October 28,
1991, RML Medical Laboratories Inc. amalgamated with Mint Gold Resources Ltd., resulting in the Company becoming a reporting issuer (as defined under
applicable securities law) in Ontario, on such date. On August 25, 1992, the Company changed its name to IMUTEC Corporation. On November 27, 1996, the
Company changed its name to Imutec Pharma Inc., and on November 19, 1998, the Company changed its name to Lorus Therapeutics Inc. On October 1, 2005
the Company continued under the Canada Business Corporations Act.
The address of the Company’s head
and registered office is 2 Meridian Road, Toronto, Ontario, Canada, M9W 4Z7. Our corporate website is www.lorusthera.com. The contents of the website
are specifically not included in this 20-F by reference.
Our common shares are listed on the
Toronto Stock Exchange (the “TSX”) under the symbol “LOR” and are listed on the American Stock Exchange (the “AMEX”)
under the symbol “LRP”.
Lorus’ subsidiaries are GeneSense
Technologies Inc. (“GeneSense”), a corporation incorporated under the laws of Canada, of which Lorus owns 100% of the issued and outstanding
share capital, and NuChem Pharmaceuticals Inc. (“NuChem”), a corporation incorporated under the laws of Ontario, of which Lorus owns 80% of
the issued and outstanding voting share capital and 100% of the issued and outstanding non-voting preference share capital.
We are a life sciences company focused
on the research and development of effective anticancer development stage therapies with high safety. We believe that we have established a diverse
anticancer product pipeline, with products in various stages of development ranging from pre-clinical compounds to multiple ongoing Phase II clinical
trials. A growing intellectual property portfolio supports this product pipeline.
Our commercial success is dependent
upon several factors, including establishing the efficacy and safety of our products in clinical trials, obtaining the necessary regulatory approvals
to enable us to market any products that may be approved and maintaining sufficient levels of funding through public and/or private
financing.
We have product candidates in three
classes of anticancer therapies: (i) immunotherapy, based on macrophage-stimulating biological response modifiers; (ii) antisense therapies, based on
synthetic segments of DNA designed to bind to the messenger RNA (mRNA) that is responsible for the production of proteins over-expressed in cancer
cells; and (iii) small molecule therapies based on anti-angiogenic, anti-proliferative and anti-metastatic agents. In addition, we have a number of
anticancer technologies in the research and pre-clinical stages of development, including tumour suppressor gene therapy, siRNA and U-Sense
technology.
Over the past three years, we have
focused on advancing our product candidates through pre-clinical and clinical testing. You should be aware that it can cost millions of dollars and
take many years before a product candidate may be approved for therapeutic use in humans. In addition, a product candidate may not meet the end points
of any Phase I, Phase II or Phase III clinical trial. See Item 3.D. “Risk Factors”.
14
Immunotherapy
Lorus’ immunotherapy product
candidates are Virulizin® and IL-17E.
Virulizin®
In 2002, we initiated a phase III
clinical trial of Virulizin® for patients with locally advanced or metastatic pancreatic cancer who had not previously received systemic
chemotherapy. In July of 2005, we announced the completion of the study and in October 2005, we announced that the results of the trial indicated that
the overall survival rate of patients who were treated with Virulizin® plus gemcitabine (a standard chemotherapy drug) was not statistically
significant when compared to those patients in the study who were given gemcitabine plus a placebo. We are currently seeking partners to continue the
clinical development of Virulizin®. See “ — Clinical Development” and “Business of the Company —
Immunotherapy”.
IL-17E
We have recently discovered a new lead
drug candidate, IL-17E, which belongs to a larger family of cytokines. In experiments with mice, IL-17E has demonstrated significant antitumour
activity against a variety of human tumours, including melanoma, pancreatic, colon, lung and ovarian tumours. We believe that these preliminary animal
results support our further investigation of the potential clinical applications of IL-17E.
Antisense
We have two lead antisense products,
GTI-2040 and GTI-2501, and other antisense molecules in pre-clinical development.
GTI-2040
Seven of the eight clinical studies
initiated for GTI-2040 have been conducted in conjunction with the United States National Cancer Institute (“NCI”) and the remaining study
was conducted by Lorus. We have initiated, are conducting or have conducted Phase II clinical trials of GTI-2040 in patients with refractory or
relapsed acute myeloid leukemia, metastatic breast cancer, non-small cell lung cancer, solid tumours, advanced unresectable colon cancer, hormone
refractory prostate cancer, advanced, end-stage renal cell cancer, and high grade myelodysplastic syndrome and acute myeloid leukemia.
GTI-2501
Our other antisense therapy, GTI-2501,
is currently in a Phase II clinical trial for the treatment of hormone refractory prostate cancer at the Toronto Sunnybrook Regional Cancer Centre,
following the successful conclusion of a Phase I clinical trial in the United States. See “ — Clinical Development” and “Business
of the Company — Antisense”.
Other
We have entered into a collaboration
agreement in respect of our antisense therapy, GTI-2601 and have other antisense molecules in pre-clinical development. See “Business of the
Company — Antisense”.
Small Molecule
Our small molecule program is in the
pre-clinical stage. See “— Clinical Development” and “Business of the Company — Small Molecule
Therapies”.
Clinical Development
The chart below illustrates our current
view of the clinical development stage of each of our products. This chart reflects the current regulatory approval process for biopharmaceuticals in
Canada and the United States (with
15
the exception of Virulizin® for malignant melanoma, which is approved for use in the private market in Mexico). See “Regulatory Requirements” for a description of the regulatory approval process in Canada and the United States. These qualitative estimates of the progress of our products are intended solely for illustrative purposes and the information contained herein is qualified in its entirety by the information appearing elsewhere or incorporated by reference in this annual report.
CLINICAL DEVELOPMENT PIPELINE
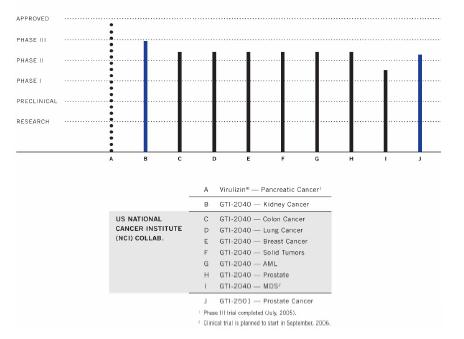
16
PRECLINICAL DEVELOPMENT PIPELINE
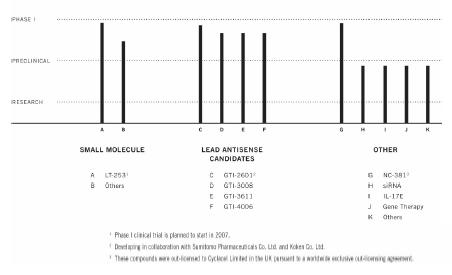
Capital Expenditures
N/A
Capital Commitments
N/A
B. |
Business Overview |
Overview
Chemotherapeutic drugs have been the
predominant medical treatment option for cancer, particularly metastatic cancer, for the past 30 years. More recently, a range of novel cancer drugs
have been developed that are efficacious while improving patient quality of life. Unlike chemotherapies, which are typically based on chemical
synthesis, these new drugs may be of biological origin, based on naturally occurring molecules, proteins or genetic material. While chemotherapy drugs
are relatively non-specific and, as a result, toxic to normal cells, these biological agents specifically target individual molecules or genes that are
involved in disease and are therefore preferentially toxic to tumour cells. The increased specificity of these drugs may result in fewer and milder
side effects, meaning that, in theory, larger and therefore, more effective doses can be administered.
We believe that the future of cancer
treatment and management lies in drugs that are effective, safe and have minimal side effects leading to improved quality of life for patients. Many of
the drugs currently approved for the treatment and management of cancer are toxic, resulting in severe side effects that limit dosing and efficacy. We
believe that a product development plan based on effective and safe drugs would have broad applications in cancer treatment. Lorus’ strategy is to
continue the development of our product pipeline using several therapeutic approaches. Each therapeutic approach is dependent on different
technologies, which we believe mitigates the
17
development risks associated with a single technology platform. In developing and evaluating our products, we evaluate the merits of each product candidate throughout the clinical trial process and consider commercialization opportunities.
Immunotherapy
Introduction
Immunotherapy is a form of treatment
that stimulates the body’s immune system to fight diseases including cancer. Immunotherapy may help the immune system to fight cancer by improving
recognition of differences between healthy cells and cancer cells. Alternatively, it may stimulate the production of specific cancer fighting
cells.
Virulizin®
Virulizin®, Lorus’
immunotherapeutic drug, has been shown in pre-clinical studies to be an immunotherapy that stimulates monocytes and macrophages to infiltrate tumour
tissue and attack tumour cells. Monocytes and macrophages are types of white blood cells that are key players in the immune response to foreign
pathogens and tumour cells. When macrophages and monocytes are activated, they produce proteins called cytokines that have the ability to kill tumour
cells directly. Our studies indicate that Virulizin® stimulates the release of tumour necrosis factor (TNF-alpha), one type of cytokine, in immune
cells to induce apoptosis (programmed cell death) of tumour cells. Our studies also indicate that Virulizin® produces fewer negative side effects
than commonly used chemotherapy agents likely because the drug works by stimulating the immune system to attack the cancer, rather than directly
killing cancerous cells.
Clinical Development
Program
In 2002, Lorus initiated a Phase III,
double-blind, multicenter, randomized study in patients with locally advanced or metastatic pancreatic cancer who had not previously received systemic
chemotherapy. This clinical trial was conducted at over 100 sites in North America and Europe with enrolment of 436 patients with advanced pancreatic
cancer. Patients enrolled in the study were randomly selected to receive treatment with either: (i) Virulizin® plus gemcitabine or (ii) placebo
plus gemcitabine. Optional second line therapy for those patients who failed to respond or became resistant to gemcitabine included Virulizin® or
placebo, alone or in combination with 5-fluorouracil (“5-FU”). All study subjects were monitored throughout the remainder of their lifespan.
The end points of the study were survival and clinical benefits. In July 2005, Lorus announced completion of “last patient visit” for the
phase III trial. Lorus announced the results of the phase III trial in October 2005 and those results are discussed in detail below.
Clinical Trial
Results
In October 2005, we released the
results of the Phase III clinical trial evaluating Virulizin® for the treatment of pancreatic cancer. The primary end points of the study were not
met. For the efficacy evaluable population, the study showed that the addition of Virulizin® to gemcitabine resulted in a median overall survival
of 6.8 months and a one-year survival rate of 27.2%, compared to 6.0 months and 16.8% for placebo plus gemcitabine. In the intent to treat population
the median overall survival rates were 6.3 months for Virulizin® plus gemcitabine (one year survival rate of 25.9%) compared to 6.0 months for
placebo plus gemcitabine (one year survival rate of 17.6%). While comparison of the median overall survival times did not reach statistical
significance, exploratory analysis did show promising trends in specific patient populations.
We are currently seeking partners to
continue the clinical development of Virulizin® in these patient specific populations.
Orphan
Drug
Lorus received Orphan Drug designation
from the FDA in February 2001 for Virulizin® in the treatment of pancreatic cancer. Orphan drug status is awarded to drugs used in the treatment
of a disease that afflicts less than
18
200,000 patients annually in the United States to encourage research and testing. This status means that the FDA will help to facilitate the drug’s development process by providing financial incentives and granting seven years of market exclusivity in the United States (independent of patent protection) upon approval of the drug in the United States. In June 2005, we announced that Virulizin® was granted Orphan Drug status in the European Union for pancreatic cancer.
IL-17E
Lorus has recently discovered a new
lead drug candidate, IL-17E, which belongs to a larger family of cytokines. In experiments with mice, IL-17E has demonstrated significant antitumour
activity against a variety of human tumours, including melanoma, pancreatic, colon, lung and ovarian tumours. Lorus believes that these preliminary
animal results support its further investigation of the potential clinical applications of IL-17E.
Antisense
Introduction
Metabolism, cell growth and cell
division are tightly controlled by complex protein signalling pathways in response to specific conditions, thereby maintaining normal function. Many
human diseases, including cancer, can be traced to faulty protein production and/or regulation. As a result, traditional therapeutics are designed to
interact with the disease-causing proteins and modify their function. A significant number of current anticancer drugs act by damaging either DNA or
proteins within cells (e.g., chemotherapy) or by inhibiting the function of proteins or small molecules (e.g., estrogen blockers, such as
Tamoxifen). Antisense therapeutics offer a novel approach to treatment in that they are designed to prevent the production of proteins causing
disease.
The premise of this therapeutic
approach is to target an earlier stage of the biochemical process than is usually possible with conventional drugs. The blueprint for protein
production is encoded in the DNA of each cell. To translate this code into protein, the cell first produces mRNAs (messenger ribonucleic acids)
specific to each protein and these act as intermediaries between the information encoded in DNA and production of the corresponding protein. Most
traditional therapies interact with the final synthesized or processed protein. Often this interaction lacks specificity that would allow for
interaction with only the intended target, resulting in undesired side effects. In contrast, this newer approach alters gene-expression at the mRNA
level, prior to protein synthesis, with specificity such that expression of only the intended target is affected. We believe that drugs based on this
approach may have broad applicability, greater efficacy and fewer side effects than conventional drugs.
We have developed a number of antisense
drugs, of which our lead products are GTI-2040 and GTI-2501. These products target the two components of ribonucleotide reductase (“RNR”).
RNR is a highly regulated, cell cycle-controlled protein required for DNA synthesis and repair. RNR is made up of two components, R1 and R2, encoded by
different genes. RNR is essential for the formation of deoxyribonucleotides, which are the building blocks of DNA. Since RNR activity is highly
elevated in tumour cell populations and is associated with tumour cell proliferation, we have developed antisense molecules specific for the mRNA of
the R1 (GTI-2501) or the R2 (GTI-2040) components of RNR. Furthermore, the R2 component also appears to be a signal molecule in cancer cells and its
elevation is believed to modify a biochemical pathway that can increase the malignant properties of tumour cells. Consequently, reducing the expression
of the RNR components in a tumour cell with antisense drugs is expected to have antitumour effects.
19
GTI-2040
Our lead antisense therapy is GTI-2040,
an antisense drug that targets the R2 component of RNR and has exhibited antitumour properties against over a dozen different human cancers in standard
mouse models, including chemotherapy resistant tumours. We have recently completed a Phase II clinical trial of GTI-2040 for advanced or metastatic
renal cell carcinoma. We have also commenced a multiple Phase II clinical trial program in cooperation with the NCI, for the study of GTI-2040 for the
treatment of acute myeloid leukemia (“AML”), breast cancer, lung cancer, colon cancer, prostate cancer and a series of solid
tumours.
Pre-clinical
Testing
Formal pre-clinical development of
GTI-2040, including manufacturing and toxicology studies, was initiated in mid-1998. Pre-clinical studies, including GLP toxicology studies in standard
animal models, have demonstrated that GTI-2040 is well tolerated at concentrations that exceed commensurate therapeutic doses in
humans.
Clinical
Development
Our clinical development for GTI-2040
has been done in conjunction with the NCI, which pays for the cost of all clinical trials. See “— Agreements — Collaboration Agreements
— National Cancer Institute”. To date, we have initiated seven clinical trials with the NCI for GTI-2040 in patients with AML, metastic
breast cancer, non-small all lung cancer, solid tumours, unresectable colon cancer, hormone refractory prostate cancer, and high grade myelodysplastic
syndrome (“MDS”) and AML. These indications were selected based on the most promising results from our preclinical studies. In addition,
Lorus conducted a study for GTI-2040 for the treatment of patients with renal cell cancer. Upon receipt of the clinical data from the ongoing NCI
clinical trials, we will analyze and make decisions regarding the strategic direction of our antisense portfolio. We continue to search for
partnerships for the future development of GTI-2040.
In September 2005, Lorus announced a
steering committee assessment of progress in the six ongoing U.S. NCI-sponsored clinical studies of GTI-2040. The committee concluded that all six
studies continue to progress without unacceptable toxicity. Combination chemotherapies under study include docetaxel, capecitabine, oxaliplatin,
cytarabine, and gemcitabine.
Acute Myeloid Leukemia
In July 2003, we announced the
FDA’s approval of the NCI-sponsored IND application for a clinical trial of GTI-2040 in combination with cytarabine, in patients with refractory
or relapsed acute myeloid leukemia. Cytarabine is the current established drug for treating AML patients.
In December 2005, we announced interim
data from the NCI-sponsored trial of GTI-2040 in acute myeloid leukemia. The data presented showed complete responses in 44% of patients 60 years of
age or younger. Patients in this trial had either failed to respond to prior therapy or had rapidly relapsed and as such had a low expectation of
response to subsequent treatment (10-20%). Complete responses in the clinical trial directly correlated with down regulation of R2, the intracellular
target of GTI-2040, demonstrating drug specificity and providing strong evidence for an antisense mechanism of action. Toxicities for the combination
were comparable to those expected for cytarabine alone and were non dose-limiting. This study is ongoing.
Metastatic Breast
Cancer
In August 2003, we announced that the
FDA had approved the NCI’s IND to begin a Phase II clinical trial to investigate GTI-2040 as a treatment for metastatic breast cancer in
combination with capecitabine. This study is ongoing.
20
Non-Small Cell Lung
Cancer
In September 2003, we received approval
from HC for initiation of a clinical trial of GTI-2040 in combination with docetaxel for the treatment of advanced non-small cell lung cancer, as part
of a Phase II clinical program of GTI-2040 in collaboration with the NCI. Interim results from this study were announced in May 2005. Our interim
results showed that the toxicity profile was determined to be acceptable for the specific combination therapy and the observed level of disease
stabilizations was encouraging given the advanced stage of the disease in this subset of patients. This study is ongoing.
Solid Tumours
In February 2004, we announced the
initiation of a Phase II clinical trial examining the use of GTI-2040 in combination with gemcitabine in patients with solid tumours. In June 2005,
results from the trial were published. The trial was intended to identify the recommended dose of GTI-2040 and its toxicity profile. At the recommended
dose GTI-2040 demonstrated a manageable toxicity profile and was generally well tolerated when given as a single agent. This study is
ongoing.
Unresectable Colon
Cancer
In May 2004, we announced the
initiation of a Phase II clinical trial examining GTI-2040 in combination with oxaliplatin and capecitabine in the treatment of advanced unresectable
colon cancer. This study is ongoing.
Hormone Refractory Prostate
Cancer
In November 2004, we announced the
initiation of a Phase II clinical trial examining GTI-2040 in combination with docetaxel and prednisone in hormone refractory prostate cancer. In
November 2005, we announced interim data from this trial. The data showed that along with an acceptable tolerability profile, nine of 22 PSA evaluable
patients demonstrated a PSA response (reductions of greater than 50%). PSA is overproduced in prostate cancer cells and is commonly used to assess
disease progression and response.
Advanced Renal Cell
Cancer
In April 2005, we announced completion
of a Phase II clinical trial of GTI-2040 in combination with capecitabine, in patients with advanced, end-stage renal cell cancer in the United States.
This trial was a single-arm pilot study examining the safety and efficacy of GTI-2040 used in combination with the anticancer agent capecitabine. The
majority of patients had failed two or more prior therapies before entering the study, exhibited extensive metastases, and were representative of a
population with very poor prognostic outcome in renal cell cancer. All 33 patients entering this study had advanced disease with multiple metastatic
sites, with or without prior removal of the primary kidney tumour. However, more than half (52%) of the patients on the recommended dose exhibited
disease stabilization or better, including one confirmed partial response. GTI-2040 was well tolerated when combined with a cytotoxic agent with
expected adverse events. Lorus is actively searching for partnerships to assist with the further development of GTI-2040 for the treatment of renal
cell cancer.
High Grade Myelodysplastic Syndrome and
AML
In June 2006, we announced a plan for a
new clinical investigation of GTI-2040 as a single agent in patients with high grade myelodysplastic syndrome (MDS) and acute myeloid leukemia (AML)
sponsored by the NCI.
Orphan Drug Status
On March 12, 2003, the FDA awarded
Orphan Drug Status to GTI-2040 for the treatment of renal cell carcinoma.
In May 2005, Lorus received Orphan Drug
designation from the FDA for GTI-2040 in the treatment of AML.
21
GTI-2501
Our other antisense therapy is
GTI-2501.
Pre-clinical
Testing
GTI-2501 has demonstrated antitumour
activity in a wide range of human cancers in standard mouse models including human breast, kidney and prostate cancers. Pre-clinical studies have
demonstrated that GTI-2501 is well tolerated in standard animal models at concentrations that exceed commensurate therapeutic doses in
humans.
Clinical Development
Program
GLP-toxicology studies for GTI-2501
were completed in November 2000 and approval of an IND was received from the FDA in February 2001. This Phase I dose-escalating study at the University
of Chicago Medical Centre was designed to establish the recommended clinical Phase II dose as well as look at the safety profile of GTI-2501. A total
of 34 patients with solid tumours or lymphoma were enrolled and have been evaluated following clinical completion. In December 2003, we announced that
a Phase II clinical trial for the treatment of hormone refractory prostate cancer (HRPC) had been initiated at the Toronto Sunnybrook Regional Cancer
Centre, in which GTI-2501 is administered in combination with docetaxel. The combination of GTI-2501 and docetaxel in this clinical trial is being
investigated in patients with asymptomatic or symptomatic HRPC where disease progression is uncontrolled. This represents the first clinical trial of
GTI-2501 in Canada following the successful conclusion of the Phase I clinical trial in 2004 in the United States. We announced expansion of this
ongoing HRPC trial to two additional sites in Canada in July 2004. The study is ongoing through 2006.
GTI-2601
On April 5, 2005 we announced that we
had signed a collaboration agreement with one of Japan’s leading pharmaceutical companies, Sumitomo Pharmaceuticals Co. Ltd.
(“Sumitomo”) and Koken Co. Ltd (“Koken”) with respect to GTI-2601, our lead antisense compound targeting thioredoxin, a gene that
is over-expressed in many tumour tissues and has been correlated with poor prognosis and chemotherapy resistance. Sumitomo and Koken have developed an
advanced delivery system based on collagen combined with macromolecules. The collaboration agreement provides that Sumitomo and Koken will further
develop their delivery technology to combine with GTI-2601, so that increased efficacy is provided with decreased doses of the antisense drug. This
agreement provides that Lorus, Sumitomo and Koken will jointly own the compounds that result from this collaboration (Lorus will share the results of
the collaboration with Sumitomo and Koken, 1:1). This collaboration continued in 2005 and into 2006.
Small Molecule Therapies
Most anticancer chemotherapeutic
treatments are DNA damaging, cytotoxic agents, designed to act on rapidly dividing cells. Treatment with these drugs typically includes unpleasant or
even serious side effects due to the inability of these drugs to differentiate between normal and cancer cells and/or due to a lack of high specificity
for the targeted protein. In addition, these drugs often lead to the development of tumour-acquired drug resistance. As a result of these limitations,
a need exists for more effective anticancer drugs. One approach is to develop small molecules with a greater specificity as anticancer drugs. Chemical
compounds weighing less than 1000 daltons (a unit of molecular weight) are designated as small or low molecular weight molecules. These molecules can
be designed to target specific proteins or receptors that are known to be involved with disease.
Low Molecular Weight Compounds
In August 2005, Lorus announced the
selection of two leading small molecule compounds from a series of novel small molecules discovered by Lorus scientists that exhibit potent anticancer
activity. The results of the further characterization of these compounds were presented in April 2006, including studies that showed that the main
mechanism of action of these compounds involves the induction of the tumour suppressor Krüppel-like factor 4, which its down-regulation is
believed to be critical in the development and progression of certain types of cancer
22
and comprise a novel anticancer mechanism of action. From these two compounds, LT-253 was selected as the lead compound for developments as a drug candidate for the treatment of colon carcinoma and non-small cell lung cancer, based on its potent in vitro and in vivo efficacy in xenograft models of human cancer, and on its safety profile. Manufacturing of a GMP product, formulation development as well as formal toxicology studies in different animal species with the aim of filing an IND application for the initiation of a Phase I clinical trial are in progress.
Other Technologies
We are currently assessing several new
technologies for their potential as new drug candidates. They include technologies in areas of tumour suppressor gene therapy, siRNA molecules
targeting RNR and U-sense compounds that we believe to have the potential to work using a unique mechanism of action to decrease the expression of
cancer relevant genes.
Gene Therapy
Researchers at Lorus have developed a
gene therapy product using the R1 gene of ribonucleotide reductase (which has been shown to act as a tumour suppressor gene) encoded in a modified
adenoviral vector (rAd5-R1) for the potential treatment of patients with colon cancer. This project is in the pre-clinical phase of
development.
siRNA
In 2003, Lorus began development of an
anticancer therapeutic based on siRNA-mediated inhibition of R2 expression. Early screening experiments have identified lead siRNA’s and
preliminary in vitro and in vivo characterization of these molecules has yielded promising results.
U-sense
Lorus has a therapeutic platform based
on short oligonucleotides that are identical to sequences in the untranslated regions of mRNA molecules. The binding of these oligonucleotides to
factors (i.e., proteins) that would otherwise bind to the mRNA has the potential to affect translation and/or stability of the mRNA and as a
result alter expression of the protein product.
Agreements
Manufacturing Agreements
Bio Vectra
dcl
In July 2004, we entered into
negotiations with Diagnostics Chemicals Limited (doing business as BioVectra dcl) in Prince Edward Island for the commercial manufacture of
Virulizin®, for which a contract was executed in October 2004. BioVectra has a cGMP facility capable of large-scale commercial production. In June
2005 Lorus announced that BioVectra had successfully produced Virulizin® in both optimized clinical and commercial batch scales. The contract
remains in force, although Bio Vectra is not currently performing any manufacturing of Virulizin®.
Licence Agreements
Ion
Pharmaceuticals and Cyclacel
In December 1997, Lorus, through
NuChem, acquired certain patent rights and a sublicense from Ion to develop and commercialize the anticancer applications of CLT and new chemical
entities related to CLT (the “NuChem Analogs”). To July 2006, NuChem had made cash payments totalling US $500,000 to Ion. The balance is
payable upon the achievement of certain milestones based on the commencement and completion of clinical trials related to the NuChem
Analogs.
23
All research and development activities
to be undertaken by NuChem are to be funded by us through subscriptions for non-participating preference shares of NuChem. As at May 31, 2006, we had
provided a total of $6,079,000 of funding to NuChem.
In September 2003, Lorus, NuChem and
Cyclacel Limited signed an exclusive worldwide license agreement for the development and commercialization of the NuChem Analogs. Under the terms of
the agreement, Lorus received upfront fees of US $400,000 and will receive milestone payments which, assuming all milestones are achieved, will total
approximately US $11.6 million for our pre-clinical compound NC 381, and similar milestone payments for each of any other compounds developed from the
compound library. In addition to these payments, we will receive royalties based on product sales. Cyclacel is responsible for all future drug
development costs.
University of
Manitoba
The University of Manitoba (the
“University”), Dr. Jim Wright, Dr. Aiping Young and Cancer Care entered into an exclusive license agreement (the “License
Agreement”) with GeneSense dated June 20, 1997 pursuant to which GeneSense was granted an exclusive worldwide license to certain patent rights
with the right to sub-license. In consideration for the exclusive license to GeneSense of the patent rights, the University and Cancer Care are
entitled to an aggregate of 1.67% of the net sales received by GeneSense from the sale of products or processes derived from the patent rights and
1.67% of all monies received by GeneSense from sub-licenses of the patent rights. GeneSense is solely responsible for the preparation, filing,
prosecution and maintenance of all patent applications and patents included in the patent rights and all related expenses. Pursuant to the terms of the
License Agreement, any and all improvements to any of the patent rights derived in whole or in part by GeneSense after the date of the License
Agreement are not included within the scope of the License Agreement and do not trigger any payment of royalties.
Collaboration Agreements
National
Cancer Institute
In February 2003, Lorus and the United
States National Cancer Institute approved clinical protocols to conduct a series of clinical trials in a Phase II program to investigate the safety and
efficacy of our lead antisense drug, GTI-2040 in breast cancer, colon cancer, non-small cell lung cancer, acute myeloid leukemia, prostate cancer, and
in a range of solid tumours. Lorus and the NCI signed a formal clinical trial agreement (expiring in October 2007) in which the NCI financially
sponsors the GTI-2040 clinical trials, while Lorus provides the clinical trial drug. All six trials were in progress as of May 31, 2006. In July 2006,
we announced a seventh trial to be conducted with the NCI for GTI-2040 for the treatment of MDS and AML.
University of
Toronto
In May 2004 we signed a collaboration
agreement with the University of Toronto to provide a further development and delivery strategy for our novel low molecular weight compounds with
anticancer and antibacterial activity. The collaboration agreement provided for payment by us to the University of Toronto of set fees and a percentage
of net revenues derived from any intellectual property developed under the agreement if and when the intellectual property is commercialized. The work
under this agreement has been completed.
Sumitomo and
Koken
In April 2005, we signed a
collaboration agreement with Sumitomo and Koken with respect to GTI-2601, our antisense compound targeting thioredoxin. Sumitomo and Koken have
developed an advanced delivery system based on collagen complexed with macromolecules. The collaboration agreement provides that Sumitomo and Koken
will further develop their delivery technology to complex with GTI-2601, so that increased efficacy is provided with decreased doses of the antisense
drug. This agreement provides that Lorus, Sumitomo and Koken will jointly own the compounds that result from this collaboration (Lorus: Sumitomo and
Koken, 1:1).
24
Other
From time to time, we enter into other
research and technology agreements with third parties under which research is conducted and monies expended. These agreements outline the
responsibilities of each participant and the appropriate arrangements in the event the research produces a product candidate.
We also have licensing agreements to
use proprietary technology of third parties in relation to our research and development. If this research ultimately results in a commercialized
product, we have agreed to pay certain royalties and licensing fees.
Business Strategy
By developing cancer therapeutics using
different mechanisms of action that may be efficacious against a wide variety of cancers, we seek to maximize our opportunity to address multiple
cancer therapeutic markets. In our efforts to obtain the greatest return on our investment in each drug candidate, we separately evaluate the merits of
each candidate throughout the clinical trial process and consider commercialization opportunities when appropriate. In the next fiscal year, we intend
to pursue partnerships and further development of our lead technologies.
Our objective is to maximize the
therapeutic value and potential commercial success of GTI-2040 and GTI-2501, and the small molecule platform. In the near term, we intend to pursue
research and early clinical development with our own funds with respect to GTI-2040, GTI-2501 and the small molecule platform. In our efforts to obtain
the greatest return on our investment in each drug candidate, we separately evaluate the merits of each candidate throughout the clinical trial process
and consider commercialization opportunities when appropriate.
To meet future financing requirements,
we intend to finance our operations through some or all of the following methods: public or private equity or debt financings, capital leases, and
collaborative and licensing agreements. We intend to pursue financing opportunities as they arise.
Public
Offering
On June 11, 2003, Lorus raised net
proceeds of $29.9 million by way of a public offering of 26,220,000 units at a price of $1.25 per unit, each unit consisting of one common share and
one-half of one purchase warrant.
Secured Convertible
Debentures
On October 6, 2004, we entered into a
Subscription Agreement (the “Agreement”) with The Erin Mills Investment Corporation (“TEMIC”) to issue an aggregate of $15 million
of secured convertible debentures (the “Debentures”) issuable in three tranches of $5 million each, in each of October 2004, January 2005 and
April 2005. The Debentures are secured by a first charge over all of the assets of the Company. All Debentures issued under the Agreement are due on
October 6, 2009 and are subject to interest payable monthly at a rate of prime plus 1% until such time as the Company’s share price reaches $1.75
for 60 consecutive trading days, at which time interest will no longer be charged. Interest is payable in common shares of Lorus until Lorus’
shares trade at a price of $1.00 or more after which interest will be payable in cash or common shares at the option of the debenture holder. Common
shares issued in payment of interest are issued at a price equal to the weighted average trading price of such shares for the ten trading days
immediately preceding their issue in respect of each interest payment. The $15.0 million principal amount of Debentures is convertible at the
holder’s option at any time into our common shares with a conversion price per share of $1.00. With the issuance of each $5.0 million debenture,
we issued to the debt holder 1,000,000 warrants with a term of five years to purchase our common shares at a price per share equal to
$1.00.
Share
Issuances
On July 13, 2006 we entered into an
agreement with High Tech Beteiligungen GmbH & Co. KG (“High Tech”) to issue 28.8 million common shares at $0.36 per share for gross
proceeds of $10.4 million. The subscription price represented a premium of 7.5% over the closing price of the common shares on the TSX on July 13,
2006. The transaction closed on August 30, 2006.
25
In connection with the closing, we also
entered into a registration rights agreement with High Tech that provides, among other things, that High Tech will have a demand right to request, an
aggregate number of five times, the registration or qualification of the Purchased Shares for resale in the United States and Canada, subject to
certain restrictions. High Tech has also been granted piggy-back rights to enable it to sell the Purchased Shares in connection with a public offering
of shares to Lorus, subject to certain exceptions. In addition, pursuant to the Share Purchase Agreement, High Tech has the right to nominate one
nominee for election to the board of directors of Lorus or, if it does not have a nominee, it will have the right to appoint an observer to the board
for as long as it owns shares.
In accordance with the terms of the
Share Purchase Agreement, Lorus agreed not to issue any common shares or securities convertible into common shares, subject to certain limited
exceptions, until July 31, 2007, at a price of less than $0.36 per common share. In addition, certain named executive officers of Lorus signed
“lock-up” agreements whereby they agreed not to dispose of their common shares for a period of 30 days following the closing date, and for
the 30 days immediately following such 30 day period, they agreed not to dispose greater than 50% of the aggregate number of common shares they hold as
at the closing date.
On July 24, 2006 we entered into an
agreement with Technifund Inc. to issue on a private placement basis, 5 million common shares at $0.36 per share for gross proceeds of $1.8 million.
The transaction closed on August 31 2006.
Revenues
The Company has not earned substantial
revenues from its drug candidates and is therefore considered to be in the development stage.
Employees
As at May 31, 2006, we employed 33
full-time persons and one part-time person in research and drug development and administration activities. Of our employees, nine hold Ph.D.s. To
encourage a focus on achieving long-term performance, employees and members of the board of directors have the ability to acquire an ownership interest
in the Company through Lorus’ stock option plan and employees can participate in the employee share purchase plan, which was established in
2005.
Our ability to develop commercial
products and to establish and maintain our competitive position in light of technological developments will depend, in part, on our ability to attract
and retain qualified personnel. There is a significant level of competition in the marketplace for such personnel. We believe that to date we have been
successful in attracting and retaining the highly skilled personnel critical to our business. We have also chosen to outsource activities where skills
are in short supply or where it is economically prudent to do so.
None of our employees are unionized,
and we consider our relations with our employees to be good.
Office Facilities
Our head office, which occupies 20,500
square feet, is located at 2 Meridian Road, Toronto, Ontario. The leased premises include approximately 8,000 square feet of laboratory and research
space. We believe that our existing facilities are adequate to meet our requirements for the near term. Our current lease expires on March 31,
2008.
Competition
The biotechnology and pharmaceutical
industries are characterized by rapidly evolving technology and intense competition. There are many companies in both these industries that are
focusing their efforts on activities similar to ours. Some of these are companies with established positions in the pharmaceutical industry and may
have substantially more financial and technical resources, more extensive research and development capabilities, and greater marketing, distribution,
production and human resources than us. In addition, we may face competition from other companies for opportunities to enter into collaborative
agreements with biotechnology and
26
pharmaceutical companies and academic institutions. Many of these other companies are not solely focused on cancer, as is the mission of our drug development. We specialize in the development of drugs that we believe will manage cancer.
Products that may compete with our
products include chemotherapeutic agents, monoclonal antibodies, antisense therapies and immunotherapies with novel mechanisms of action. These are
drugs that are delivered by specific means and are targeting cancers with large disease populations. We also expect that we may experience competition
from established and emerging pharmaceutical and biotechnology companies that have other forms of treatment for the cancers that we target. There are
many drugs currently in development for the treatment of cancer that employ a number of novel approaches for attacking these cancers. Cancer is a
complex disease with more than 100 indications requiring drugs for treatment. The drugs in competition with our product candidates have specific
targets for attacking the disease, targets which are not necessarily the same as ours. These competitive drugs therefore could potentially also be used
together in combination therapies with our drugs to manage the disease.
Government Regulation
Overview
Regulation by government authorities in
Canada, the United States, Mexico and the European Union is a significant factor in our current research and drug development activities. To clinically
test, manufacture and market drug products for therapeutic use, we must satisfy the rigorous mandatory procedures and standards established by the
regulatory agencies in the countries in which we currently operate or intend to operate.
The laws of most of these countries
require the licensing of manufacturing facilities, carefully controlled research and the extensive testing of products. Biotechnology companies must
establish the safety and efficacy of their new products in clinical trials and establish cGMP and control over marketing activities before being
allowed to market their products. The safety and efficacy of a new drug must be shown through clinical trials of the drug carried out in accordance
with the mandatory procedures and standards established by regulatory agencies.
The process of completing clinical
trials and obtaining regulatory approval for a new drug takes a number of years and requires the expenditure of substantial resources. Once a new drug
or product license application is submitted, we cannot assure you that a regulatory agency will review and approve the application in a timely manner.
Even after initial approval has been obtained, further studies, including post-marketing studies, may be required to provide additional data on
efficacy and safety necessary to confirm the approved indication or to gain approval for the use of the new drug as a treatment for clinical
indications other than those for which the new drug was initially tested. Also, regulatory agencies require post-marketing surveillance programs to
monitor a new drug’s side effects. Results of post-marketing programs may limit or expand the further marketing of new drugs. A serious safety or
effectiveness problem involving an approved new drug may result in a regulatory agency requiring withdrawal of the new drug from the market and
possible civil action. We cannot assure you that we will not encounter such difficulties or excessive costs in our efforts to secure necessary
approvals, which could delay or prevent us from manufacturing or marketing our products.
In addition to the regulatory product
approval framework, biotechnology companies, including Lorus, are subject to regulation under local provincial, state and federal law, including
requirements regarding occupational safety, laboratory practices, environmental protection and hazardous substance control, and may be subject to other
present and future local, provincial, state, federal and foreign regulation, including possible future regulation of the biotechnology
industry.
Canada
In Canada, the manufacture and sale of
drugs are controlled by Health Canada (“HC”). New drugs (sometimes referred to as drug candidates or product candidates) must pass through a
number of testing stages, including pre-clinical testing and clinical trials. Pre-clinical testing involves testing the new drug’s chemistry,
pharmacology and toxicology in vitro and in vivo. Successful results (that is, potentially valuable pharmacological activity combined
with an acceptable low level of toxicity) enable the developer of the drug candidate to file a clinical trial application (“CTA”) to begin
clinical trials involving humans.
27
To study a drug in Canadian patients, a
CTA submission must be filed with HC. The CTA submission must contain specified information, including the results of the pre-clinical tests completed
at the time of the submission and any available information regarding use of the drug in humans. In addition, since the method of manufacture may
affect the efficacy and safety of a new drug, information on manufacturing methods and standards and the stability of the drug substance and dosage
form must be presented. Production methods and quality control procedures must be in place to ensure an acceptably pure product, essentially free of
contamination, and to ensure uniformity with respect to all quality aspects.
Provided HC does not reject a CTA
submission, clinical trials can begin. Clinical trials for product candidates to treat cancer are generally carried out in three phases. Phase I
involves studies to evaluate toxicity and ideal dose levels in humans. The new drug is administered to human patients who have met the clinical trial
entry criteria to determine pharmacokinetics, human tolerance and prevalence of adverse side effects. Phases II and III involve therapeutic studies. In
Phase II, efficacy, dosage, side effects and safety are established in a small number of patients who have the disease or disorder that the new drug is
intended to treat. In Phase III, there are controlled clinical trials in which the drug candidate is administered to a large number of patients who are
likely to receive benefit from the new drug. In Phase III, the effectiveness of the drug candidate is compared to that of standard accepted methods of
treatment in order to provide sufficient data for the statistical proof of safety and efficacy for the new drug.
If clinical studies establish that a
drug candidate has value, the manufacturer submits a new drug submission (“NDS”) application to HC for marketing approval. The NDS contains
all information known about the drug candidate, including the results of pre-clinical testing and clinical trials. Information about a substance
contained in an NDS includes its proper name, its chemical name, and details on its method of manufacturing and purification, and its biological,
pharmacological and toxicological properties. The NDS also provides information about the dosage form of the new drug, including a quantitative listing
of all ingredients used in its formulation, its method of manufacture, manufacturing facility information, packaging and labelling, the results of
stability tests, and its diagnostic or therapeutic claims and side effects, as well as details of the clinical trials to support the safety and
efficacy of the new drug. Furthermore, for biological products, an on-site evaluation is required prior to the issuance of a notice of compliance
(“NOC”). All aspects of the NDS are critically reviewed by HC. If an NDS is found satisfactory, an NOC is issued permitting the new drug to
be sold. In Canada an establishment license must be obtained prior to marketing the product.
HC has a policy of priority evaluation
of new drug submissions for all drugs intended for serious or life-threatening diseases for which no drug product has received regulatory approval in
Canada and for which there is reasonable scientific evidence to indicate that the proposed drug candidate is safe and may provide effective
treatment.
The monitoring of a new drug does not
cease once it is on the market. For example, a manufacturer of a new drug must report any new information received concerning serious side effects, as
well as the failure of the new drug to produce desired effects. As well, if HC determines it to be in the interest of public health, a notice of
compliance for a new drug may be suspended and the new drug may be removed from the market.
An exception to the foregoing
requirements relating to the manufacture and sale of a new drug is the limited authorization that may be available in respect of the sale of drug
candidates for emergency treatment. Under a special access program, HC may authorize the sale of a quantity of a new drug for human use to a specific
practitioner for the emergency treatment of a patient under the practitioner’s care. Prior to authorization, the practitioner must supply HC with
information concerning the medical emergency for which the new drug is required, such data as is in the possession of the practitioner with respect to
the use, safety and efficacy of the new drug, the names of the institutions at which the new drug is to be used and such other information as may be
requested by HC. In addition, the practitioner must agree to report to both the drug manufacturer and HC the results of the new drug’s use in the
medical emergency, including information concerning adverse reactions, and must account to HC for all quantities of the new drug made
available.
The Canadian regulatory approval
requirements for new drugs outlined above are similar to those of other major pharmaceutical markets. While the testing carried out in Canada is often
acceptable for the purposes of regulatory submissions in other countries, individual regulatory authorities may request supplementary
testing
28
during their assessment of any submission. We cannot assure you that the clinical testing conducted under HC authorization or the approval of regulatory authorities of other countries will be accepted by regulatory authorities of such other countries outside of Canada.
United States
In the United States, the United States
Food and Drug Administration (“FDA”) controls the manufacture and sale of new drugs. New drugs require FDA approval of a marketing
application (e.g., an NDA or FDA application) prior to commercial sale. To obtain marketing approval, data from adequate and well-controlled
clinical investigations, demonstrating to the FDA’s satisfaction a new drug’s safety and effectiveness for its intended use, are required.
Such data are generated in studies conducted pursuant to an IND submission, similar to that required for a CTA in Canada. As in Canada, clinical
studies are characterized as Phase I, Phase II and Phase III trials or a combination thereof. In a marketing application, the manufacturer must also
demonstrate the identity, potency, quality and purity of the active ingredients of the new drug involved, and the stability of those ingredients.
Further, the manufacturing facilities, equipment, processes and quality controls for the new drug must comply with the FDA’s cGMP regulations for
drugs or biological products both in a pre-licensing inspection before product licensing and in subsequent periodic inspections after licensing. In the
case of a biological product, an establishment license must be obtained prior to marketing and batch releasing.
A five-year period of market
exclusivity for a drug comprising a new chemical entity (“NCE”) is available to an applicant that succeeds in obtaining FDA approval of an
NCE, provided the active ingredient of the NCE has never before been approved in an NDA. During this exclusivity period, the FDA may not approve any
abbreviated application filed by another sponsor for a generic version of the NCE. Further, a three-year period of market exclusivity for a new use or
indication for a previously approved drug is available to an applicant that submits new clinical studies that are essential to support the new use or
indication. During the latter period of exclusivity, the FDA may not approve an abbreviated application filed by another sponsor for a generic version
of the product for that use or indication.
The FDA has “fast track”
regulations intended to accelerate the approval process for the development, evaluation and marketing of new drugs used to diagnose or treat
life-threatening and severely debilitating illnesses for which no satisfactory alternative therapies exist. “Fast track” designation affords
early interaction with the FDA in terms of protocol design and permits, although it does not require, the FDA to issue marketing approval after
completion of Phase II clinical trials (although the FDA will require subsequent clinical trials or even post-approval efficacy
studies).
C. |
Organizational Structure |
Lorus Therapeutics Inc. was
incorporated under the Business Corporations Act (Ontario) on September 5, 1986 under the name RML Medical Laboratories Inc. On October 28,
1991, RML Medical Laboratories Inc. amalgamated with Mint Gold Resources Ltd., resulting in the Company becoming a reporting issuer (as defined under
applicable securities law) in Ontario, on such date. On August 25, 1992, the Company changed its name to IMUTEC Corporation. On November 27, 1996, the
Company changed its name to Imutec Pharma Inc., and on November 19, 1998, the Company changed its name to Lorus Therapeutics Inc. On October 1, 2005
the Company continued under the Canada Business Corporations Act.
The address of the Company’s head
and registered office is 2 Meridian Road, Toronto, Ontario, Canada, M9W 4Z7. Our corporate website is www.lorusthera.com. The contents of the website
are specifically not included in this 20-F by reference.
D. |
Property, Plant and Equipment |
Our head office, which occupies 20,500
square feet, is located at 2 Meridian Road, Toronto, Ontario. The leased premises include approximately 8,000 square feet of laboratory and research
space. We believe that our existing facilities are adequate to meet our requirements for the near term. Our current lease expires on March 31,
2008.
29
Item
4A. |
Unresolved Staff Comments |
Not applicable.
Item
5. |
Operating and Financial Review and Prospects |
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL
CONDITION AND RESULTS OF OPERATIONS
A. |
Operating Results |
The following discussion should be read
in conjunction with the audited consolidated financial statements for the year ended May 31, 2006 and the accompanying notes (the “Financial
Statements”) set forth elsewhere in this report. The Financial Statements, and all financial information discussed below, have been prepared in
accordance with Canadian generally accepted accounting principles (“GAAP”). Significant differences between Canadian and United States GAAP
are identified in Note 17 to the Financial Statements. All amounts are expressed in Canadian dollars unless otherwise noted. In this Management’s
Discussion and Analysis, “Lorus”, the “Company’, “we”, “us” and “our” each refers to Lorus
Therapeutics Inc.
Overview
Lorus Therapeutics Inc. is a life
sciences company focused on the discovery, research and development of effective anticancer therapies with a high safety profile. Lorus has worked
diligently to establish a diverse, marketable anticancer product pipeline, with products in various stages of development ranging from preclinical to
multiple Phase II clinical trials. A growing intellectual property portfolio supports our diverse product pipeline.
Our success is dependent upon several
factors, including establishing the efficacy and safety of our products in clinical trials, securing strategic partnerships, obtaining the necessary
regulatory approvals to market our products and maintaining sufficient levels of funding through public and/or private financing.
We believe that the future of cancer
treatment and management lies in drugs that are effective, safe and have minimal side effects, and therefore improve a patient’s quality of life.
Many of the cancer drugs currently approved for the treatment and management of cancer are toxic with severe side effects, and we therefore believe
that a product development plan based on effective and safe drugs could have broad applications in cancer treatment. Lorus’ strategy is to
continue the development of our product pipeline using several therapeutic approaches. Each therapeutic approach is dependent on different
technologies, thereby mitigating the development risks associated with a single technology platform. We evaluate the merits of each product throughout
the clinical trial process and consider commercialization as appropriate. The most advanced anticancer drugs in our pipeline, each of which flow from
different platform technologies, are: immunotherapeutics; antisense and small molecules.
Our net loss for 2006 totalled $17.9
million ($0.10 per share) compared to a net loss of $22.1 million ($0.13 per share) in 2005. Research and development expenses in 2006 decreased to
$10.2 million from $14.4 million in 2005. The close of the Virulizin® Phase III clinical trial in 2006 as well as staff reductions resulting from
the November 2005 corporate changes (described below) contributed to the decrease over 2005. We utilized cash of $13.1 million in our operating
activities in 2006 compared with $18.7 million in 2005; the lower utilization is consistent with lower research and development activities and lower
general and administrative expenses offset by lower interest income. At the end of 2006 we had cash and cash equivalents and short-term investments of
$8.3 million compared to $21.5 million at the end of 2005.
30
Selected Annual Financial
Data
The following selected consolidated
financial data have been derived from, and should be read in conjunction with, the accompanying audited consolidated financial statements for the year
ended May 31, 2006 which are prepared in accordance with Canadian GAAP.
Consolidated Statements of
Loss and Deficit
(amounts in Canadian
000’s except for per common share data)
| Years Ended May 31 |
|||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2006 |
|
2005 |
|
2004 |
||||||||||
REVENUE |
$ | 26 | $ | 6 | $ | 608 | |||||||||
EXPENSES |
|||||||||||||||
Cost of
sales |
3 | 1 | 28 | ||||||||||||
Research and
development |
10,237 | 14,394 | 26,785 | ||||||||||||
General and
administrative |
4,334 | 5,348 | 4,915 | ||||||||||||
Stock-based
compensation |
1,205 | 1,475 | — | ||||||||||||
Depreciation and amortization |
771 | 564 | 420 | ||||||||||||
Operating
expenses |
16,550 | 21,782 | 32,148 | ||||||||||||
Interest
expense |
882 | 300 | — | ||||||||||||
Accretion in
carrying value of secured convertible debentures |
790 | 426 | — | ||||||||||||
Amortization of
deferred financing charges |
87 | 84 | — | ||||||||||||
Interest income |
(374 | ) | (524 | ) | (1,239 | ) | |||||||||
Loss for the period |
17,909 | 22,062 | 30,301 | ||||||||||||
Basic and diluted loss per common share |
$ | 0.10 | $ | 0.13 | $ | 0.18 | |||||||||
Weighted average number of common shares outstanding used in the calculation of basic and diluted loss per
share |
173,523 | 172,112 | 171,628 | ||||||||||||
Total Assets |
$ | 11,461 | $ | 27,566 | $ | 34,424 | |||||||||
Total Long-term liabilities |
$ | 11,002 | $ | 10,212 | $ | — | |||||||||
Accounting Policy
Changes
Variable
Interest Entities
Effective June 1, 2005, the Company
adopted the recommendations of CICA Handbook Accounting Guideline 15 (AcG-15), Consolidation of Variable Interest Entities, effective for fiscal years
beginning on or after November 1, 2004. Variable interest entities (VIEs) refer to those entities that are subject to control on a basis other than
ownership of voting interests. AcG-15 provides guidance for identifying VIEs and criteria for determining which entity, if any, should consolidate
them. The adoption of AcG-15 did not have an effect on the financial position, results of operations or cash flows in the current period or the prior
period presented.
31
Financial Instruments —
Disclosure and Presentation
Effective June 1, 2005, the Company
adopted the amended recommendations of CICA Handbook Section 3860, Financial Instruments — Disclosure and Presentation, effective for fiscal years
beginning on or after November 1, 2004. Section 3860 requires that certain obligations that may be settled at the issuer’s option in cash or the
equivalent value by a variable number of the issuer’s own equity instruments be presented as a liability. The Company has determined that there is
no impact on the Financial Statements resulting from the adoption of the amendments to Section 3860 either in the current period or the prior period
presented.
Accounting for Convertible
Debt Instruments
On October 17, 2005, the CICA issued
EIC 158, Accounting for Convertible Debt Instruments applicable to convertible debt instruments issued subsequent to the date of the EIC. EIC 158
discusses the accounting treatment of convertible debentures in which upon conversion, the issuer is either required or has the option to satisfy all
or part of the obligation in cash. The EIC discusses various accounting issues related to this type of convertible debt. The Company has determined
that there is no impact on the Financial Statements resulting from the adoption of EIC 158 either in the current period or the prior period
presented.
Section 3831, Non-Monetary
Transactions
In June 2005, the CICA released a new
Handbook Section 3831, Non-monetary Transactions, effective for all non-monetary transactions initiated in periods beginning on or after January 1,
2006. This standard requires all non-monetary transactions to be measured at fair value unless they meet one of four very specific criteria. Commercial
substance replaces culmination of the earnings process as the test for fair value measurement. A transaction has commercial substance if it causes an
identifiable and measurable change in the economic circumstances of the entity. Commercial substance is a function of the cash flows expected by the
reporting entity.
Critical Accounting
Policies
The Company periodically reviews its
financial reporting and disclosure practices and accounting policies to ensure that they provide accurate and transparent information relative to the
current economic and business environment. As part of this process, the Company has reviewed its selection, application and communication of critical
accounting policies and financial disclosures. Management has discussed the development and selection of the critical accounting policies with the
Audit Committee of the Board of Directors and the Audit Committee has reviewed the disclosure relating to critical accounting policies in this
Management’s Discussion and Analysis. Other important accounting polices are described in note 2 of the Financial Statements.
Drug Development
Costs
We incur costs related to the research
and development of pharmaceutical products and technologies for the management of cancer. These costs include internal and external costs for
preclinical research and clinical trials, drug costs, regulatory compliance costs and patent application costs. All research costs are expensed as
incurred as required under GAAP.
Development costs, including the cost
of drugs for use in clinical trials, are expensed as incurred unless they meet the criteria under GAAP for deferral and amortization. The Company
continually assesses its activities to determine when, if ever, development costs may qualify for capitalization. By expensing the research and
development costs as required under GAAP, the value of the product portfolio is not reflected on the Company’s Financial
Statements.
Stock-Based
Compensation
We have applied the fair value based
method to expense stock options awarded since June 1, 2002 using the Black-Scholes option-pricing model as allowed under CICA Handbook Section 3870.
The model estimates the fair value of fully transferable options, without vesting restrictions, which significantly differs from the stock option
awards issued by Lorus. The model also requires four highly subjective assumptions including future stock price
32
volatility and expected time until exercise, which greatly affect the calculated values. The increase or decrease of one of these assumptions could materially increase or decrease the fair value of stock options issued and the associated expense.
Valuation Allowance for Future
Tax Assets
We have a net tax benefit resulting
from non-capital losses carried forward, and scientific research and experimental development expenditures. In light of the recent net losses and
uncertainty regarding our future ability to generate taxable income, management is of the opinion that it is not more likely than not that these tax
assets will be realized in the foreseeable future and hence, a full valuation allowance has been recorded against these income tax assets.
Consequently, no future income tax assets or liabilities are recorded on the balance sheets. The generation of future taxable income could result in
the recognition of some portion or all of these benefits, which could result in a material improvement in our results of operations through the
recovery of future income taxes.
Valuation of Long Lived
Assets
We
periodically review the useful lives and the carrying values of our long lived
assets. We review for impairment in long lived assets whenever events or changes
in circumstances indicate that the carrying amount of the assets may not be
recoverable. If the sum of the undiscounted future cash flows expected to result
from the use and eventual disposition of an asset is less than its carrying
amount, it is considered to be impaired. An impairment loss is measured at the
amount by which the carrying amount of the asset exceeds its fair value; which
is estimated as the expected future cash flows discounted at a rate commensurate
with the risks associated with the recovery of the asset.
Recent Accounting
Pronouncements Yet To Be Adopted — Canadian GAAP
Comprehensive
Income and Equity
In January 2005, the CICA released new
Handbook Section 1530, Comprehensive Income, and Section 3251, Equity. Section 1530 establishes standards for reporting comprehensive income. The
section does not address issues of recognition or measurement for comprehensive income and its components. Section 3251 establishes standards for the
presentation of equity and changes in equity during the reporting period. The requirements in this section are in addition to Section
1530.
Section 3855, Financial
Instruments — Recognition and Measurement
CICA Handbook Section 3855 establishes
standards for the recognition and measurement of all financial instruments, provides a characteristics-based definition of a derivative instrument,
provides criteria to be used to determine when a financial instrument should be recognized, and provides criteria to be used to determine when a
financial liability is considered to be extinguished.
Section 3865,
Hedges
Section 3865 establishes standards for
when and how hedge accounting may be applied. Hedge accounting is optional.
These three Sections are effective for
fiscal years beginning on or after October 1, 2006. An entity adopting these Sections for a fiscal year beginning before October 1, 2006 must adopt all
the Sections simultaneously.
Recent Accounting
Pronouncements Yet To Be Adopted — US GAAP
In December 2004, the FASB revised SFAS
No. 123 to require companies to recognize n the income statement the grant-date fair value of stock options and other equity based compensation issued
to employees, but expressed no preference for a type of valuation model (SFAS 123R). The way an award is classified will affect the measurement of
compensation cost. Liability-classified awards are re-measured to fair value at each balance sheet
33
date until the award is settled. Equity-classified awards are measured at grant-date fair value and the grant-date fair value is recognized over the requisite service period. Such awards are not subsequently re-measured.
In April 2005, the staff of the Securities and Exchange
Commission issued Staff Accounting Bulletin No. 107 (SAB 107) to provide additional guidance regarding the application of SFAS 123R. SAB 107 permits
registrants to choose an appropriate valuation technique or model to estimate the fair value of share options, assuming consistent application, and
provides guidance for the development of assumptions used in the valuation process. Based upon SEC rules issued in April 2005, SFAS 123R is effective
for fiscal years that begin after June 15, 2005 and will be adopted by the Company effective June 1, 2006. Additionally, SAB 107 discusses disclosures
to be made under “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in registrants’ periodic
reports. The Company has not yet determined the effect of this new standard on its consolidated financial position and results of
operations.
In December 2004, FASB issued Financial
Accounting Standard 153: Exchanges of Nonmonetary Assets as an amendment of APB Opinion No. 29. The guidance in APB Opinion No. 29, Accounting for
Nonmonetary Transactions, is based on the principle that exchanges of nonmonetary assets should be measured based on the fair value of the assets
exchanged. The guidance in that Opinion, however, included certain exceptions to that principle. This Statement amends Opinion 29 to eliminate the
exception for nonmonetary exchanges of similar productive assets and replaces it with a general exception for exchanges of nonmonetary assets that do
not have commercial substance. Nonmonetary exchange has commercial substance if the future cash flows of the entity are expected to change
significantly as a result of the exchange. This statement is effective for years beginning after June 15, 2005. The Company has not entered into any
non-monetary transactions and as such this section is not applicable.
In May 2005, the FASB issued SFAS No.
154, Accounting Changes and Error Corrections (SFAS 154), which replaces APB No. 20, Accounting Change and SFAS No. 3, Reporting Accounting Changes in
Interim Financial Statements—An Amendment of APB Opinion No. 28. SFAS 154 provides guidance on the accounting for and reporting of accounting
changes and error corrections. It establishes retrospective application, on the latest practicable date, as the required method for reporting a change
in accounting principle and the reporting of a correction of an error. SFAS 154 is effective for accounting changes and corrections of errors made in
fiscal years beginning after December 15, 2005. Management believes that the adoption of this statement will not have a material effect on the
Company’s consolidated financial condition or results of operations.
Operating
Results
Revenues
Revenues for the year increased to $26
thousand compared with 2005 revenue of $6 thousand which decreased compared with $608 thousand in 2004. The increase in revenue in 2006 is due to lab
work performed by Lorus personnel on behalf of other companies. The decrease in 2005 compared with 2004 is the result of a licensing agreement Lorus
entered into during 2004 with Cyclacel Ltd. in connection with the out licensing of our clotrimazole analog library of anticancer drug candidates. The
agreement included an initial license fee of $546 thousand received in 2004 with the potential of additional license fees of up to U.S.$11.6 million
that may be earned if Cyclacel achieves certain defined research and development milestones. We do not expect that any of these milestones will be
achieved in the next 12 months. The balance of the revenue earned during 2004 relates to product and royalty revenues from the sale of Virulizin®
to our distributor in the Mexican market, Mayne Pharma. As of July 31, 2005, our contract with Mayne Pharma to distribute Virulizin® in Mexico was
terminated as a result of Mayne Pharma ceasing operations in Mexico and Brazil. We do not anticipate product revenue in fiscal 2007 from any of our
other anticancer drugs currently under development.
Research and
Development
Research and development expenses
totalled $10.2 million in 2006 compared to $14.4 million in 2005 and $26.8 million in 2004. The decrease in spending compared with 2005 is due to the
close of our Virulizin® Phase III clinical trial for the treatment of advanced pancreatic cancer in 2006 as well as a reduction in headcount in
November 2005 as described under corporate changes. Although many expenditures related to the trial continued, as the results of the trial were
compiled and analyzed and the trial was wound up, the costs were less in comparison
34
with the prior year when the trial was fully enrolled and underway. The significant decrease in expenditures in 2005 in comparison with 2004 is primarily the result of two factors. First, in 2004 the Phase III clinical trial of Virulizin® was progressing through a heavy enrolment period resulting in many up front costs, including personnel, drug manufacturing and testing, combination drug purchases and contract research organization costs. In 2005, the study and the associated costs wound down to the point of last patient visit in Q1 2006. Second, we incurred expenditures in 2004 related to the upfront manufacturing of GTI-2040 for the U.S. National Cancer Institute (NCI) sponsored Phase II clinical trials as well as GTI-2501 for our Phase I/II prostate trial. We have had, and continue to have, a sufficient drug supply on hand such that no additional costs were incurred during 2005 and 2006.
Of the total research and development
expenditures incurred during the year, Virulizin® accounted for $6.2 million or 61% of the total spending. During the past year as we wound down
the Phase III clinical trial we focused the majority of the Company’s time and resources on Virulizin®.
General and
Administrative
General and administrative expenses
totalled $4.3 million in 2006 compared to $5.3 million in 2005 and $4.9 million in 2004. The decrease of $1.0 million during 2006 is due to reductions
in headcount in November 2005 as described under corporate changes as well as lower legal, consulting and investor relations costs, the result of
changes made to reduce our cash burn rate. The increase in expenditures in 2005 of $400 thousand compared with 2004 was primarily due to additional
administrative personnel as we were preparing for commercialization in the event of successful Phase III clinical results.
Stock-Based
Compensation
Stock- based compensation expense
totalled $1.2 million in 2006 compared with $1.5 million in 2005 and nil in 2004. The decrease in stock-based compensation expense in 2006, despite an
increase in the number of options issued is the result of reduced fair values on the stock options issued, due to a decline in our stock price, as well
as a significant number of unvested options that were forfeited during the year, reducing the overall expense. During 2006, employees of the Company
(excluding directors and officers) were given the opportunity to choose between keeping 100% of the options they held at the existing exercise prices
or forfeiting 50% of the options held in exchange for having the remaining 50% of the exercise prices of the options re-priced to $0.30 per share.
Employees holding 2,290,000 stock options opted for re-pricing their options, resulting in the amendment of the exercise price of 1,145,000 stock
options and the forfeiture of 1,145,000 stock options during the quarter ended February 28, 2006. The 2005 expense represents the amortization of the
estimated fair value of stock options granted since June 1, 2002 applicable to the current service period as well as a charge of $208 thousand recorded
in the second quarter of 2005 representing the increase in value attributed to the shareholder approved amendment to the stock option plan to extend
the contractual life of all options outstanding from five-years to ten-years.
Depreciation and
Amortization
Depreciation and amortization expenses
increased to $771 thousand in 2006 compared to $564 thousand in 2005 and $420 thousand in 2004. The increase in expense in 2006 compared with 2005 is
due to a write-down of $250 thousand taken on certain furniture and equipment whose carrying value was deemed to be unrecoverable and in excess of the
fair value of the underlying assets offset by a lower level of capital expenditures in 2006. The increase in expense in 2005 compared with 2004 is due
to the acquisition of additional capital related to the scale up of our manufacturing process, as well as a write-down of $75 thousand taken on certain
equipment whose carrying value was deemed to be unrecoverable and in excess of the estimated future undiscounted cash flows of the underlying
assets.
Interest
Expense
Non-cash interest expense was $882
thousand in 2006 compared with $300 thousand in 2005 and nil in 2004. These amounts represent interest at a rate of prime +1% on the $15 million
convertible debentures. The increase in interest expense in 2006 compared with 2005 is a combination of higher interest rates due to increases in the
prime rate, as well as the full amount of the debentures outstanding for the entire year, rather than part of the year as in 2005. In 2005, the
interest accrued based on the cash advanced beginning October 6, 2004 when the first
35
tranche of $5 million was advanced through to May 31, 2005 when the entire $15 million had been advanced. All interest accrued on the debentures to date has been paid in common shares of the Company.
Accretion in Carrying Value of
Secured Convertible Debentures
Accretion in the carrying value of the
debentures amounted to $790 thousand in 2006 compared with $426 thousand in 2005 and nil in 2004. The accretion charges arise as under GAAP and the
Company has allocated the proceeds from each tranche of the debentures to the debt and equity instruments issued on a relative fair value basis
resulting in the $15.0 million debentures having an initial cumulative carrying value of $9.8 million as of their dates of issuance. Each reporting
period, the Company is required to accrete the carrying value of the convertible debentures such that at maturity on October 6, 2009, the carrying
value of the debentures will be the face value of $15.0 million. The increase in expense in 2006 compared with 2005 is due to a full year of accretion
in 2006 compared with a partial year in 2005.
Amortization of Deferred
Financing Charges
Amortization of deferred financing
charges totalled $87 thousand in 2006 compared with $84 thousand in 2005 and nil in 2004. The deferred financing charges relate to the convertible
debenture transaction and will be amortized using the effective interest rate method over the five-year life of the debt commencing October 6,
2004.
Interest and Other
Income
Interest income totalled $374 thousand
in 2006 compared to $524 thousand in 2005 and $1.2 million in 2004. The decrease from 2005 to 2006 is due to a lower average cash and short-term
investment balance in 2006 offset by higher interest rates during 2006. The decrease in 2005 compared with 2004 is the result of significantly lower
cash and short-term investment balances in 2005 compared with 2004.
Loss for the
Year
Net loss for the year decreased to
$17.9 million or $0.10 per share in 2006 compared to $22.1 million or $0.13 per share in 2005 and $30.3 million or $0.18 per share in 2004. The
decrease in net loss in 2006 compared with 2005 is due to lower research and development costs resulting from the close of our Virulizin® Phase
III clinical trial as well as staff reductions due to corporate changes, lower general and administrative costs due to staff reductions and lower
legal, consulting and investor relations charges off set by lower interest income due to reduced cash and short term investment balances as well as
higher non-cash interest, accretion and depreciation and amortization expense. The decrease in net loss in 2005 compared with 2004 is primarily due to
lower research and development costs resulting from the wind down of the Phase III Virulizin® clinical trial, as well as no GTI-2040 or GTI-2501
drug production in 2005, offset by lower interest revenue, and non-cash expenses associated with stock based compensation expense, and non-cash charges
related to the convertible debentures including accretion, interest and amortization of deferred financing charges.
Corporate
Changes
In November 2005, as a means to
conserve cash and refocus operations, Lorus scaled back some activities related to the Virulizin® technology and implemented a workforce reduction
of approximately 39% or 22 employees. As a result, we have recorded severance compensation expense for former employees of $557 thousand. Of this
expense, $468 thousand is presented in the income statement as general and administrative expense and $89 thousand as research and development expense.
Accounts payable and accrued liabilities at May 31, 2006 include severance and compensation expense liabilities relating to the Company’s November
2005 corporate changes of $154 thousand that will be paid out by December 2006.
36
Quarterly Results of
Operations
The following table sets forth certain
unaudited consolidated statements of operations data for each of the eight most recent fiscal quarters that, in management’s opinion, have been
prepared on a basis consistent with the audited consolidated financial statements contained elsewhere in this annual report and includes all
adjustments, consisting only of normal recurring adjustments, necessary for a fair presentation of the information presented.
Research and development expenses have
decreased throughout 2006 in comparison with the same quarter in the prior year. This reduction is due to the close of our Phase III Virulizin®
clinical trial as well as corporate changes in November 2005 to reduce headcount.
General and administrative expenses
increased for the quarter ended November 30, 2005 due to severance charges recorded during the quarter resulting from the termination of personnel in
the November 2005 corporate changes. Expenditures have continued to decline since Q2 2006 due to reduced headcount as well as reduced consulting,
patent costs and investor relation costs.
Net loss decreased in Q3 and Q4 of 2006
as the result of reduced research and development and general and administrative expenditures. During the quarter ended May 31, 2006, research and
development expenditures were significantly lower than prior periods as minimal clinical, regulatory and compliance costs related to Virulizin were
incurred vs. prior quarters when work associated with the Phase III clinical trial will still ongoing. General and administrative expenses decreased
during the three months ended May 31, 2006 compared with prior quarters due to headcount reductions (in November 2005) as well as lower legal and
corporate communication costs associated with budget cutbacks.
Research and development expenditures
decreased in the quarter ended May 31, 2005 compared with prior quarters due to lower clinical trial costs associated with the Virulizin Phase III
clinical trial as it wound down to last patient visit in July 2005. General and administrative expenditures decreased slightly during the three month
period ended May 31, 2005 compared with prior periods due to lower consulting costs.
| |
Fiscal 2006 Quarter Ended |
|
Fiscal 2005 Quarter Ended |
|
|||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (Amounts in 000’s except for per common share data) |
|
May 31, 2006 |
|
Feb. 28, 2006 |
|
Nov. 30, 2005 |
|
Aug. 31, 2005 |
|
May 31, 2005 |
|
Feb. 28, 2005 |
|
Nov. 30, 2004 |
|
Aug. 31, 2004 |
|||||||||||||||||||
Revenue |
$ | 14 | $ | 5 | $ | 6 | $ | 1 | $ | — | $ | 3 | $ | 1 | $ | 2 | |||||||||||||||||||
Research and
development |
1,353 | 2,296 | 2,631 | 3,957 | 2,332 | 3,175 | 3,838 | 5,049 | |||||||||||||||||||||||||||
General and
administrative |
730 | 909 | 1,619 | 1,076 | 1,506 | 1,484 | 1,333 | 1,025 | |||||||||||||||||||||||||||
Net
loss |
(2,970 | ) | (4,095 | ) | (5,102 | ) | (5,742 | ) | (4,598 | ) | (5,274 | ) | (5,945 | ) | (6,245 | ) | |||||||||||||||||||
Basic and
diluted net loss per share |
$ | (0.02 | ) | $ | (0.02 | ) | $ | (0.03 | ) | $ | (0.03 | ) | $ | (0.03 | ) | $ | (0.03 | ) | $ | (0.03 | ) | $ | (0.04 | ) | |||||||||||
Cash used in
operating activities |
$ | (1,940 | ) | $ | (3,956 | ) | $ | (2,360 | ) | $ | (4,809 | ) | $ | (3,789 | ) | $ | (4,106 | ) | $ | (4,966 | ) | $ | (5,860 | ) | |||||||||||
Disclosure Controls and
Procedures
Disclosure controls and procedures are
designed to provide reasonable assurance that all material information required to be publicly disclosed by a public company is gathered and
communicated to management, including the certifying officers, on a timely basis so that appropriate decisions can be made regarding public disclosure.
As at the end of May 31, 2006, the certifying officers and other members of management evaluated the effectiveness of our disclosure controls and
procedures (as this term is defined in the rules adopted by Canadian
37
securities regulatory authorities and the United States Securities and Exchange Commission). This evaluation included a review of our existing disclosure and insider trading policy, compliance with regard to that policy, the disclosure controls currently in place surrounding our interim and annual financial statements, MD&A and other required documents and discussions with management surrounding the process of communicating material information to management and in turn the certifying officers and all procedures taking into consideration the size of the company and the number of employees. Based on the evaluation described above, the certifying officers have concluded that, as of May 31, 2006, the disclosure controls and procedures were effective to provide reasonable assurance that the information we are required to disclose on a continuous basis in annual and interim filings and other reports is recorded, processed, summarized and reported or disclosed on a timely basis as required.
Outstanding Share
Data
As at September 30, 2006, the number of
issued and outstanding common shares of the Company was 209,625,949. In addition, there were 3,000,000 warrants to purchase 3,000,000 common shares of
the Company and 12,665,000 stock options outstanding that can be exercised into an equal number of common shares. The convertible debentures are
convertible into 15,000,000 common shares of the Company.
B. |
Liquidity and capital resources |
Since its inception, Lorus has financed
its operations and technology acquisitions primarily from equity and debt financing, the exercise of warrants and stock options, and interest income on
funds held for future investment. We expect to continue to finance the GTI-2501 Phase II clinical trial and the development of our small molecule
program from internal resources until their anticipated completion. The ongoing costs of the six GTI-2040 Phase II clinical trials will continue to be
borne by the US NCI with Lorus continuing to be responsible for any additional GTI-2040 manufacturing costs.
We have not earned substantial revenues
from our drug candidates and are therefore considered to be in the development stage. The continuation of our research and development activities and
the commercialization of the targeted therapeutic products are dependent upon our ability to successfully finance and complete our research and
development programs through a combination of equity financing and payments from strategic partners. We have no current sources of payments from
strategic partners. In addition, we will need to repay or refinance the secured convertible debentures on their maturity should the holder not choose
to convert the debentures into common shares. There can be no assurance that additional funding will be available at all or on acceptable terms to
permit further clinical development of our products or to repay the convertible debentures on maturity. If we are not able to raise additional funds,
we may not be able to continue as a going concern and realize our assets and pay our liabilities as they fall due. The financial statements do not
reflect adjustments that would be necessary if the going concern assumption were not appropriate. If the going concern basis were not appropriate for
these financial statements, then adjustments would be necessary in the carrying value of the assets and liabilities, the reported revenues and expenses
and the balance sheet classifications used.
Our current level of cash and
short-term investments and the additional funds available upon the successful closing of the subscription agreements (described below) are sufficient
to execute our current planned expenditures for the next twelve months.
Operating Cash
Requirements
Lorus utilized cash in operating
activities of $13.1 million in 2006 compared with $18.7 million in 2005 and $28.1 million in 2004. The decrease in cash used in operating activities in
2006 is due to lower research and development and general and administrative expenses, as described above, offset by lower interest income. The
significant decrease in cash used in operating activities in 2005 compared with 2004 is due to lower research and development expenses, offset by lower
interest income.
Cash
Position
At May 31, 2006, Lorus had cash and
cash equivalents and short-term investments totalling $8.3 million compared to $21.5 million at the end of 2005. The Company invests in highly rated
and liquid debt instruments.
38
Investment decisions are made in accordance with an established investment policy administered by senior management and overseen by the Board of Directors. Working capital (representing primarily cash and cash equivalents and short term investments) at May 31, 2006 was $5.8 million as compared to $18.5 million at May 31, 2005. As discussed below, subsequent to year-end, we entered into subscription agreements to raise gross proceeds of $12.2 million through the issuance of 33.8 million common shares of Lorus. Cash and short-term investments will therefore increase by $12.2 million in gross proceeds.
We do not expect to generate positive
cash flow from operations in the next several years due to additional research and development costs, including costs related to drug discovery,
preclinical testing, clinical trials, manufacturing costs and operating expenses associated with supporting these activities. Negative cash flow will
continue until such time, if ever, that we receive regulatory approval to commercialize any of our products under development and revenue from any such
products exceeds expenses.
We may seek to access the public or
private equity markets from time to time, even if we do not have an immediate need for additional capital at that time. We intend to use our resources
to fund our existing drug development programs and develop new programs from our portfolio of preclinical research technologies. The amounts actually
expended for research and drug development activities and the timing of such expenditures will depend on many factors, including the progress of the
Company’s research and drug development programs, the results of preclinical and clinical trials, the timing of regulatory submissions and
approvals, the impact of any internally developed, licensed or acquired technologies, our ability to find suitable partnership agreements to assist
financially with future development, the impact from technological advances, determinations as to the commercial potential of the Company’s
compounds and the timing and development status of competitive products.
Financing
On July 13, 2006 we entered into an
agreement with High Tech Beteiligungen GmbH & Co. KG (“High Tech”) to issue 28.8 million common shares at $0.36 per share for gross
proceeds of $10.4 million. The subscription price represented a premium of 7.5% over the closing price of the common shares on the TSX on July 13,
2006. The transaction closed on August 30, 2006. In connection with the closing, we also entered into a registration rights agreement with High Tech
that provides, among other things, that High Tech will have a demand right to request, an aggregate number of five times, the registration or
qualification of the Purchased Shares for resale in the United States and Canada, subject to certain restrictions. High Tech has also been granted
piggy-back rights to enable it to sell the Purchased Shares in connection with a public offering of shares to Lorus, subject to certain exceptions. In
addition, pursuant to the Share Purchase Agreement, High Tech will have the right to nominate one nominee on the board of directors of Lorus or, if it
does not have a nominee, it will have the right to appoint an observer to the board for as long as it owns shares.
In accordance with the terms of the
Share Purchase Agreement, Lorus agreed not to issue any common shares or securities convertible into common shares, subject to certain limited
exceptions, until July 31, 2007, at a price of less than $0.36 per common share. In addition, certain named executive officers of Lorus signed
“lock-up” agreements whereby they agreed not to dispose of their common shares for a period of 30 days following the closing date, and for
the 30 days immediately following such 30 day period, they agreed not to dispose greater than 50% of the aggregate number of common shares they hold as
at the closing date.
On July 24, 2006 we entered into an
agreement with Technifund Inc. to issue on a private placement basis, 5 million common shares at $0.36 per share for gross proceeds of $1.8 million.
The transaction closed on August 31 2006.
On October 6, 2004, we entered into an
agreement to raise aggregate net proceeds of $13.9 million through the issuance of secured convertible debentures and warrants. The debentures are
secured by a first charge over all of the assets of the Company. We received $4.4 million on October 6, 2004 (representing a $5.0 million debenture
less an investor fee representing 4% of the $15.0 million to be received under the agreement), and $5.0 million on each of January 14 and April 15,
2005. All debentures issued under this agreement are due on October 6, 2009 and are subject to interest payable monthly at a rate of prime +1% until
such time as the Company’s share price reaches $1.75 for 60 consecutive trading days, at which time, interest will no longer be charged. Interest
is payable in common shares of Lorus until Lorus’ shares trade at a price of $1.00 or more after which interest will be payable
in
39
cash or common shares at the option of the debenture holder. Common shares issued in payment of interest will be issued at a price equal to the weighted average trading price of such shares for the ten trading days immediately preceding their issue in respect of each interest payment. For the year ended May 31, 2006, the Company has issued 2,153,000 common shares in settlement of $882 thousand in interest. For the year ended May 31, 2005 the Company issued 421,000 common shares in settlement of $300 thousand in interest.
The $15.0 million principal amount of
debentures is convertible at the holder’s option at any time into common shares of the Company with a conversion price per share of
$1.00.
The Company issued to the debt holder
3,000,000 warrants expiring October 6, 2009 to buy common shares of the Company at a price per share equal to $1.00.
In addition, in 2005, Lorus issued
common shares on the exercise of stock options for proceeds of $112 thousand.
On June 11, 2003, Lorus raised net
proceeds of $29.9 million by way of a public offering of 26,220,000 units at a price of $1.25 per unit, each unit consisting of one common share and
one-half of one purchase warrant. In 2004, Lorus issued common shares on the exercise of stock options for proceeds of $171 thousand.
Use of
Proceeds
In our prospectus dated June 3, 2003,
we indicated that the proceeds to be received from that financing would be used as follows: $12 million for the product development of our
immunotherapy platform, $11 million for the product development of our antisense platform and $2 million for preclinical and discovery programs. It was
anticipated that the balance of funding would be used for working capital and general purposes. Since the date of the prospectus, we have incurred
$38.0 million in research and development expenses on our immunotherapy platform, $11.6 million on our antisense platform, and $1.8 million on
preclinical and discovery programs. The additional spending on our immunotherapy platform was funded through cash and short term investments held by
the Company prior to the 2003 offering, as well as the October 6, 2004 $15.0 million convertible debenture financing, and is the direct result of the
expansion of the Virulizin® Phase III clinical trial. The spending anticipated in the 2003 prospectus on our antisense platform and preclinical
and discovery programs was to be incurred over a number of years, including 2004, 2005 and 2006. We have sufficient funds available at the end of 2006
to fund the remaining $200 thousand to be spent on preclinical and discovery programs.
Subsequent
Events
On July 13, 2006 we entered into an
agreement with HighTech Beteiligungen GmbH & Co. KG (“HighTech”) to issue 28.8 million common shares at $0.36 per share for gross
proceeds of $10.4 million. The subscription price represented a premium of 7.5% over the closing price of the common shares on the Toronto Stock
Exchange on July 13, 2006. The transaction closed on August 30, 2006. In connection with the transaction, HighTech received demand registration rights
that will enable HighTech to request the registration or qualification of the common shares for resale in the United States and Canada, subject to
certain restrictions. These demand registration rights will expire on June 30, 2012. In addition, HighTech has the right to nominate one nominee to the
board of directors of Lorus or, if it does not have a nominee, it will have the right to appoint an observer to the board. Upon completion of the
transaction, HighTech will hold approximately 14% of the issued and outstanding common shares of Lorus Therapeutics Inc.
On July 24, 2006 Lorus entered into an
agreement with Technifund Inc. to issue on a private placement basis, 5 million common shares at $0.36 per share for gross proceeds of $1.8 million.
The transaction closed on August 31, 2006.
On September 19, 2006 the Company
announced that Dr. Jim A Wright would step down as the President and Chief Executive Officer of Lorus effective September 21, 2006. The departure of
Dr. Wright resulted in a liability based on a mutual separation agreement executed subsequent to the quarter end of approximately $500 thousand. The
amount is expected to be paid by the end of the third quarter 2007.
40
C. |
Research and development, patents and licenses, etc. |
Certain information concerning research
and development and intellectual property is set forth in Item 4, “Information of the Company”
D. |
Trend information |
The Company does not currently know of
any material trends that would be material to our operations.
E. |
Off-balance sheet arrangements |
As at May 31, 2006, we have not entered
into any off-balance sheet arrangements.
F. |
Tabular disclosure of contractual obligations |
As at May 2006, we had contractual
obligations requiring annual payments as follows:
| (Amounts in 000s) |
Less than 1 year |
1-3 years |
4-5 years |
5+ years |
Total |
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Operating
leases |
139 | 126 | — | — | 265 | |||||||||||||||||
Convertible
Debentures1 |
— | — | 15,000 | — | 15,000 | |||||||||||||||||
Total |
139 | 126 | 15,000 | — | 15,265 | |||||||||||||||||
1 The convertible debentures as described above may be converted into common shares of Lorus at a conversion price of
$1.00 per share. In the event that the holder does not convert the debentures, Lorus has an obligation to repay the $15.0 million in
cash.
Reference Information
Item
6. |
Directors, Senior Management and Employees |
A. |
Directors and Senior Management |
The following table and notes thereto
provide the name, province or state and country of residence, positions with the Company and term of office of each person who serves as a director or
executive officer of Lorus as at the date hereof.
Each director has been elected or
appointed to serve until the next annual meeting or until a successor is elected or appointed. We have an Audit Committee, a Nominating and Corporate
Governance Committee, a Compensation Committee and an Environment, Health and Safety Committee; the members of each such committee are shown below. As
at May 31, 2006, our directors and executive officers, as a group, beneficially owned, directly or indirectly, or exercised control over 4,631,000 or
approximately 2.7% of our common shares.
| Name, Province/State and Country of Residence |
Position |
Director or Officer Since |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
J.
KEVIN BUCHI(1) (3) Pennsylvania, United States |
Director |
December
2002 |
||||||||
DONALD W. PATERSON(1)(3) Ontario, Canada |
Director |
July
1991 |
||||||||
ALAN STEIGROD(2) Florida, United States |
Director |
May
2001 |
||||||||
GRAHAM STRACHAN(1)(3)(4) Ontario, Canada |
Chairman, Director |
May
2001 |
||||||||
41
DR. JIM WRIGHT Ontario, Canada |
Former President and Chief Executive Officer, Director |
October
1999 |
||||||||
GEORG LUDWIG(2) Eschen, Liechtenstein |
Director |
September
2006 |
||||||||
DR. MICHAEL MOORE(2) Surrey, United Kingdom |
Director |
September
2006 |
||||||||
DR. AIPING YOUNG(4) Ontario, Canada |
President and Chief Executive Officer, Director |
October
1999 |
||||||||
ELIZABETH WILLIAMS Ontario, Canada |
Director of Finance and Acting Chief Financial Officer |
November
2005 |
||||||||
(1) |
Member of Audit Committee. |
(2) |
Member of the Compensation Committee. |
(3) |
Member of the Nominating and Corporate Governance Committee. |
(4) |
Member of Environment, Health and Safety Committee. |
The principal occupation and employment
of each of the foregoing persons for the past five years is set forth below:
J. Kevin Buchi: Mr. Buchi is
executive vice president and chief financial officer of Cephalon, Inc., an international biopharmaceutical company. Mr. Buchi is responsible for
finance, accounting, manufacturing and information systems and has been involved in raising significant financing for Cephalon. He is a certified
public accountant and has received a master’s degree in management from the J.L. Kellogg Graduate School of Management at Northwestern
University.
Donald W. Paterson: Mr. Paterson
is President of Cavandale Corporation, a corporation principally engaged in providing strategic corporate consulting to emerging growth companies
within the technology industry.
Alan Steigrod: Mr. Steigrod is
Managing Director of Newport Healthcare Ventures, a consulting firm for the healthcare industry, located in Newport Beach, California.
Graham Strachan: From 2002 to
the present, Mr. Strachan has been the President of GLS Business Development Inc., a life-science consulting firm located in Etobicoke, Ontario. From
1986 to 2002, Mr. Strachan was the President and Chief Executive Officer of Allelix Biopharmaceuticals Inc.
Georg Ludwig: Mr. Ludwig is the
Managing Director of ConPharm Anstalt, a consulting and management company specializing in life sciences funds. Prior to January 2005, Mr. Ludwig was a
Managing Director at HighTech Private Equity, a leading European venture capital fund focused exclusively on providing financial support for the
development of innovative products based on applied technologies and life sciences.
Dr. Michael Moore: Dr. Moore is
currently the Chief Executive Officer of Piramed Limited, a UK-based biopharmaceutical company with a focus on novel classes of small molecule,
anti-inflammatory and anti-tumour agents. Prior to joining Piramed in August 2003, Dr. Moore held several progressive positions in the
biopharmaceutical industry (1988-2003), culminating in the position of Chief Scientific Officer and Research Director at Xenova Group
plc.
Dr. Jim Wright: Dr. Wright
stepped down as Lorus’ President and CEO effective September 21, 2006 and continues to serve as a Director. Dr. Wright co-founded GeneSense in
1996, and served as its President, Chief Scientific Officer and a director before becoming our President and Chief Scientific Officer in October 1999
on our acquisition of GeneSense. Dr. Wright served as the Company’s CEO since October 2001.
Dr. Aiping Young: Dr. Young was
appointed President and CEO effective September 21, 2006 and as a Director on October 6, 2006. Prior to her appointment, Dr. Young had been our Chief
Operating Officer since
42
November 20, 2003 and was a cofounder with Dr. Wright of GeneSense Technologies Inc. Dr. Young previously held the position of Senior Vice President, Research and Development and Chief Technical Officer at Lorus.
Elizabeth Williams: Prior to
joining Lorus in July 2004, Ms. Williams was an Audit Manager with Ernst and Young LLP. Ms. Williams is a chartered accountant and has received a
bachelor’s degree in business administration. Ms. Williams lectured on introductory auditing at Wilfrid Laurier University during
2005.
The following table outlines other
public company directorships of our directors:
| Board Member |
|
Company |
||||
|---|---|---|---|---|---|---|
Jim A.
Wright |
— |
|||||
Graham
Strachan |
Amorfix Biotechnologies Inc. Ibex Technologies Inc. |
|||||
J. Kevin
Buchi |
Encysive Pharmaceuticals Celator Pharmaceuticals |
|||||
Donald
Peterson |
ANGOSS Software Corporation NewGrowth Inc. Homeserve Technologies Inc. Utility Corporation |
|||||
Alan
Steigrod |
Sepracor Inc. Poniard Pharmaceuticals Inc. |
|||||
Michael
Moore |
— |
|||||
George
Ludwig |
— |
|||||
B. |
Compensation |
Summary of Executive Compensation
The following table provides a summary of compensation earned
during each of the last three fiscal years by our Chief Executive Officer, our Chief Financial Officer (or acting Chief Financial Officer) and for the
next three most highly compensated executive officers (the “named executive officers”). The figures are in Canadian dollars.
Summary Compensation Table
| Annual Compensation |
Long-Term Compensation Awards |
|||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Name and Principal Position |
Fiscal Year |
Salary ($) |
Bonus ($) |
Other Annual Compensation ($) |
Securities Under Options/ SARs Granted (#)(1) |
All Other Compensation ($) |
||||||||||||||||||||
DR. JIM A. WRIGHT(4) |
2006 | 345,442 | 53,000 | Nil | 947,500 | Nil | ||||||||||||||||||||
Former President
and Chief |
2005 | 313,586 | 95,760 | Nil | 228,000 | Nil | ||||||||||||||||||||
Executive
Officer |
2004 | 285,000 | 102,600 | Nil | 570,000 | Nil | ||||||||||||||||||||
DR. AIPING YOUNG(4) |
2006 | 259,692 | 32,000 | Nil | 1,194,144 | Nil | ||||||||||||||||||||
President and
Chief |
2005 | 222,697 | 46,125 | Nil | 250,000 | Nil | ||||||||||||||||||||
Executive
Officer |
2004 | 197,945 | 45,390 | Nil | 225,000 | Nil | ||||||||||||||||||||
43
MS. ELIZABETH WILLIAMS(2) |
2006 | 88,631 | 7,000 | Nil | 228,035 | Nil | ||||||||||||||||||||
Director of
Finance, Acting |
2005 | 84,163 | 7,990 | Nil | 52,388 | Nil | ||||||||||||||||||||
Chief Financial
Officer |
2004 | Nil | Nil | Nil | Nil | Nil | ||||||||||||||||||||
MR. PAUL VAN DAMME(3) |
2006 | 110,813 | 11,000 | Nil | Nil | 74,633 | ||||||||||||||||||||
Former Chief
Financial |
2005 | 152,654 | 35,030 | Nil | 202,500 | 37,000 | ||||||||||||||||||||
Officer |
2004 | Nil | Nil | Nil | Nil | Nil |
(1) Options granted are net of forfeitures.
(2) Ms. Williams started with Lorus on June 14, 2004; hence,
there are no amounts relating to Ms. Williams’ compensation for 2004.
(3) Mr. Van Damme started with Lorus on September 7, 2004; hence,
there are no amounts relating to Mr. Van Damme’s compensation for 2004. Mr. Van Damme resigned from his position on November 9, 2005. The amount
of “All Other Compensation” relates to a lump sum amount paid pursuant to our separation agreement with Mr. Van Damme.
(4) On September 21, 2006 Dr. Wright stepped down as Lorus’
President and Chief Executive Officer and was replaced by Dr. Aiping Young, the Company’s Chief Operating Officer. The 2006 compensation amounts
relate to Dr. Wright’s performance as the Company’s President and Chief Operating Officer and Dr. Young’s performance as the
Company’s Chief Operating Officer.
Directors’ Compensation
During the fiscal year ended May 31,
2006, each director who was not an officer of the Company or a representative of a shareholder, was entitled to receive 50,000 stock options (the Chair
received 100,000) and, at his election, common shares, deferred share units and/or cash compensation for attendance at the board of directors of the
Company (the “Board”) committee meetings. Compensation consisted of an annual fee of $15,000 (the Chair received $35,000) and $1,500 per
Board meeting attended ($4,500 to the Chair of a Board meeting). Members of the Audit Committee received an annual fee of $8,000 (the Chair received
$10,000). Members of the Compensation Committee received an annual fee of $5,000, and members of the Nominating and Corporate Governance Committee and
the Environment, Health and Safety Committee received annual fees of $4,000 (the Chair of each of the three committees received
$5,000).
In September 2005, stock options to
purchase 300,000 common shares at a price of $0.68 per share expiring September 13, 2015 were granted, in aggregate, to our directors. These options
vested 50% upon issuance and the remaining 50% will vest after one year. In addition, the Company reimbursed the directors for expenses incurred in
attending meetings of the Board and committees of the Board.
Directors are entitled to participate
in our Deferred Share Unit Plan. See “Equity Compensation Plans—Directors and Officers’ Deferred Share Unit Plan”.
Management Contracts
Under
the employment agreement with Dr. Jim A. Wright dated October 29, 1999, as amended,
Dr. Wright’s position was President and Chief Executive Officer of the
Company for an annual salary of $327,600. If within 24 months of a change of
control of Lorus, Dr. Wright’s employment was terminated without cause
or if his responsibilities were reduced, then he was entitled to receive the
equivalent of two years’ of his basic salary, two years of his annual bonus
(based on the average of the bonus granted to him in the three preceding years),
and two years of benefits. If Dr. Wright’s employment is terminated without
cause and not subsequent to a change of control of Lorus, Dr. Wright was entitled
to 18 months’ salary and benefits and 18 months’ of pro-rated annual
bonus (and he was required to mitigate his loss after the first 12 months and
account to Lorus for any severance payment beyond the first 12 months). The
employment agreement provided that the Company may at any time assign the Chief
Executive Officer to perform other functions that are consistent with his skills,
experience and position within the Company. Dr. Wright reported directly to the
Board. The bonus and options allocation of the President and Chief Executive
Officer was determined by the Board and was awarded based 100% on achievement
of corporate objectives.
44
In September 2006 the Company entered
into a mutual separation agreement with Dr. Jim Wright. This agreement provided 18 months of severance to Dr. Wright.
Under the employment agreement with Dr.
Aiping Young dated September 21, 2006, Dr. Young’s position is President and Chief Executive Officer of the Company for an annual salary of
$300,000. This agreement provides for a notice period equal to 18 months in the event of termination without cause or a resignation due to change in
role as a result of a change in control of the Company. The employment agreement provides that the Company may at any time assign the President and
Chief Executive Officer to perform other functions that are consistent with her skills, experience and position within the Company. Dr. Young reports
to the Board of Directors. The bonus and options allocation of the President and Chief Executive Officer is as recommended by the Board of Directors
and is awarded based 100% on achievement of corporate objectives. The agreement also provides for certain option grants including a one time grant of 1
million options vesting immediately, 1 million options of which the vesting is contingent upon certain events as well as an annual option grant of
500,000 options vesting upon the achievement of corporate objectives.
Under the employment agreement with Ms.
Elizabeth Williams dated May 31, 2004, Ms. Williams’ position is Director of Finance and Controller of the Company for an annual salary of
$89,000. This agreement provides for a notice period equal to the greater of one month and the applicable notice entitlement under employment
legislation in the event of termination. Ms. Williams reports to the Chief Executive Officer. The bonus and options allocation of the Director of
Finance is as recommended by the Chief Executive Officer.
Vacation allocation on a calendar year
basis for each Named Executive Officer is four weeks of paid vacation, pro rated to reflect a period of employment less than a full calendar year.
Salary and bonus amounts for each of the Named Executive Officers for the fiscal year 2006 were as set out in the above Summary Compensation
Table.
The following table sets forth certain
details as at the end of the last fiscal year ended May 31, 2006 with respect to compensation plans pursuant to which equity securities of the Company
are authorized for issuance.
| Plan Category |
# of Common Shares to be issued upon exercise of outstanding options |
Weighted-average exercise price of outstanding options |
# of Common Shares remaining available for future issuance under the equity compensation plans |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Plans
approved by Shareholders(1) |
10,300,000 | $ | 0.70 | 7,832,390 | ||||||||||
Plans not
approved by Shareholders |
— | — | — | |||||||||||
Total |
10,300,000 | $ | 0.70 | 7,832,390 | ||||||||||
(1) |
This includes options granted and reserved for issuance pursuant to our amended 1993 Stock Option Plan, amended 2003 Stock Option Plan and our Alternate Compensation Plan. |
Equity Compensation Plans
Stock Option
Plans
Our original stock option plan was
established in 1993 (the “1993 Stock Option Plan”); however, due to significant developments in the laws relating to share option plans and
our future objectives, in November 2003 we created a new stock option plan (the “2003 Stock Option Plan”, and together with the 1993 Stock
Option Plan, the “Stock Option Plans”), ratified by our shareholders, pursuant to which all future grants of stock options would be
made.
On January 1, 2005, the TSX amended its
rules (the “TSX Rules”) to provide that, among other things, the maximum number of shares issuable under a stock option plan of a TSX issuer
may be a rolling number based on a
45
fixed percentage of the number of outstanding shares of such issuer from time to time. Previously, the TSX Rules required a stock option plan to have a fixed number of shares issuable thereunder. The amended TSX Rules require that a stock option plan with a rolling maximum be approved by the shareholders of an issuer every three years.
At our last annual meeting held on
September 13, 2005, shareholders of the Company approved an amendment to the Stock Option Plans to provide that the number of shares available for
issue is a rolling rate of 15% of the issued common shares of the Company. This amendment increased the number of options issuable under the Stock
Option plans from 20,582,081 to 25,921,000. Shareholders also approved amendments to remove all prior limits on grants of options and issuance of
common shares to any one individual and for individual insiders under the 1993 Stock Option Plan and 5% limits for individual insiders under the 2003
Stock Option Plan, and to replace such limits with the 10% limit for insiders as a group as provided under the amended TSX Rules.
The Stock Option Plans enables us to
grant share options to employees, directors, and individuals in special contract relationships. The number of common shares reserved issuable pursuant
to the Stock Option Plans is currently at a maximum of 25,921,000 common shares, amounting to 14.8 per cent of the common shares outstanding as of May
31, 2006. Of this amount a total of 7,832,390 options to acquire common shares are at May 31, 2006, outstanding under the Stock Option Plans. This
amounts to 4.8 per cent of the common shares outstanding.
Our Board, within certain limitations,
determines the terms, conditions and limitations of options granted under the Stock Option Plans. Under the Stock Option Plans, (a) the number of
common shares reserved for insiders, at any time, under all share compensation arrangements, cannot exceed 10 per cent of issued and outstanding common
shares and (b) the number of common shares issued to any one insider, within any one year period, under all share compensation arrangements, cannot
exceed 10 per cent of issued and outstanding common shares and (c) the number of common shares reserved for issuance under all share compensation
arrangements may not exceed 5 per cent of issued and outstanding common shares. The Stock Option Plans may be amended by our Board for the proper
administration of such plan, subject to regulatory approval, if required. The exercise price per common share is determined by the Board but shall not
be less than the closing price of the Common shares on the TSX on the day prior to the day on which the option is granted. The Board fixes the term of
each option when the option is granted, but may not be greater than 10 years from the date on which the option is granted. In general, the right of an
optionee to exercise an option commences on the first anniversary date of the option grant and the optionee is entitled to purchase, on a cumulative
basis, 50 per cent after the first year and 25 per cent of the optioned common shares in each of the next two years. However, in certain circumstances,
options are granted entitling an optionee to purchase 100 per cent of the common shares earlier than the general pattern. In the event that our
relationship with an optionee terminates, the provisions of the Stock Option Plans specify the applicable period for exercising options dependent upon
the event giving rise to the termination and the position of the optionee with Lorus. Entitlement to unvested options ceases immediately upon
termination of employment and entitlement to vested options ceases three months after employment is terminated. The Board is entitled to provide
exceptions to this termination as deemed appropriate. For all optionees, entitlement continues for nine months in the event of death. The ability of an
optionee to exercise an option under the Stock Option Plans may be accelerated in the event of a change of control of Lorus. The exercise price per
Common Share is payable in full on the date of exercise. Options granted under the Stock Option Plans are not assignable.
During the period June 1 to May 31,
2006, options to purchase 6,721,000 common shares were granted under the Stock Option Plans at exercise prices between $0.26 and $0.78 per Common
Share. During the year ended May 31, 2006, we granted options to employees, other than executive officers of the Company, to purchase 2,912,706 common
shares, being 43.3% of the total incentive stock options granted during the year to employees and executive officers.
Performance Based Compensation
Plans
Executive officers of the Company are
eligible to participate in a performance related compensation plan (the “Compensation Plan”). The Compensation Plan provides for
potential annual cash bonus payments and annual granting of options to purchase common shares under our 2003 Stock Option Plan. The potential annual
cash bonus and annual granting of options to each executive officer are conditional upon the achievement by the Company and each executive officer of
predetermined objectives reviewed by the Compensation Committee and approved by the Board. See “Compensation Committee” and “Report on
Executive Compensation”.
46
Directors and Officers’
Alternate Compensation Plan
The Directors’ and Officers’
Alternate Compensation Plan was terminated by resolution of the Board, effective September 13, 2005.
Employee Share Purchase
Plan
In November 2004, the Board adopted the
Employee Share Purchase Plan (“ESPP”), effective January 1, 2005. For the year ended May 31, 2006, a total of 293,000 common shares had been
purchased by employees and executive officers under the ESPP at prices per share between $0.2295 and $0.578 per Common Share. During fiscal 2006, under
the ESPP, executive officers as a group purchased 90,000 common shares at a weighted average purchase price of $0.31 per Common Share and employees,
excluding executive officers, as a group purchased 203,000 common shares at an average exercise price of $0.29 per Common Share.
The purpose of the ESPP is to assist
the Company to retain the services of its employees, to secure and retain the services of new employees and to provide incentives for such persons to
exert maximum efforts for the success of the Company. The ESPP provides a means by which employees of the Company and its affiliates may purchase
common shares at a 15% discount through accumulated payroll deductions. Eligible participants in the ESPP include all employees, including executive
officers, who work at least 20 hours per week and are customarily employed by the Company or an affiliate of the Company for at least six months per
calendar year. Generally, each offering is of three months’ duration with purchases occurring every month. Participants may authorize payroll
deductions of up to 15% of their base compensation for the purchase of common shares under the ESPP.
Deferred Profit Sharing
Plan
We have a Deferred Profit Sharing Plan
(“DPSP”) matching program which is available to all employees. The DPSP matching program provides 100% matching of employee contributions
into each employee’s Group RRSP account up to a maximum of 3% of the employee’s gross earnings. We began making contributions to the
employees’ Group Retirement Savings Plan in fiscal 1998. Beginning February 2001, our contributions have been paid into an employer-sponsored
DPSP.
Directors’ and
Officers’ Deferred Share Unit Plan
We have a deferred share unit plan for
directors and officers (the “Deferred Share Unit Plan”). Under the Deferred Share Unit Plan, participating directors may elect to receive
either a portion or all of their annual fees for acting as a director (“Annual Fees”) from us in deferred share units. Under the Deferred
Share Unit Plan, the Compensation Committee may at any time during the period between the annual meetings of our shareholders, in its discretion
recommend the Company credit to each participating director who has elected under the terms of the Deferred Share Unit Plan, the number of units equal
to the gross amount of the Annual Fees to be deferred divided by the fair market value of the common shares. The fair market value of the common shares
is determined as the closing price of the common shares on the TSX on the day immediately preceding such recommendation by the Compensation Committee
or such other amount as determined by the Board and permitted by the stock exchanges or other market(s) upon which the common shares are from time to
time listed for trading and by any other applicable regulatory authority (collectively, the “Regulatory Authorities”).
In addition, the participating
directors may elect under the Deferred Share Unit Plan to receive deferred share units in satisfaction for meeting fees earned by the Participating
Directors as a result of attendance at meetings of the Board held between the annual meetings of our shareholders by the credit to each Participating
Director of the number of units equal to the gross amount of the meeting fees to be deferred divided by the fair market value of the common shares,
being the closing price of the common shares on the TSX on the day immediately preceding the recommendation by the Compensation Committee or such other
amount as determined by the Board and permitted by the Regulatory Authorities.
The Deferred Share Unit Plan is
administered by the Board (in consultation with the Compensation Committee) and, subject to regulatory requirements, may be amended by the Board
without shareholder approval. When a participating director ceases to hold the position of director and is no longer otherwise employed by us,
the
47
participating director receives either (a) a lump sum cash payment equal to the number of deferred share units held multiplied by the then fair market value of the common shares on the date of termination, or (b) the number of common shares that can be acquired in the open market with the amount described in (a), either case being subject to withholding for income tax. The Board may terminate the Deferred Share Unit Plan any time before or after any allotment or accrediting of deferred share units thereunder.
Option Grants During Fiscal Year 2006
The following tables set forth the
options granted to and exercised by each of the Named Executive Officers during the year ended May 31, 2006:
Option/SAR Grants During the Most Recently Completed
Financial Year
| Name and Principal Position |
|
Securities Under Options/SARs Granted (#)(1) |
|
% of Total Options/SARs Granted to Employees in Financial Year (%) |
|
Exercise or Base Price ($/Security) |
|
Market Value of Securities Underlying Options/SARs on the Date of Grant ($/Security) |
|
Expiration Date |
||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
DR. JIM A. WRIGHT(5) |
300,000 | (2) | 2.08 | 0.78 | 0.78 | July 19, 2015 | ||||||||||||||||
Former President
and Chief |
807,500 | (3) | 12.01 | 0.30 | 0.30 | Oct. 10, 2010 to | ||||||||||||||||
Executive
Officer |
July 19, 2015 | |||||||||||||||||||||
DR. AIPING YOUNG(5) |
208,333 | (2) | 3.10 | 0.78 | 0.78 | July 19, 2015 | ||||||||||||||||
President and
Chief |
75,000 | (4) | 1.12 | 0.78 | 0.78 | July 19, 2015 | ||||||||||||||||
Executive
Officer |
50,000 | 0.74 | 0.26 | 0.26 | Nov. 30, 2015 | |||||||||||||||||
| 50,000 | 0.74 | 0.30 | 0.30 | Jan. 5, 2016 | ||||||||||||||||||
| 200,000 | (2) | 2.98 | 0.30 | 0.30 | Jan. 5, 2016 | |||||||||||||||||
| 610,811 | (3) | 9.09 | 0.30 | 0.30 | Oct. 10, 2010 to July 19, 2015 |
|||||||||||||||||
MS. ELIZABETH WILLIAMS |
54,487 | (4) | 0.81 | 0.78 | 0.78 | July 19, 2015 | ||||||||||||||||
Director of
Finance, Acting |
50,000 | 0.74 | 0.26 | 0.26 | Nov. 30, 2015 | |||||||||||||||||
Chief Financial
Officer |
50,000 | 0.74 | 0.30 | 0.30 | Jan. 5, 2016 | |||||||||||||||||
| 20,000 | 0.30 | 0.30 | 0.30 | Jan. 5, 2016 | ||||||||||||||||||
| 53,548 | (3) | 0.80 | 0.30 | 0.30 | July 20, 2014 to July 19, 2015 |
|||||||||||||||||
MR. PAUL VAN DAMME |
Nil | Nil | Nil | Nil | Nil | |||||||||||||||||
Former Chief
Financial Officer |
||||||||||||||||||||||
(1) |
Options granted are net of forfeitures. |
(2) |
These options are incentive options granted to certain Named Executive Officers to purchase common shares. The options vest immediately upon the attainment of specific undertakings; failing to achieve the undertakings will result in forfeiture on the specified deadline. |
(3) |
These options were granted as an incentive to senior management. Options granted represented 50% of the options held by the individual prior to January 6, 2006. |
(4) |
These options were granted on July 20, 2005 in respect of corporate and personal performance during the year ended May 31, 2006. The options vest on the basis of 50% on the first anniversary and 25% on the second and third anniversaries of the date of granting. |
(5) |
On September 21, 2006 Dr. Wright stepped down as Lorus’ President and Chief Executive Officer and was replaced by Dr. Aiping Young, the Company’s Chief Operating Officer. The 2006 option amounts relate to Dr. |
48
| Wrights performance as the Company’s President and Chief Operating Officer and Dr. Young’s performance as the Company’s Chief Operating Officer. |
Aggregated Option/SAR Exercises During the Most
Recently Completed
Financial Year and Financial Year-End Option/SAR Values
Financial Year and Financial Year-End Option/SAR Values
| Name |
|
Securities Acquired on Exercise (#) |
|
Aggregate Value Realized ($) |
|
Unexercised Options/SARs at May 31, 2006 (#) Exercisable/ Unexercisable |
|
Value of Unexercised in-the-Money Options/SARs at May 31, 2006 ($) Exercisable/ Unexercisable |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
DR. JIM A. WRIGHT Former President and Chief Executive Officer |
Nil | Nil | 2,310,000/112,500 |
38,500/1,875 |
||||||||||||||
DR. AIPING YOUNG President and Chief Executive Officer |
Nil | Nil | 1,644,941/487,500 |
29,228/19,812 |
||||||||||||||
MS. ELIZABETH WILLIAMS Acting Chief Financial Officer |
Nil | Nil | 39,921/241,022 |
655/10,817 |
||||||||||||||
MR. PAUL VAN DAMME Former Chief Financial Officer |
Nil | Nil | Nil |
Nil |
||||||||||||||
C. |
Board Practices |
Lorus is authorized to have a board of
at least one director and no more than ten. Lorus currently has five directors. Directors are elected for a term of about one year, from annual meeting
to annual meeting, or until an earlier resignation, death or removal. Each officer serves at the discretion of the Board or until an earlier
resignation, death or removal. There are no family relationships among any of our directors or officers.
Committees of the Board of Directors
The Company has an Audit Committee, a
Nominating and Corporate Governance Committee, a Compensation Committee and an Environment, Health and Safety Committee.
The members of these committees are as
follows:
Audit
Committee: |
J. Kevin Buchi,
Donald W. Paterson and Graham Strachan |
|||||
Compensation
Committee: |
Alan Steigrod,
Georg Ludwig and Michael Moore |
|||||
Nominating and Corporate
Governance Committee: |
Donald W.
Paterson, Graham Strachan and J. Kevin Buchi |
|||||
Environment, Health and
Safety Committee: |
Graham Strachan
and Dr. Aiping Young |
|||||
Compensation Committee.
Our board of directors, upon the advice
of the Compensation Committee, determines executive compensation. During the period from September 13, 2005 to June 30, 2006 the Compensation Committee
was comprised of three independent directors, Mr. Buchi, Mr. Strachan and Mr. Steigrod. Mr. Steigrod is the Chair of the Compensation Committee. The
Compensation Committee met three times during the above period.
49
Compensation Committee
Mandate
The Compensation Committee’s
mandate is to review, and advise the board of directors on, the recruitment, appointment, performance, compensation, benefits and termination of
executive officers. The Compensation Committee also administers and reviews procedures and policies with respect to our stock option plan, employee
benefit programs, pay equity and employment equity. The philosophy of the Compensation Committee regarding executive officer compensation is to reward
performance and to provide a total compensation package that will attract and retain qualified, motivated and achievement oriented executive
officers.
The Compensation Committee attempts to
create compensation arrangements that will align the interests of our executive officers and our shareholders. The key components of executive officer
compensation are base salary, potential annual cash bonuses and annual participation in the stock option plan.
Audit Committee.
Pursuant to Canadian securities laws,
our board of directors has determined that Messrs. Buchi, Paterson and Strachan are financially literate, as all have experience in reviewing and
analysing the financial reports and ascertaining the financial position of a corporation. Mr. Buchi is a certified public accountant and holds the
position of Chief Financial Officer in a public pharmaceutical company. Pursuant to United States securities laws, Mr. Buchi is also an audit committee
“financial expert”. Mr. Paterson, in his position as President of Cavandale Corporation, is educated and experienced in reading and analyzing
financial statements. Mr. Strachan has experience reading and analysing financial statements both as President of his own life science consulting firm
and in a prior position as President, Chief Executive Officer and a director of a biopharmaceutical company. Additionally, we believe that all three
members of the audit committee qualify as “independent” as that term is defined in the relevant Canadian and United States securities laws
and stock exchange rules relating to the composition of the audit committee.
Audit Committee
Mandate
The Audit Committee’s mandate is
to assist the board of directors in fulfilling its oversight responsibilities. In particular, the Audit Committee:
(a) |
serves as an independent and objective party to monitor the integrity of our financial reporting process and systems of internal controls regarding finance, accounting, and legal compliance, including the review of our financial statements, MD&A and annual and interim results; |
(b) |
identifies and monitors the management of the principal risks that could impact our financial reporting; |
(c) |
monitors the independence and performance of our independent auditors, including the pre-approval of all audit fees and all permitted non-audit services; |
(d) |
provides an avenue of communication among the independent auditors, management, and our board of directors; and |
(e) |
encourages continuous improvement of, and foster adherence to, our policies, procedures and practices at all levels. |
The Audit Committee is also responsible
for implementing and overseeing our whistle-blowing procedures.
D. |
Employees |
As at May 31, 2006, we employed 33
full-time persons and one part-time person in research and drug development and administration activities. Of our employees, nine hold Ph.D.s. To
encourage a focus on achieving long-term performance, employees and members of the board of directors have the ability to acquire an
ownership
50
interest in the Company through Lorus’ stock option plan and employees can participate in the employee share purchase plan, which was established in 2005.
Our ability to develop commercial
products and to establish and maintain our competitive position in light of technological developments will depend, in part, on our ability to attract
and retain qualified personnel. There is a significant level of competition in the marketplace for such personnel. We believe that to date we have been
successful in attracting and retaining the highly skilled personnel critical to our business. We have also chosen to outsource activities where skills
are in short supply or where it is economically prudent to do so.
None of our employees are unionized,
and we consider our relations with our employees to be good.
E. |
Share Ownership |
The following table sets forth
information regarding beneficial ownership of our common shares as of November 17, 2006, by our officers and directors individually and as a
group.
| Number of Shares Beneficially Owned |
Percentage of Shares Outstanding |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
Jim A.
Wright |
4,428,521 | 2.12 | % | |||||||
Aiping H.
Young |
14,340 | 0.00 | % | |||||||
Elizabeth
Williams |
6,852 | 0.00 | % | |||||||
Michael
Moore |
Nil | 0.00 | % | |||||||
Georg
Ludwig* |
29,090,000 | 13.92 | % | |||||||
Donald
Paterson |
125,260 | 0.06 | % | |||||||
Graham
Strachan |
10,000 | 0.00 | % | |||||||
Alan
Steigrod |
Nil | 0.00 | % | |||||||
Kevin Buchi |
50,000 | 0.02 | % | |||||||
All directors and executive officers as a group (seven persons) |
33,724,973 | 16.14 | % | |||||||
* |
Mr. Ludwig is affiliated with HighTech in his capacity as managing director. |
See item 6.B for a description of arrangements pursuant to which
employees may become involved in the capital of Lorus.
Item
7. |
Major Shareholders and Related Party Transactions |
A. |
Major Shareholders |
To the knowledge of our directors and
officers, as of the date hereof, no person or company beneficially owns, directly or indirectly, or exercises control or direction over, more than 5%
of the outstanding common shares, other than as described below.
On July 13, 2006, Lorus entered into a
share purchase agreement with High Tech Beteiligungen GmbH & Co. KG (“High Tech”) to issue 28.8 million common shares at $0.36 per share
for gross proceeds of $10.4 million. The transaction closed on August 30, 2006. As of November 17, 2006 based solely on 13-G and 13-D reports filed
with the Securities and Exchange Commission, High Tech holds approximately 14% of the issued and outstanding common shares of Lorus.
On July 24, 2006 Lorus entered into a
share purchase agreement with Technifund Inc. (“Technifund”) to issue, on a private placement basis, 5,000,000 common shares at $0.36 for
gross proceeds of $1,800,000. The transaction closed on August 31, 2006. As of November 17, 2006 based solely on 13-G and 13-D reports filed with the
Securities and Exchange Commission, Technifund holds approximately 7% of the issued and outstanding common shares of Lorus.
51
B. |
Related Party Transactions |
See Item 7.A.
In 2006, we did not enter into any
transactions with related parties. In order to effectively execute our business strategy, we expect to continue outsourcing various functions to the
expertise of third-parties such as contract manufacturing organizations, contract research organizations, and other research organizations. These
relationships are with non-related third-parties and occur at arm’s length and on normal commercial terms.
C. |
Interests of Experts and Counsel |
Not applicable.
Item
8. |
Financial Information |
A. |
Consolidated Financial Statements and Other Financial Information |
See Item 17.
B. |
Significant Changes |
None.
Item
9. |
The Offer and Listing |
A. |
Offer and Listing details |
Not applicable, except for Item 9A (4).
Price Range of Common Stock and Trading
Markets
Our common shares are currently listed
on the TSX under the symbol “LOR” and on the AMEX under the symbol “LRP”. The following table sets out the price ranges and trading
volumes of our common shares on the TSX and Amex for the periods indicated:
| American Stock Exchange/Amex (US$) |
Toronto Stock Exchange/TSX (CDN$) |
||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Five most recent full fiscal years: |
High |
Low |
High |
Low |
|||||||||||||||
Year ended
May 31, 2006 |
0.79 | 0.19 | 0.92 | 0.22 | |||||||||||||||
Year ended
May 31, 2005 |
0.70 | 0.45 | 0.94 | 0.57 | |||||||||||||||
Year ended
May 31, 2004 |
1.09 | 0.60 | 1.47 | 0.83 | |||||||||||||||
Year ended
May 31, 2003 |
1.40 | 0.18 | 2.04 | 0.31 | |||||||||||||||
Year ended
May 31, 2002 |
1.16 | 0.41 | 1.80 | 0.63 | |||||||||||||||
Year ended
May 31, 2006 |
0.79 | 0.19 | 0.92 | 0.22 | |||||||||||||||
Quarter ended
May 31, 2006 |
0.36 | 0.30 | 0.42 | 0.34 | |||||||||||||||
Quarter ended
February 28, 2006 |
0.42 | 0.19 | 0.49 | 0.22 | |||||||||||||||
Quarter ended
November 30, 2005 |
0.79 | 0.22 | 0.92 | 0.25 | |||||||||||||||
Quarter ended
August 31, 2005 |
0.68 | 0.55 | 0.84 | 0.60 | |||||||||||||||
Year ended
May 31, 2005 |
0.70 | 0.45 | 0.94 | 0.57 | |||||||||||||||
Quarter ended
May 31, 2005 |
0.68 | 0.55 | 0.94 | 0.58 | |||||||||||||||
Quarter ended
February 28, 2005 |
0.70 | 0.46 | 0.88 | 0.66 | |||||||||||||||
Quarter ended
November 30, 2004 |
0.69 | 0.55 | 0.86 | 0.57 | |||||||||||||||
Quarter ended
August 31, 2004 |
0.69 | 0.45 | 0.82 | 0.67 | |||||||||||||||
52
October
2006 |
0.27 | 0.20 | 0.30 | 0.23 | ||||||||||||||
September
2006 |
0.31 | 0.25 | 0.34 | 0.28 | ||||||||||||||
August
2006 |
0.34 | 0.26 | 0.39 | 0.30 | ||||||||||||||
July
2006 |
0.32 | 0.23 | 0.38 | 0.28 | ||||||||||||||
June
2006 |
0.33 | 0.28 | 0.37 | 0.31 | ||||||||||||||
May
2006 |
0.35 | 0.30 | 0.38 | 0.34 |
B. |
Plan of Distribution |
Not applicable.
C. |
Markets |
See Item 9.A.
D. |
Selling Shareholders |
Not applicable.
E. |
Dilution |
Not applicable.
F. |
Expense of the Issue |
Not applicable.
Item
10. |
Additional Information |
A. |
Share Capital |
Description of Securities
Our authorized share capital consists
of an unlimited number of common shares, without par value.
As of May 31, 2006, 175,262,548 common
shares were issued and outstanding. In addition, as of May 31, 2006 there were 13,470,000 common shares issuable upon the exercise of outstanding stock
options at a weighted average exercise price of $0.48 per share, 4,662,390 common shares reserved for future grant or issuance under our stock option
plan and 3,000,000 common shares issuable upon the exercise of outstanding warrants at a weighted-average exercise price of $1.00 per
share.
B. |
Articles of Incorporation and By-laws |
We are incorporated pursuant to the
laws of Canada. Our Articles of Incorporation and By-laws provide no restrictions as to the nature of our business operations. Under Canadian law, a
director must inform us, at a meeting of the Board of Directors, of any interest in a material contract or proposed material contract with us.
Directors may not vote in respect of any such contracts made with us or in any such contract in which a director is interested, and such directors
shall not be counted for purposes of determining a quorum. However, these provisions do not apply to (i) a contract relating primarily to their
remuneration as a director, officer, employee or agent, (ii) a contract for their indemnity or insurance as allowed under the Canada Business
Corporations Act, or (iii) a contract with an affiliate.
We are authorized to issue an unlimited
number of common shares. Our stockholders have no rights to share in our profits, are subject to no redemption or sinking fund provisions, have no
liability for further capital calls and are not subject to any discrimination due to number of shares owned. By not more than 50 days or less than
seven days in advance of a dividend, the Board of Directors may establish a record date for the determination of the persons entitled to such
dividend.
53
The rights of holders of our common
stock can be changed at any time in a stockholder meeting where the modifications are approved by 66 2/3% of the shares represented by proxy or in
person at a meeting at which a quorum exists.
All holders of our common stock are
entitled to vote at annual or special meetings of stockholders, provided that they were stockholders as of the record date. The record date for
stockholder meetings may precede the meeting date by no more than 50 days and not less than 21 days, provided that notice by way of advertisement is
given to stockholders at least seven days before such record date. Notice of the time and place of meetings of stockholders may not be less than 21 or
greater than 50 days prior to the date of the meeting. There are no:
• |
limitations on share ownership; |
• |
provisions of the Articles or by-laws that would have the effect of delaying, deferring or preventing a change of control of our company; |
• |
by-law provisions that govern the ownership threshold above which stockholder ownership must be disclosed; and |
• |
conditions imposed by the Articles or by-laws governing changes in capital, but Alberta law requires any changes to the terms of share capital be approved by 66b% of the shares represented by proxy or in person at a stockholders’ meeting convened for that purpose at which a quorum exists. |
Common Stock
Each holder of record of common stock
is entitled to one vote for each share held on all matters properly submitted to the stockholders for their vote, except matters which are required to
be voted on as a particular class or series of stock. Cumulative voting for directors is not permitted.
Holders of outstanding shares of common
stock are entitled to those dividends declared by the Board of Directors out of legally available funds. In the event of liquidation, dissolution or
winding up our affairs, holders of common stock are entitled to receive, pro rata, our net assets available after provision has been made for the
preferential rights of the holders of preferred stock. Holders of outstanding common stock have no pre-emptive, conversion or redemption rights. All of
the issued and outstanding shares of common stock are, and all unissued shares of common stock, when offered and sold will be, duly authorized, validly
issued, fully paid and non-assessable. To the extent that additional shares of common stock may be issued in the future, the relative interests of the
then existing stockholders may be diluted. There were 175,262,548 common shares issued and outstanding at May 31, 2006.
Convertible Debentures
On October 6, 2004, we entered into an
agreement to raise aggregate net proceeds of $13.9 million through the issuance of secured convertible debentures and warrants. The debentures are
secured by a first charge over all of the assets of the Company. We received $4.4 million on October 6, 2004 (representing a $5.0 million debenture
less an investor fee representing 4% of the $15.0 million to be received under the agreement), and $5.0 million on each of January 14 and April 15,
2005. All debentures issued under this agreement are due on October 6, 2009 and are subject to interest payable monthly at a rate of prime +1% until
such time as the Company’s share price reaches $1.75 for 60 consecutive trading days, at which time, interest will no longer be charged. Interest
is payable in common shares of Lorus until Lorus’ shares trade at a price of $1.00 or more after which interest will be payable in cash or common
shares at the option of the debenture holder. Common shares issued in payment of interest will be issued at a price equal to the weighted average
trading price of such shares for the ten trading days immediately preceding their issue in respect of each interest payment. For the year ended May 31,
2006, the Company has issued 2,153,000 common shares in settlement of $882 thousand in interest. For the year ended May 31, 2005 the Company issued
421,000 common shares in settlement of $300 thousand in interest.
The $15.0 million principal amount of
debentures is convertible at the holder’s option at any time into common shares of the Company with a conversion price per share of
$1.00.
54
The Company issued to the debt holder
3,000,000 warrants expiring October 6, 2009 to buy common shares of the Company at a price per share equal to $1.00.
Shares Eligible for Future Sale
Future sales of substantial amounts of
our common stock in the public market or even the perception that such sales may occur, could adversely affect the market price for our common stock
and could impair our future ability to raise capital through an offering of our equity securities.
At May 31, 2006 there were 10,300,000
options outstanding under the plan to purchase an equal number of shares of common stock. The outstanding options are exercisable at a weighted average
price per share of $0.70.
Indemnification of Executive Officers and
Directors
We have agreed to indemnify our
executive officers and directors for all costs, charges and expenses, including an amount paid to settle an action or satisfy a judgment, reasonably
incurred by them in respect of any civil, criminal or administrative action or proceeding to which they are made a party by reason of being or having
been a director or officer, if (a) they acted honestly and in good faith with a view to our best interests, and (b) in the case of a criminal or
administrative action or proceeding that is enforced by a monetary penalty, they had reasonable grounds for believing that their conduct was
lawful.
C. |
Material Contracts |
The Company has entered into material
contracts that are other than in the ordinary course of business during the previous two years, as follows:
Please refer to Item 4 — Business
Overview — Business Strategy, for details of the share purchase agreements entered into with each of High Tech and Technifund. Please refer to
Item 4 — Business Overview — Business Strategy — Secured Convertible Debentures, for details of the subscription agreement, debentures
and warrants entered into with TEMIC.
Other than the agreements described in
the preceding paragraph, we have not, during our financial year ending May 31, 2006, entered into any material contracts other than contracts in the
ordinary course of business. The Company is not a party to any other material contracts entered into since January 1, 2002 and still in
effect.
D. |
Exchange Controls |
There is no law or governmental decree
or regulation in Canada that restricts the export or import of capital, or affects the remittance of dividends, interest or other payments to
non-resident holders of our voting shares, other than withholding tax requirements.
There is no limitation imposed by
Canadian law or by our Articles or our other charter documents on the right of a non-resident to hold or vote voting shares, other than as provided by
the Investment Canada Act, the North American Free Trade Agreement Implementation Act (Canada) and the World Trade Organization Agreement
Implementation Act.
The Investment Canada Act requires
notification and, in certain cases, advance review and approval by the government of Canada of the acquisition by a non-Canadian of control of a
Canadian business, all as defined in the Investment Canada Act. Generally, the threshold for review will be higher in monetary terms for a member of
the World Trade Organization or North American Free Trade Agreement.
55
E. |
Taxation |
U.S. Federal Income Tax Consequences
The following is a summary of the
anticipated material U.S. federal income tax consequences to a U.S. Holder (as defined below) arising from and relating to the acquisition, ownership,
and disposition of common shares of the Company (“Common Shares”).
This summary is for general information
purposes only and does not purport to be a complete analysis or listing of all potential U.S. federal income tax consequences that may apply to a U.S.
Holder as a result of the acquisition, ownership, and disposition of Common Shares. In addition, this summary does not take into account the individual
facts and circumstances of any particular U.S. Holder that may affect the U.S. federal income tax consequences of the acquisition, ownership, and
disposition of Common Shares. Accordingly, this summary is not intended to be, and should not be construed as, legal or U.S. federal income tax advice
with respect to any U.S. Holder. Each U.S. Holder should consult its own financial advisor, legal counsel, or accountant regarding the U.S. federal,
U.S. state and local, and foreign tax consequences of the acquisition, ownership, and disposition of Common Shares.
Scope of this Disclosure
Authorities
This summary is based on the Internal
Revenue Code of 1986, as amended (the “Code”), Treasury Regulations (whether final, temporary, or proposed), published rulings of the
Internal Revenue Service (“IRS”), published administrative positions of the IRS, the Convention Between Canada and the United States of
America with Respect to Taxes on Income and on Capital, signed September 26, 1980, as amended (the “Canada-U.S. Tax Convention”), and U.S.
court decisions that are applicable and, in each case, as in effect and available, as of the date of this Annual Report. Any of the authorities on
which this summary is based could be changed in a material and adverse manner at any time, and any such change could be applied on a retroactive basis.
This summary does not discuss the potential effects, whether adverse or beneficial, of any proposed legislation that, if enacted, could be applied on a
retroactive basis.
U.S.
Holders
For purposes of this summary, a
“U.S. Holder” is a beneficial owner of Common Shares that, for U.S. federal income tax purposes, is (a) an individual who is a citizen or
resident of the U.S., (b) a corporation, or any other entity classified as a corporation for U.S. federal income tax purposes, that is created or
organized in or under the laws of the U.S. or any state in the U.S., including the District of Columbia, (c) an estate if the income of such estate is
subject to U.S. federal income tax regardless of the source of such income, or (d) a trust if (i) such trust has validly elected to be treated as a
U.S. person for U.S. federal income tax purposes or (ii) a U.S. court is able to exercise primary supervision over the administration of such trust and
one or more U.S. persons have the authority to control all substantial decisions of such trust.
Non-U.S.
Holders
For purposes of this summary, a
“non-U.S. Holder” is a beneficial owner of Common Shares other than a U.S. Holder. This summary does not address the U.S. federal income tax
consequences of the acquisition, ownership, and disposition of Common Shares to non-U.S. Holders. Accordingly, a non-U.S. Holder should consult its own
financial advisor, legal counsel, or accountant regarding the U.S. federal, U.S. state and local, and foreign tax consequences (including the potential
application of and operation of any tax treaties) of the acquisition, ownership, and disposition of Common Shares.
U.S. Holders Subject to
Special U.S. Federal Income Tax Rules Not Addressed
This summary does not address the U.S.
federal income tax consequences of the acquisition, ownership, and disposition of Common Shares to U.S. Holders that are subject to special provisions
under the Code, including
56
the following U.S. Holders: (a) U.S. Holders that are tax-exempt organizations, qualified retirement plans, individual retirement accounts, or other tax-deferred accounts; (b) U.S. Holders that are financial institutions, insurance companies, real estate investment trusts, or regulated investment companies; (c) U.S. Holders that are broker-dealers, dealers, or traders in securities or currencies that elect to apply a mark-to-market accounting method; (d) U.S. Holders that have a “functional currency” other than the U.S. dollar; (e) U.S. Holders that are liable for the alternative minimum tax under the Code; (f) U.S. Holders that own Common Shares as part of a straddle, hedging transaction, conversion transaction, constructive sale, or other arrangement involving more than one position; (g) U.S. Holders that acquired Common Shares in connection with the exercise of employee stock options or otherwise as compensation for services; (h) U.S. Holders that hold Common Shares other than as a capital asset within the meaning of Section 1221 of the Code; or (i) U.S. Holders that own, directly or indirectly, 10% or more, by voting power or value, of the outstanding shares of the Company. U.S. Holders that are subject to special provisions under the Code, including U.S. Holders described immediately above, should consult their own financial advisor, legal counsel or accountant regarding the U.S. federal, U.S. state and local, and foreign tax consequences of the acquisition, ownership, and disposition of Common Shares.
If an entity that is classified as
partnership (or “pass-through” entity) for U.S. federal income tax purposes holds Common Shares, the U.S. federal income tax consequences to
such partnership (or “pass-through” entity) and the partners of such partnership (or owners of such “pass-through” entity)
generally will depend on the activities of the partnership (or “pass-through” entity) and the status of such partners (or owners). Partners
of entities that are classified as partnerships (or owners of “pass-through” entities) for U.S. federal income tax purposes should consult
their own financial advisor, legal counsel or accountant regarding the U.S. federal income tax consequences of the acquisition, ownership, and
disposition of Common Shares.
Tax Consequences Other than
U.S. Federal Income Tax Consequences Not Addressed
This summary does not address the U.S.
state and local, U.S. federal estate and gift, or foreign tax consequences to U.S. Holders of the acquisition, ownership, and disposition of Common
Shares. Each U.S. Holder should consult its own financial advisor, legal counsel, or accountant regarding the U.S. state and local, U.S. federal estate
and gift, and foreign tax consequences of the acquisition, ownership, and disposition of Common Shares. (See ”Taxation—Canadian
Taxation” below).
U.S. Federal Income Tax Consequences of the Acquisition,
Ownership, and Disposition of Common Shares
Distributions on Common Shares
General
Taxation of Distributions
A U.S. Holder that receives a
distribution, including a constructive distribution, with respect to the Common Shares will be required to include the amount of such distribution in
gross income as a dividend (without reduction for any Canadian income tax withheld from such distribution) to the extent of the current or accumulated
“earnings and profits” of the Company. To the extent that a distribution exceeds the current and accumulated “earnings and profits”
of the Company, such distribution will be treated (a) first, as a tax-free return of capital to the extent of a U.S. Holder’s tax basis in the
Common Shares and, (b) thereafter, as gain from the sale or exchange of such Common Shares. (See more detailed discussion at “Disposition of
Common Shares” below).
Reduced Tax Rates for Certain
Dividends
For taxable years beginning after
December 31, 2002 and before January 1, 2011, a dividend paid by the Company generally will be taxed at the preferential tax rates applicable to
long-term capital gains if (a) the Company is a “qualified foreign corporation” (as defined below), (b) the U.S. Holder receiving such
dividend is an individual, estate, or trust, and (c) such dividend is paid on Common Shares that have been held by such U.S. Holder for at least 61
days during the 121-day period beginning 60 days before the “ex-dividend date” (i.e., the first date that a purchaser of such Common Shares
will not be entitled to receive such dividend).
The Company generally will be a
“qualified foreign corporation” under Section 1(h)(11) of the Code (a “QFC”) if (a) the Company is incorporated in a possession of
the U.S., (b) the Company is eligible for the benefits
57
of the Canada-U.S. Tax Convention, or (c) the Common Shares are readily tradable on an established securities market in the U.S. However, even if the Company satisfies one or more of such requirements, the Company will not be treated as a QFC if the Company is a “passive foreign investment company” (as defined below) for the taxable year during which the Company pays a dividend or for the preceding taxable year. In 2003, the U.S. Department of the Treasury (the “Treasury”) and the IRS announced that they intended to issue Treasury Regulations providing procedures for a foreign corporation to certify that it is a QFC. Although these Treasury Regulations were not issued in 2004, the Treasury and the IRS have confirmed their intention to issue these Treasury Regulations. It is expected that these Treasury Regulations will obligate persons required to file information returns to report a distribution with respect to a foreign security issued by a foreign corporation as a dividend from a QFC if the foreign corporation has, among other things, certified under penalties of perjury that the foreign corporation was not a “passive foreign investment company” for the taxable year during which the foreign corporation paid the dividend or for the preceding taxable year.
As discussed below, the Company does
not believe that it was a “passive foreign investment company” for the taxable year ended December 31, 2005, and does not expect that it will
be a “passive foreign investment company” for the taxable year ending December 31, 2006. (See more detailed discussion at “Additional
Rules that May Apply to U.S. Holders” below). However, there can be no assurance that the IRS will not challenge the determination made by the
Company concerning its “passive foreign investment company” status or that the Company will not be a “passive foreign investment
company” for the current or any future taxable year. Accordingly, although the Company expects that it may be a QFC, there can be no assurances
that the IRS will not challenge the determination made by the Company concerning its QFC status, that the Company will be a QFC for the current or any
future taxable year, or that the Company will be able to certify that it is a QFC in accordance with the certification procedures issued by the
Treasury and the IRS.
If the Company is not a QFC, a dividend
paid by the Company to a U.S. Holder, including a U.S. Holder that is an individual, estate, or trust, generally will be taxed at ordinary income tax
rates (and not at the preferential tax rates applicable to long-term capital gains). The dividend rules are complex, and each U.S. Holder should
consult its own financial advisor, legal counsel, or accountant regarding the dividend rules.
Distributions Paid in Foreign
Currency
The amount of a distribution paid to a
U.S. Holder in foreign currency generally will be equal to the U.S. dollar value of such distribution based on the exchange rate applicable on the date
of receipt. A U.S. Holder that does not convert foreign currency received as a distribution into U.S. dollars on the date of receipt generally will
have a tax basis in such foreign currency equal to the U.S. dollar value of such foreign currency on the date of receipt. Such a U.S. Holder generally
will recognize ordinary income or loss on the subsequent sale or other taxable disposition of such foreign currency (including an exchange for U.S.
dollars).
Dividends Received
Deduction
Dividends paid on the Common Shares
generally will not be eligible for the “dividends received deduction.” The availability of the dividends received deduction is subject to
complex limitations that are beyond the scope of this discussion, and a U.S. Holder that is a corporation should consult its own financial advisor,
legal counsel, or accountant regarding the dividends received deduction.
Disposition of Common
Shares
A U.S. Holder will recognize gain or
loss on the sale or other taxable disposition of Common Shares in an amount equal to the difference, if any, between (a) the amount of cash plus the
fair market value of any property received and (b) such U.S. Holder’s tax basis in the Common Shares sold or otherwise disposed of. Any such gain
or loss generally will be capital gain or loss, which will be long-term capital gain or loss if the Common Shares are held for more than one year. Gain
or loss recognized by a U.S. Holder on the sale or other taxable disposition of Common Shares generally will be treated as “U.S. source” for
purposes of applying the U.S. foreign tax credit rules. (See more detailed discussion at “Foreign Tax Credit” below).
58
Preferential tax rates apply to
long-term capital gains of a U.S. Holder that is an individual, estate, or trust. There are currently no preferential tax rates for long-term capital
gains of a U.S. Holder that is a corporation. Deductions for capital losses and net capital losses are subject to complex limitations under the Code.
For a U.S. Holder that is an individual, estate, or trust, capital losses may be used to offset capital gains and up to U.S.$3,000 of ordinary income.
An unused capital loss of a U.S. Holder that is an individual, estate, or trust generally may be carried forward to subsequent taxable years, until
such net capital loss is exhausted. For a U.S. Holder that is a corporation, capital losses may be used to offset capital gains, and an unused capital
loss generally may be carried back three years and carried forward five years from the year in which such net capital loss is
recognized.
Foreign Tax
Credit
A U.S. Holder who pays (whether
directly or through withholding) Canadian income tax with respect to dividends paid on the Common Shares generally will be entitled, at the election of
such U.S. Holder, to receive either a deduction or a credit for such Canadian income tax paid. Generally, a credit will reduce a U.S. Holder’s
U.S. federal income tax liability on a dollar-for-dollar basis, whereas a deduction will reduce a U.S. Holder’s income subject to U.S. federal
income tax. This election is made on a year-by-year basis and applies to all foreign taxes paid (whether directly or through withholding) by a U.S.
Holder during a year.
Complex limitations apply to the
foreign tax credit, including the general limitation that the credit cannot exceed the proportionate share of a U.S. Holder’s U.S. federal income
tax liability that such U.S. Holder’s “foreign source” taxable income bears to such U.S. Holder’s worldwide taxable income. In
applying this limitation, a U.S. Holder’s various items of income and deduction must be classified, under complex rules, as either “foreign
source” or “U.S. source.” In addition, this limitation is calculated separately with respect to specific categories of income (including
“passive income,” “high withholding tax interest,” “financial services income,” “general income,” and certain
other categories of income). Dividends paid by the Company generally will constitute “foreign source” income and generally will be
categorized as “passive income” or, in the case of certain U.S. Holders, “financial services income.” However, for taxable years
beginning after December 31, 2006, the foreign tax credit limitation categories are reduced to “passive category income” and “general
category income” (and the other categories of income, including “financial services income,” are eliminated). The foreign tax credit
rules are complex, and each U.S. Holder should consult its own financial advisor, legal counsel, or accountant regarding the foreign tax credit
rules.
Information Reporting; Backup
Withholding Tax
Payments made within the U.S., or by a
U.S. payor or U.S. middleman, of dividends on, and proceeds arising from certain sales or other taxable dispositions of, Common Shares generally will
be subject to information reporting and backup withholding tax, at the rate of 28%, if a U.S. Holder (a) fails to furnish such U.S. Holder’s
correct U.S. taxpayer identification number (generally on Form W-9), (b) furnishes an incorrect U.S. taxpayer identification number, (c) is notified by
the IRS that such U.S. Holder has previously failed to properly report items subject to backup withholding tax, or (d) fails to certify, under penalty
of perjury, that such U.S. Holder has furnished its correct U.S. taxpayer identification number and that the IRS has not notified such U.S. Holder that
it is subject to backup withholding tax. However, U.S. Holders that are corporations generally are excluded from these information reporting and backup
withholding tax rules. Any amounts withheld under the U.S. backup withholding tax rules will be allowed as a credit against a U.S. Holder’s U.S.
federal income tax liability, if any, or will be refunded, if such U.S. Holder furnishes required information to the IRS. Each U.S. Holder should
consult its own financial advisor, legal counsel, or accountant regarding the information reporting and backup withholding tax rules.
Additional Rules that May Apply to U.S.
Holders
If the Company is a “controlled
foreign corporation” or a “passive foreign investment company” (each as defined below), the preceding sections of this summary may not
describe the U.S. federal income tax consequences to U.S. Holders of the acquisition, ownership, and disposition of Common Shares.
59
Controlled Foreign
Corporation
The Company generally will be a
“controlled foreign corporation” under Section 957 of the Code (a “CFC”) if more than 50% of the total voting power or the total
value of the outstanding shares of the Company is owned, directly or indirectly, by citizens or residents of the U.S., domestic partnerships, domestic
corporations, domestic estates, or domestic trusts (each as defined in Section 7701(a)(30) of the Code), each of which own, directly or indirectly, 10%
or more of the total voting power of the outstanding shares of the Company (a “10% Shareholder”).
If the Company is a CFC, a 10%
Shareholder generally will be subject to current U.S. federal income tax with respect to (a) such 10% Shareholder’s pro rata share of the
“subpart F income” (as defined in Section 952 of the Code) of the Company and (b) such 10% Shareholder’s pro rata share of the earnings
of the Company invested in “United States property” (as defined in Section 956 of the Code). In addition, under Section 1248 of the Code, any
gain recognized on the sale or other taxable disposition of Common Shares by a U.S. Holder that was a 10% Shareholder at any time during the five-year
period ending with such sale or other taxable disposition generally will be treated as a dividend to the extent of the “earnings and profits”
of the Company that are attributable to such Common Shares. If the Company is both a CFC and a “passive foreign investment company” (as
defined below), the Company generally will be treated as a CFC (and not as a “passive foreign investment company”) with respect to any 10%
Shareholder.
The Company does not believe that it
has previously been, or currently is, a CFC. However, there can be no assurance that the Company will not be a CFC for the current or any future
taxable year.
Passive Foreign Investment
Company
The Company generally will be a
“passive foreign investment company” under Section 1297 of the Code (a ”PFIC”) if, for a taxable year, (a) 75% or more of the gross
income of the Company for such taxable year is passive income or (b) on average 50% or more of the assets held by the Company either produce passive
income or are held for the production of passive income, based on the fair market value of such assets (or on the adjusted tax basis of such assets, if
the Company is not publicly traded and either is a “controlled foreign corporation” or makes an election). “Passive income”
includes, for example, dividends, interest, certain rents and royalties, certain gains from the sale of stock and securities, and certain gains from
commodities transactions. However, for transactions entered into on or before December 31, 2004, gains arising from the sale of commodities generally
are excluded from passive income if (a) a foreign corporation holds the commodities directly (and not through an agent or independent contractor) as
inventory or similar property or as dealer property, (b) such foreign corporation incurs substantial expenses related to the production, processing,
transportation, handling, or storage of the commodities, and (c) gross receipts from sales of commodities that satisfy the requirements of clauses (a)
and (b) constitute at least 85% of the total gross receipts of such foreign corporation. For transactions entered into after December 31, 2004, gains
arising from the sale of commodities generally are excluded from passive income if substantially all of a foreign corporation’s commodities are
(a) stock in trade of such foreign corporation or other property of a kind which would properly be included in inventory of such foreign corporation,
or property held by such foreign corporation primarily for sale to customers in the ordinary course of business, (b) property used in the trade or
business of such foreign corporation that would be subject to the allowance for depreciation under Section 167 of the Code, or (c) supplies of a type
regularly used or consumed by such foreign corporation in the ordinary course of its trade or business.
For purposes of the PFIC income test
and assets test described above, if the Company owns, directly or indirectly, 25% or more of the total value of the outstanding shares of another
foreign corporation, the Company will be treated as if it (a) held a proportionate share of the assets of such other foreign corporation and (b)
received directly a proportionate share of the income of such other foreign corporation. In addition, for purposes of the PFIC income test and asset
test described above, “passive income” does not include any interest, dividends, rents, or royalties that are received or accrued by the
Company from a “related person” (as defined in Section 954(d)(3) of the Code), to the extent such items are properly allocable to the income
of such related person that is not passive income.
If the Company is a PFIC, the U.S.
federal income tax consequences to a U.S. Holder of the acquisition, ownership, and disposition of Common Shares will depend on whether such U.S.
Holder makes an election to treat
60
the Company as a “qualified electing fund” or “QEF” under Section 1295 of the Code (a “QEF Election”) or a mark-to-market election under Section 1296 of the Code (a “Mark-to-Market Election”). A U.S. Holder that does not make either a QEF Election or a Mark-to-Market Election will be referred to in this summary as a “Non-Electing U.S. Holder.”
Under Section 1291 of the Code, any
gain recognized on the sale or other taxable disposition of Common Shares, and any “excess distribution” (as defined in Section 1291(b) of
the Code) paid on the Common Shares, must be rateably allocated to each day in a Non-Electing U.S. Holder’s holding period for the Common Shares.
The amount of any such gain or excess distribution allocated to prior years of such Non-Electing U.S. Holder’s holding period for the Common
Shares generally will be subject to U.S. federal income tax at the highest tax applicable to ordinary income in each such prior year. A Non-Electing
U.S. Holder will be required to pay interest on the resulting tax liability for each such prior year, calculated as if such tax liability had been due
in each such prior year.
A U.S. Holder that makes a QEF Election
generally will not be subject to the rules of Section 1291 of the Code discussed above. However, a U.S. Holder that makes a QEF Election generally will
be subject to U.S. federal income tax on such U.S. Holder’s pro rata share of (a) the “net capital gain” of the Company, which will be
taxed as long-term capital gain to such U.S. Holder, and (b) and the “ordinary earnings” of the Company, which will be taxed as ordinary
income to such U.S. Holder. A U.S. Holder that makes a QEF Election will be subject to U.S. federal income tax on such amounts for each taxable year in
which the Company is a PFIC, regardless of whether such amounts are actually distributed to such U.S. Holder by the Company.
A U.S. Holder that makes a
Mark-to-Market Election generally will not be subject to the rules of Section 1291 of the Code discussed above. A U.S. Holder may make a Mark-to-Market
Election only if the Common Shares are “marketable stock” (as defined in Section 1296(e) of the Code). A U.S. Holder that makes a
Mark-to-Market Election will include in gross income, for each taxable year in which the Company is a PFIC, an amount equal to the excess, if any, of
(a) the fair market value of the Common Shares as of the close of such taxable year over (b) such U.S. Holder’s tax basis in such Common Shares. A
U.S. Holder that makes a Mark-to-Market Election will, subject to certain limitations, be allowed a deduction in an amount equal to the excess, if any,
of (a) such U.S. Holder’s adjusted tax basis in the Common Shares over (b) the fair market value of such Common Shares as of the close of such
taxable year.
The Company does not believe that it
was a PFIC for the taxable year ended December 31, 2005, and does not expect that it will be a PFIC for the taxable year ending December 31, 2006.
However, the determination of whether the Company was, or will be, a PFIC for a taxable year depends, in part, on the application of complex U.S.
federal income tax rules, which are subject to various interpretations. In addition, whether the Company will be a PFIC for the taxable year ending
December 31, 2006 and each subsequent taxable year depends on the assets and income of the Company over the course of each such taxable year and, as a
result, cannot be predicted with certainty as of the date of this Annual Report. Accordingly, there can be no assurance that the IRS will not challenge
the determination made by the Company concerning its PFIC status or that the Company will not be a PFIC for the current or any future taxable
year.
The PFIC rules are complex, and each
U.S. Holder should consult its own financial advisor, legal counsel, or accountant regarding the PFIC rules and how the PFIC rules may affect the U.S.
federal income tax consequences of the acquisition, ownership, and disposition of Common Shares.
Canadian Taxation
The following summary fairly describes,
as of the date hereof, the principal Canadian federal income tax considerations under the Income Tax Act (Canada) (the “ITA”)
generally applicable to an owner of Common Shares who is not and has not been or deemed to be resident in Canada for purposes of the ITA at any time
while such Holder holds the Common Shares, is a resident of the U.S. for purposes of the Canada-U.S. Tax Convention, and who, for purposes of the ITA,
at all relevant times: holds the Common Shares as capital property; does not have a “permanent establishment” or “fixed base” in
Canada, as defined in the Canada-U.S. Tax Convention; does not use or hold (and is not deemed to use or hold) the Common Shares in carrying on a
business in Canada for purposes of the ITA; and deals at arm’s length and is not affiliated with the Company within the meaning of the ITA (a
“Holder”). The Common Shares will generally constitute capital property to a Holder unless such Holder holds such
61
Common Shares in the course of carrying on a business of trading or dealing in securities or has acquired such Common Shares in a transaction or transactions considered to be an adventure in the nature of trade.
This summary is not applicable to a
Holder an interest in which is a “tax shelter investment” as defined in the ITA, to a Holder who is a “financial institution” for
purposes of the “mark-to-market” rules contained in the ITA, or to a Holder who is a “specified financial institution” for the
purposes of the ITA. Such Holders should consult their own tax advisors.
This summary is based on the current
provisions of the ITA, the regulations thereunder (the “Regulations”), the Canada-U.S. Tax Convention, all specific proposed amendments to
the ITA or the Regulations publicly announced by or on behalf of the Canadian Minister of Finance prior to the date hereof, (the “Specific
Proposals”) and the Company’s understanding of the current published administrative and assessing practices of the Canada Revenue Agency (the
“CRA”). This summary assumes the Specific Proposals will be enacted as proposed but no assurance can be given that this will be the case and
this summary does not otherwise take into account or anticipate any changes in administrative practice or in law, whether by way of judicial,
governmental or legislative decision or action, nor does it take into account any income tax laws or considerations of any province or territory of
Canada or any jurisdiction other than Canada, which may differ from the Canadian federal income tax consequences described in this
document.
This summary is of a general nature
only, is not exhaustive of all possible tax considerations applicable to an investor, and is not intended to be relied on as legal or tax advice or
representations to any particular investor. Consequently, investors are urged to seek independent tax advice in respect of their particular
circumstances and the consequences to them of the acquisition, ownership or disposition of Common Shares having regard to their particular
circumstances.
Dividends
Under the Canada-U.S. Tax Convention,
dividends paid or credited, or deemed to be paid or credited, on the Common Shares to a Holder generally will be subject to Canadian withholding tax at
the rate of 15% of the gross amount of those dividends. If a Holder is a company within the meaning of the Canada-U.S. Tax Convention and owns 10% or
more of the Company’s voting stock, the rate is reduced from 15% to 5%.
Under the Canada-U.S. Tax Convention,
dividends paid to religious, scientific, literary, educational or charitable organizations or certain pension, retirement or employee benefit
organizations that have complied with administrative procedures specified by the CRA are exempt from the aforementioned Canadian withholding tax so
long as such organization is resident in and exempt from tax in the U.S. Such exemption does not apply to the extent the dividends are received in
connection with a trade or business carried on by such Holder or where the Company is related to such Holder.
Disposition of Common
Shares
A Holder will only be subject to
taxation in Canada under the ITA on capital gains realized by the Holder on a disposition or deemed disposition of the Common Shares if such shares
constitute “taxable Canadian property” within the meaning of the ITA at the time of the disposition or deemed disposition and the Holder is
not afforded relief under the Canada-U.S. Tax Convention. In general, the Common Shares will not be “taxable Canadian property” to a Holder
if, at the time of their disposition, they are listed on a stock exchange that is prescribed in the Regulations (which includes the American Stock
Exchange), unless:
• |
at any time within the 60-month period immediately preceding the disposition or deemed disposition, the Holder, persons not dealing at arm’s length with the Holder, or the Holder together with such non-arm’s length persons, owned 25% or more of the issued shares of any class or series of the Company’s capital stock; |
• |
the Holder was formerly resident in Canada and, upon ceasing to be a Canadian resident, elected under the ITA to have the Common Shares deemed to be “taxable Canadian property”; or |
• |
the Holder’s Common Shares were acquired in a tax deferred exchange in consideration for property that was itself “taxable Canadian property.” |
62
If a Holder’s Common Shares are
“taxable Canadian property,” such Holder will recognize a capital gain (or a capital loss) for the taxation year during which the Holder
disposes, or is deemed to have disposed of, the Common Shares. Such capital gain (or capital loss) will be equal to the amount by which the proceeds of
disposition exceed (or are less than) the Holder’s adjusted cost base of such Common Shares and any reasonable costs of making the disposition.
One-half of any such capital gain (a “taxable capital gain”) must be included in income in computing the Holder’s income and one half of
any such capital loss (an “allowable capital loss”) is generally deductible by the Holder from taxable capital gains arising in the year of
disposition. To the extent a Holder has insufficient taxable capital gains in the current taxation year against which to apply an allowable capital
loss, the deficiency will constitute a net capital loss for the current taxation year and may generally be carried back to any of the three preceding
taxation years or carried forward to any future taxation year, to the extent and under the circumstances described in the ITA.
F. |
Dividends and Paying Agents |
Not applicable.
G. |
Statement by Experts |
Not applicable.
H. |
Documents on Display |
We are subject to the information and
reporting requirements of the Securities Exchange Act of 1934, as amended, and file periodic reports and other information with the SEC. However, as a
foreign private issuer, we are exempt from the rules and regulations under the Exchange Act prescribing the furnishing and content of proxy statements,
and our officers, directors and principal stockholders are exempt from the reporting and short-swing profit recovery provisions contained in Section 16
of the Exchange Act. Our reports and other information filed with the SEC may be inspected at the public reference facilities maintained by the SEC at
100 F Street, N.E., Washington, D.C. 20549. Copies of these materials may be obtained at prescribed rates from the SEC at that address. Our reports and
other information can also be inspected at no charge on the SEC’s Web site at www.sec.gov.
We are also subject to the information
and reporting requirements of the Securities Act (Ontario) and the Canada Business Corporations Act. Such reports and information can be inspected at
no charge on the website www.sedar.com.
If you are a stockholder, you may
request a copy of these filings at no cost by contacting us at:
2 Meridian Road
Toronto, Ontario, M9W 4Z7
Canada
Phone (416) 798-1200
Fax (416) 798-2200
Toronto, Ontario, M9W 4Z7
Canada
Phone (416) 798-1200
Fax (416) 798-2200
I. |
Subsidiary Information |
Lorus’ subsidiaries are GeneSense
Technologies Inc. (“GeneSense”), a corporation incorporated under the laws of Canada, of which Lorus owns 100% of the issued and outstanding
share capital, and NuChem Pharmaceuticals Inc. (“NuChem”), a corporation incorporated under the laws of Ontario, of which Lorus owns 80% of
the issued and outstanding voting share capital and 100% of the issued and outstanding non-voting preference share capital.
Item
11. |
Qualitative and Quantitative Disclosures about Market Risk |
Refer to notes 13 and 15 of the consolidated financial statements
in Item 17.
63
Risk Factors
See item 3.D.
Item
12. |
Description of Securities Other Than Equity Securities |
Not applicable.
PART II
Item
13. |
Defaults, Dividends, Arrearages and Delinquencies |
Not applicable.
Item
14. |
Material Modifications to the Rights of Security Holders and Use of Proceeds |
Not applicable.
Item
15. |
Controls and Procedures |
(a) |
Evaluation of disclosure controls and procedures |
As of the end of Lorus’ fiscal
year ended May 31, 2006, an evaluation of the effectiveness of Lorus’ “disclosure controls and procedures” (as such term is defined in
Rules 13a-15(e) and 15d-15(e) of the Exchange Act), was carried out by Lorus’ management with the participation of the principal executive officer
and principal financial officer. Based upon on that evaluation, Lorus’ principal executive officer and principal financial have concluded that as
of the end of that fiscal year, Lorus’ disclosure controls and procedures are effective to ensure that information required to be disclosed by
Lorus in reports that it files or submits under the Exchange Act is (i) recorded, processed, summarized and reported within the time periods specified
in SEC rules and forms and (ii) accumulated and communicated to Lorus’ management, including its principal executive officer and principal
financial officer, to allow timely decisions regarding required disclosure.
It should be noted that while
Lorus’ principal executive officer and principal financial officer believe that Lorus’ disclosure controls and procedures provide a
reasonable level of assurance that they are effective, they do not expect that Lorus’ disclosure controls and procedures or internal control over
financial reporting will prevent all errors and fraud. A control system, no matter how well conceived or operated, can provide only reasonable, not
absolute, assurance that the objectives of the control system are met.
(b) |
Management’s annual report on internal control over financial reporting |
Not applicable.
(c) |
Attestation report of the independent registered public accounting firm |
Not applicable.
(d) |
Changes in internal controls |
There has been no change in Lorus’
internal control over financial reporting during the period covered by this report that has materially affected, or is reasonably likely to materially
affect, Lorus’ internal control over financial reporting.
64
Item
16. |
[Reserved] |
Item
16A. |
Audit Committee Financial Expert |
Our Board of Directors has determined
that J. Kevin Buchi, a director of the Company and the Chairman of the Audit Committee, possesses the attributes required of an “audit committee
financial expert,” and is “independent,” under applicable AMEX rules.
Item
16B. |
Code of Ethics |
We have adopted a Code of Ethics, which
applies to all of our officers, directors, employees and consultants. The Code of Ethics is publicly available on our website at www.lorusthera.com. A
copy of the Code of Ethics is also available upon written request from our Director of Finance at our offices located at 2 Meridian Road, Toronto,
Ontario M9W 4Z7. There were no amendments to, or waivers granted under, the Code of Ethics during our fiscal year ended May 31, 2006.
Item
16C. |
Principal Accountant Fees and Services |
KPMG LLP has served as our principal
independent auditors since October 1994. The total fees billed for professional services by KPMG LLP (our external auditors) for the years ended May
31, 2006 and 2005 are as follows:
| |
2006 |
|
2005 |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|
Audit
Fees |
$ | 198,500 | $ | 167,326 | ||||||
Audit-Related
Fees |
— | — | ||||||||
Tax
Fees |
$ | 13,100 | $ | 24,400 | ||||||
All Other
Fees |
— | — | ||||||||
Total |
$ | 211,600 | $ | 191,726 | ||||||
Audit fees consist of the fees paid
with respect to the audit of our consolidated annual financial statements, quarterly reviews and accounting assistance. Tax fees relate to assistance
provided with respect to proposed financing transactions and review of tax returns.
Pre-Approval Policies and Procedures
The audit committee of our board of
directors has, pursuant to the audit committee charter, adopted specific responsibilities and duties regarding the provision of services by our
external auditors, currently KPMG LLP. Our charter requires audit committee pre-approval of all permitted audit and audit-related services. Any audit
and non-audit services must also be submitted to the audit committee for review and approval. Under the charter, all permitted services to be provided
by KPMG LLP must be pre-approved by the audit committee.
Subject to the charter, the audit
committee may establish fee thresholds for a group of pre-approved services. The audit committee then recommends to the board of directors approval of
the fees and other significant compensation to be paid to the independent auditors.
No services were provided by KPMG LLP
under a de minimus exemption for our fiscal year ended May 31, 2006.
Item
16D. |
Exemptions from the Listing Standards for Audit Committees |
Not applicable.
65
Item
16E. |
Purchases of Equity Securities by the Issuer and Affiliated Purchasers |
Not applicable.
PART III
Item
17. |
Financial Statements |
The Consolidated Financial Statements of Lorus Therapeutics Inc.
are attached as follows:
| Page |
||||||
|---|---|---|---|---|---|---|
Managements
Responsibility for Financial Reporting |
F-1 |
|||||
Report of
Independent Registered Public Accounting Firm |
F-2 |
|||||
Consolidated
Balance Sheets as of May 31, 2006 and 2005 |
F-3 |
|||||
Consolidated
Statements of Loss and Deficit for the years ended May 31, 2006, 2005 and 2004 |
F-4 |
|||||
Consolidated
Statements of Cash Flows for the years ended May 31, 2006, 2005 and 2004 |
F-5 |
|||||
Notes to
Consolidated Financial Statements |
F-6 |
|||||
Item
18. |
Financial Statements |
We have responded to Item 17 in lieu of responding to this
Item.
Item
19. |
Exhibits |
| Number | Exhibit |
| 1.1 * | Articles of Amalgamation |
| 1.2 * | By-laws of the Registrant |
| 1.3 ** | Articles of Amendment, dated August 25, 1992, regarding name change of Company to Imutec Corporation |
| 1.4 *** | Articles of Amendment, dated November 27, 1996, regarding name change of Company to Imutec Pharma Inc. |
| 1.5 **** | Articles of Amendment dated November 19, 1998, regarding name change of Company to Lorus Therapeutics Inc. |
| 1.6 | Certificate of Continuance dated October 1, 2005 |
| 2.1 | Share Purchase Agreement dated as of July 13, 2006 between Lorus and High Tech Beteiligungen GmbH & Co. KG (“High Tech”) |
| 2.2 | Registration Rights Agreement dated as of August 30, 2006 between Lorus and High Tech |
| 2.3 | Share Purchase Agreement dated as of July 24, 2006 between Lorus and Technifund Inc. |
| 2.4 ***** | Subscription Agreement entered into with The Erin Mills Investment Corporation dated October 6, 2004 |
| 2.5 | Convertible Secured Debentures issued to The Erin Mills Investment Corporation on April 15, 2005, January 14, 2005 and October 6, 2004 |
| 4.1 | Stock Option Plan |
| 4.2 | Form of Officer and Director Indemnity Agreement |
| 4.3 * | Amalgamation Agreement dated August 23, 1991, among the Company, Mint Gold Resources Ltd., Harry J. Hodge and Wayne Beach. |
| 8.1 | List of Subsidiaries |
| 11.1 | Code of Business Conduct and Ethics |
| 12.1 | Certification of Chief Executive Officer pursuant to Section 302 of the Sarbanes-Oxley Act |
| 12.2 | Certification of Chief Financial Officer pursuant to Section 302 of the Sarbanes-Oxley Act |
| 13.1 | Certification of Chief Executive Officer pursuant to Section 906 of the Sarbanes-Oxley Act |
| 13.2 | Certification of Chief Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act |
| * | Incorporated by reference to File 0-19763, Registration Statement on Form 20-FR, dated March 4, 1992. |
| ** | Incorporated by reference to File 0-19763, Annual Report on Form 20-F, dated September 28, 1992. |
| *** | Incorporated by reference to File 0-19763, Annual Report on Form 20-F, dated November 18, 1998. |
| **** | Incorporated by reference to File 0-1963, Annual Report on Form 20-F dated November 30, 1999. |
| ***** | Incorporated by reference to File 1-32001, Form 6-K dated February 10, 2005. |
66
13.2 |
Certification of Chief Financial Officer pursuant to Section 906 of the Sarbanes-Oxley Act |
|||||
Signatures
The registrant hereby certifies that it meets all of the
requirements for filing on Form 20-F and that it has duly caused and authorized the undersigned to sign this annual report on its
behalf.
LORUS THERAPEUTICS
INC.
By: |
/s/ Aiping H. Young Name: Aiping H. Young Title: President and Chief Executive Officer Date: November 21, 2006 |
By: |
/s/ Elizabeth Williams Name: Elizabeth Williams Title: Director of Finance and Acting Chief Financial Officer Date: November 21, 2006 |
67
MANAGEMENT’S RESPONSIBILITY FOR FINANCIAL
REPORTING
The accompanying consolidated financial statements of Lorus
Therapeutics Inc. and other financial information contained in this annual report are the responsibility of Management and have been approved by the
Board of Directors of the Company.
The consolidated financial statements have been prepared in
conformity with Canadian generally accepted accounting principles, using Management’s best estimates and judgments where appropriate. In the
opinion of Management, these consolidated financial statements reflect fairly the financial position and the results of operations and cash flows of
the Company within reasonable limits of materiality. The financial information contained elsewhere in this annual report has been reviewed to ensure
consistency with that in the consolidated financial statements. The integrity and objectivity of data in the financial statements and elsewhere in this
annual report are the responsibility of Management.
In discharging its responsibility for the integrity and fairness
of the financial statements, management maintains a system of internal controls designed to provide reasonable assurance, at appropriate cost, that
transactions are authorized, assets are safeguarded and proper records are maintained. Management believes that the internal controls provide
reasonable assurance that financial records are reliable and form a proper basis for the preparation of the consolidated financial statements, and that
assets are properly accounted for and safeguarded. The internal control process includes management’s communication to employees of policies that
govern ethical business conduct.
The Board of Directors, through an Audit Committee, oversees
management’s responsibilities for financial reporting. This committee, which consists of three independent directors, reviews the audited
consolidated financial statements and recommends the financial statements to the Board for approval. Other key responsibilities of the Audit Committee
include reviewing the adequacy of the Company’s existing internal controls, audit process and financial reporting with management and the external
auditors.
The consolidated financial statements have been audited by KPMG
LLP, Chartered Accountants, who are independent auditors appointed by the shareholders of the Company upon the recommendation of the Audit Committee.
Their report follows. The independent auditors have free and full access to the Audit Committee.
/s/ Aiping H.
Young |
/s/ Elizabeth
Williams |
|||||||||
Aiping H.
Young President and Chief Executive Officer |
Elizabeth
Williams Director of Finance (Acting Chief Financial Officer) |
|||||||||
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING
FIRM
To the Board of Directors of Lorus Therapeutics
Inc.
We have audited the consolidated balance sheets of Lorus
Therapeutics Inc. (the “Company”) as at May 31, 2006 and 2005 and the consolidated statements of loss and deficit and cash flows for each of
the years in the three-year period ended May 31, 2006. These financial statements are the responsibility of the Company’s management. Our
responsibility is to express an opinion on these consolidated financial statements based on our audits.
We conducted our audits in accordance with the standards of the
Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform an audit to obtain reasonable assurance
whether the financial statements are free of material misstatement. An audit includes examining, on a test basis, evidence supporting the amounts and
disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by management,
as well as evaluating the overall financial statement presentation. We believe that our audits provide a reasonable basis for our audit
opinion.
In our opinion, these consolidated financial statements present
fairly, in all material respects, the financial position of the Company as at May 31, 2006 and 2005 and the results of its operations and its cash
flows for each of the years in the three-year period ended May 31, 2006 in accordance with Canadian generally accepted accounting
principles.
The accompanying consolidated financial statements have been
prepared assuming that the Company will continue as a going concern. As discussed in note 1 to the financial statements, the Company has suffered
recurring losses from operations and has a net shareholders’ deficiency that raise substantial doubt about its ability to continue as a going
concern. Management’s plan in regard to these matters is also described in note 1. The financial statements do not include any adjustments that
might result from the outcome of this uncertainty.
As discussed in note 3 to the financial statements, the Company
adopted new accounting policies related to the consolidation of variable interest entities, financial instruments — disclosure and presentation as
a liability of certain obligations that may be settled at the Company’s option in cash or the equivalent value by a variable number of the
Company’s own equity instruments, accounting for convertible debt instruments and accounting for non-monetary transactions.
Canadian generally accepted accounting principles vary in certain
significant respects from accounting principles generally accepted in the United States of America. Information relating to the nature and effect of
such differences is presented in note 17 to the consolidated financial statements.
We did not audit the consolidated financial statements of loss
and deficit and cash flows for the period from inception on September 6, 1986 to May 31, 2006 in accordance with the standards of the Public Company
Accounting Oversight Board (United States).
/s/ KPMG LLP
Chartered Accountants
Toronto, Canada
Toronto, Canada
November 17, 2006
F-2
Lorus Therapeutics Inc.
Consolidated Balance Sheets
| (amounts in Canadian 000’s) |
As at May 31, 2006 |
As at May 31, 2005 |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
ASSETS |
||||||||||
Current |
||||||||||
Cash and cash
equivalents |
$ | 2,692 | $ | 2,776 | ||||||
Short-term
investments (note 5) |
5,627 | 18,683 | ||||||||
Prepaid expenses and other assets |
515 | 1,126 | ||||||||
| 8,834 | 22,585 | |||||||||
Long-term |
||||||||||
Fixed assets
(note 6) |
885 | 1,581 | ||||||||
Deferred
financing charges (note 13) |
481 | 568 | ||||||||
Goodwill |
606 | 606 | ||||||||
Acquired patents and licenses (note 7) |
655 | 2,226 | ||||||||
| 2,627 | 4,981 | |||||||||
| $ | 11,461 | $ | 27,566 | |||||||
LIABILITIES |
||||||||||
Current |
||||||||||
Accounts
payable |
$ | 555 | $ | 1,069 | ||||||
Accrued liabilities |
2,460 | 3,019 | ||||||||
| 3,015 | 4,088 | |||||||||
Long-term |
||||||||||
Secured
convertible debentures (note 13) |
11,002 | 10,212 | ||||||||
SHAREHOLDERS’ EQUITY (DEFICIENCY) |
||||||||||
Share capital
(note 8) |
||||||||||
Common
shares |
145,001 | 144,119 | ||||||||
Equity
portion of secured convertible debentures (note 13) |
3,814 | 3,814 | ||||||||
Stock options
(note 8 (c)) |
4,525 | 4,252 | ||||||||
Contributed
surplus (note 8 (b)) |
7,665 | 6,733 | ||||||||
Warrants |
991 | 991 | ||||||||
Deficit accumulated during development stage |
(164,552 | ) | (146,643 | ) | ||||||
| (2,556 | ) | 13,266 | ||||||||
| $ | 11,461 | $ | 27,566 | |||||||
See accompanying notes to audited consolidated
financial statements
Basis of Presentation (note 1)
Commitments and Guarantees (note 14)
Canada and United States Accounting Policy Differences (note 17)
Subsequent Events (note 19)
Basis of Presentation (note 1)
Commitments and Guarantees (note 14)
Canada and United States Accounting Policy Differences (note 17)
Subsequent Events (note 19)
F-3
Lorus Therapeutics Inc.
Consolidated Statements of Loss and
Deficit
| (amounts in Canadian 000’s except for per common
share data) |
Years Ended May 31 |
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2006 |
|
2005 |
|
2004 |
|
Period from inception Sept. 5, 1986 to May 31, 2006 |
|||||||||||
REVENUE |
$ | 26 | $ | 6 | $ | 608 | $ | 706 | ||||||||||
EXPENSES |
||||||||||||||||||
Cost of
sales |
3 | 1 | 28 | 87 | ||||||||||||||
Research and
development (note 11) |
10,237 | 14,394 | 26,785 | 110,475 | ||||||||||||||
General and
administrative |
4,334 | 5,348 | 4,915 | 47,475 | ||||||||||||||
Stock-based
compensation (note 9) |
1,205 | 1,475 | — | 6,750 | ||||||||||||||
Depreciation and amortization (note 6) |
771 | 564 | 420 | 8,823 | ||||||||||||||
Operating
expenses |
16,550 | 21,782 | 32,148 | 173,610 | ||||||||||||||
Interest
expense (note 13) |
882 | 300 | — | 1,182 | ||||||||||||||
Accretion in
carrying value of secured convertible debentures (note 13) |
790 | 426 | — | 1,216 | ||||||||||||||
Amortization
of deferred financing charges |
87 | 84 | — | 171 | ||||||||||||||
Interest income |
(374 | ) | (524 | ) | (1,239 | ) | (10,921 | ) | ||||||||||
Loss for the period |
17,909 | 22,062 | 30,301 | 164,552 | ||||||||||||||
Deficit,
beginning of period |
146,643 | 121,804 | 91,503 | — | ||||||||||||||
Impact of change in accounting for stock options |
— | 2,777 | — | — | ||||||||||||||
Deficit, beginning of period (as restated) |
146,643 | 124,581 | 91,503 | — | ||||||||||||||
Deficit, end of period |
$ | 164,552 | $ | 146,643 | $ | 121,804 | $ | 164,552 | ||||||||||
Basic and diluted loss per common share |
$ | 0.10 | $ | 0.13 | $ | 0.18 | ||||||||||||
Weighted average number of common shares outstanding used in the calculation of basic and diluted loss per
share |
173,523 | 172,112 | 171,628 | |||||||||||||||
F-4
Lorus Therapeutics Inc.
Consolidated Statements of Cash Flows
| (amounts in Canadian 000’s) |
Years Ended May 31 |
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2006 |
|
2005 |
|
2004 |
|
Period from inception Sept. 5, 1986 to May 31, 2006 |
|||||||||||
OPERATING
ACTIVITIES |
||||||||||||||||||
Loss for the
period |
$ | (17,909 | ) | $ | (22,062 | ) | $ | (30,301 | ) | $ | (164,552 | ) | ||||||
Add items not
requiring a current outlay of cash: |
||||||||||||||||||
Stock-based
compensation (note 9) |
1,205 | 1,475 | — | 6,750 | ||||||||||||||
Interest
expense (note 13) |
882 | 300 | — | 1,182 | ||||||||||||||
Accretion in
carrying value of secured convertible debentures (note 13) |
790 | 426 | — | 1,216 | ||||||||||||||
Amortization
of deferred financing charges (note 13) |
87 | 84 | — | 171 | ||||||||||||||
Depreciation
and amortization (note 6) |
2,342 | 2,260 | 2,123 | 20,729 | ||||||||||||||
Other |
— | (38 | ) | 245 | 707 | |||||||||||||
Net change in non-cash working capital balances related to operations (note 12) |
(462 | ) | (1,166 | ) | (129 | ) | 1,592 | |||||||||||
Cash used in operating activities |
(13,065 | ) | (18,721 | ) | (28,062 | ) | (132,205 | ) | ||||||||||
INVESTING
ACTIVITIES |
||||||||||||||||||
Maturity
(purchase) of short-term investments, net |
13,056 | 6,974 | (1,438 | ) | (5,627 | ) | ||||||||||||
Business
acquisition, net of cash received |
— | — | — | (539 | ) | |||||||||||||
Acquired
patents and licenses |
— | — | — | (715 | ) | |||||||||||||
Additions to
fixed assets |
(75 | ) | (599 | ) | (383 | ) | (6,049 | ) | ||||||||||
Cash proceeds on sale of fixed assets |
— | — | — | 348 | ||||||||||||||
Cash provided by (used in) investing activities |
12,981 | 6,375 | (1,821 | ) | (12,582 | ) | ||||||||||||
FINANCING
ACTIVITIES |
||||||||||||||||||
Issuance of
debentures, net |
— | 12,948 | — | 12,948 | ||||||||||||||
Issuance of
warrants, net |
— | 991 | 4,537 | 37,405 | ||||||||||||||
Issuance of
common shares |
— | 112 | 25,512 | 97,371 | ||||||||||||||
Additions to deferred financing charges |
— | — | — | (245 | ) | |||||||||||||
Cash provided by financing activities |
— | 14,051 | 30,049 | 147,479 | ||||||||||||||
(Decrease)
increase in cash and cash equivalents during the period |
(84 | ) | 1,705 | 166 | 2,692 | |||||||||||||
Cash and cash equivalents, beginning of period |
2,776 | 1,071 | 905 | — | ||||||||||||||
Cash and cash equivalents, end of period |
$ | 2,692 | $ | 2,776 | $ | 1,071 | $ | 2,692 | ||||||||||
See accompanying notes to audited
consolidated financial statements
F-5
1. |
Basis of presentation |
Lorus Therapeutics Inc.
(“Lorus” or “the Company”) is a biopharmaceutical company specializing in the research and development of pharmaceutical products
and technologies for the management of cancer. With products in various stages of evaluation, from pre-clinical through to Phase II trials, Lorus
develops therapeutics that seek to manage cancer with efficacious low-toxicity compounds that improve patients’ quality of life.
The Company has not earned substantial
revenues from its drug candidates and is therefore considered to be in the development stage. The continuation of the Company’s research and
development activities is dependent upon the Company’s ability to successfully finance its cash requirements through a combination of equity
financing and payments from strategic partners. The Company has no current sources of payments from strategic partners. In addition, the Company will
need to repay or refinance the secured convertible debentures on their maturity should the holder not chose to convert the debentures into common
shares. There can be no assurance that additional funding will be available at all or on acceptable terms to permit further clinical development of the
Company’s products or to repay the convertible debentures on maturity. If the Company is not able to raise additional funds, it may not be able to
continue as a going concern and realize its assets and pay its liabilities as they fall due. The financial statements do not reflect adjustments that
would be necessary if the going concern assumption were not appropriate. If the going concern basis were not appropriate for these financial
statements, then adjustments would be necessary in the carrying value of the assets and liabilities, the reported revenues and expenses and the balance
sheet classifications used.
However, Management believes that the
Company’s current level of cash and short-term investments and the additional funds available upon the successful closing of the subscription
agreements described in note 19 will be sufficient to execute the Company’s current planned expenditures for the next twelve
months.
2. |
Significant accounting policies |
Principles of
consolidation
The consolidated financial statements
include the accounts of Lorus, its 80% owned subsidiary, NuChem Pharmaceuticals Inc. (“NuChem”), and its wholly owned subsidiary, GeneSense
Technologies Inc. (“GeneSense”) which are all located in Canada. The results of operations for acquisitions are included in these
consolidated financial statements from the date of acquisition. All significant intercompany balances and transactions have been eliminated on
consolidation.
The consolidated financial statements
have been prepared by management in accordance with accounting principles generally accepted in Canada and comply, in all material respects, with
accounting principles generally accepted in the United States, except as disclosed in note 17, “Canada and United States Accounting Policy
Differences.”
Revenue
Recognition
Revenue includes product sales revenue,
license revenue and royalty revenue.
The Company recognizes revenue from
product sales when persuasive evidence of an arrangement exists, delivery has occurred, the Company’s price to the customer is fixed or
determinable, and collectibility is reasonably assured. The Company allows customers to return product within a specified period of time before and
after its expiration date. Provisions for these returns are estimated based on historical return and exchange levels, and third-party data with respect
to inventory levels in the Company’s distribution channels.
License fees are comprised of initial
fees and milestone payments derived from a worldwide exclusive license agreement. Non-refundable license fees are recognized when the Company has no
further involvement or obligation to perform under the arrangement, the fee is fixed and determinable and collection of the amount is deemed probable.
Future non-refundable milestone payments receivable upon the achievement of third party performance are recognized upon the achievement of specified
milestones
F-6
when the milestone payment is substantive in nature, achievement of the milestone was not reasonably assured at the inception of the agreement and the Company has no further significant involvement or obligation to perform under the arrangement.
The Company earned royalties from its
distributor during the years ended May 31, 2005 and 2004. Royalties from the distribution agreement are recognized when the amounts are reasonably
determinable and collection is reasonably assured. In 2006 the distribution agreement was terminated and no royalties were earned during the year ended
May 31, 2006.
Cash
Equivalents
The Company considers unrestricted cash
on hand, in banks, in term deposits and in commercial paper with original maturities of three months or less as cash and cash
equivalents.
Short-Term
Investments
Lorus invests in high quality fixed
income government and corporate instruments with low credit risk.
Short-term investments, which consist
of fixed income securities with a maturity of more than three months, are recorded at their accreted value as they are held to maturity instruments.
All investments held at year end approximate fair value, mature within one year and are denominated in Canadian dollars.
Fixed
Assets
Fixed assets are recorded at cost less
accumulated depreciation and amortization. The Company records depreciation and amortization at rates which are expected to charge operations with the
cost of the assets over their estimated useful lives as follows:
Furniture and
equipment |
straight line over three to five years |
Leasehold
improvements |
straight line over the lease term |
Research and
Development
Research costs are charged to expense
as incurred. Development costs, including the cost of drugs for use in clinical trials, are expensed as incurred unless they meet the criteria under
Canadian generally accepted accounting principles for deferral and amortization. No development costs have been deferred to date.
Goodwill and Acquired Patents
and Licenses
Intangible assets with finite lives
acquired in a business combination or other transaction are amortized over their estimated useful lives which have been assessed as seven
years.
Goodwill represents the excess of the
purchase price over the fair value of net identifiable assets acquired in the GeneSense business combination. Goodwill acquired in a business
combination is tested for impairment on an annual basis and at any other time if an event occurs or circumstances change that would indicate that
impairment may exist. When the carrying value of a reporting unit’s goodwill exceeds its fair value, an impairment loss is recognized in an amount
equal to the excess.
The Company capitalized the cost of
acquired patent and license assets on the acquisitions of GeneSense and the NuChem compounds. The nature of this asset is such that it is categorized
as an intangible asset with a finite life. The carrying value of acquired research and development assets does not necessarily reflect its present or
future value. The amount recoverable is dependent upon the continued advancement of the drugs through research, clinical trials and ultimately to
commercialization. It is not possible to predict the outcome of future research and development programs.
The Company has identified no
impairment relating to goodwill and intangible assets for 2006 and 2005.
F-7
Impairment of Long-Lived
Assets
The Company periodically reviews the
useful lives and the carrying values of its long-lived assets. The Company reviews for impairment in long-lived assets whenever events or changes in
circumstances indicate that the carrying amount of the assets may not be recoverable. If the sum of the undiscounted expected future cash flows
expected to result from the use and eventual disposition of an asset is less than its carrying amount, it is considered to be impaired. An impairment
loss is measured at the amount by which the carrying amount of the asset exceeds its fair value, which is estimated as the expected future cash flows
discounted at a rate proportionate with the risks associated with the recovery of the asset.
Stock-Based
Compensation
The Company has a stock-based
compensation plan described in note 9. Prior to June 1, 2004, stock based awards were accounted for using the intrinsic method with the exception of
options with contingent vesting criteria for which the settlement method was used. On June 1, 2004, the Company adopted the fair value method of
accounting for stock-based awards to employees, officers and directors granted or modified after June 1, 2004. This method requires the Company to
expense, over the vesting period, the fair value of all employee stock-based awards granted or modified since June 1, 2002. The Company applied this
change retroactively, without restatement of prior periods. The impact to the financial statements arising from adoption of the fair value method was
an increase to the deficit and stock option balances presented in shareholders’ equity (deficiency) of $2.8 million at June 1, 2004. Stock options
and warrants awarded to non-employees are accounted for using the fair value method and expensed as the service or product is received. Consideration
paid on the exercise of stock options and warrants is credited to capital stock. The fair value of performance-based options is recognized over the
estimated period to achievement of performance conditions. Fair value is determined using the Black-Scholes option pricing model.
The Company has a deferred share unit
plan that provides directors the option of receiving payment for their services in the form of share units rather than common shares or cash. Share
units entitle the director to elect to receive, on termination of their services to the Company, an equivalent number of common shares, or the cash
equivalent of the market value of the common shares at that future date. Lorus records an expense and a liability equal to the market value of the
shares issued. The accumulated liability is adjusted for market fluctuations on a quarterly basis.
Shares issued under the Alternate
Compensation Plan are accounted for using the fair value of the common shares on the day they are granted.
Investment Tax
Credits
The Company is entitled to Canadian
federal and provincial investment tax credits, which are earned as a percentage of eligible research and development expenditures incurred in each
taxation year. Investment tax credits are accounted for as a reduction of the related expenditure for items of a current nature and a reduction of the
related asset cost for items of a long-term nature, provided that the Company has reasonable assurance that the tax credits will be
realized.
Income
Taxes
Income taxes are reported using the
asset and liability method. Under this method, future tax assets and liabilities are recorded for the future tax consequences attributable to
differences between the consolidated financial statement carrying amounts of assets and liabilities and their respective tax bases, and operating loss
and research and development expenditure carry forwards. Future tax assets and liabilities are measured using enacted or substantially enacted tax
rates expected to apply when the asset is realized or the liability is settled. The effect on future tax assets and liabilities of a change in tax
rates is recognized in income in the period that enactment or substantive enactment occurs. A valuation allowance is recorded for the portion of the
future tax assets where the realization of any value is uncertain for which management has deemed to be 100% of the assets available.
Loss Per
Share
Basic net loss per common share is
calculated by dividing the net loss by the weighted average number of common shares outstanding during the year. Diluted net loss per common share is
calculated by dividing the net loss by the sum of the weighted average number of common shares outstanding and the dilutive
F-8
common equivalent shares outstanding during the year. Common equivalent shares consist of the shares issuable upon exercise of stock options, warrants and conversion of the convertible debentures calculated using the treasury stock method. Common equivalent shares are not included in the calculation of the weighted average number of shares outstanding for diluted net loss per common share when the effect would be anti-dilutive.
Deferred Financing
Charges
Deferred financing charges, comprised
primarily of legal costs, represent costs related to the issuance of the Company’s convertible debentures. Deferred financing charges are
amortized using the effective interest rate method over the five year term of the convertible debentures.
Segmented
Information
The Company is organized and operates
as one operating segment, the research, development, and commercialization of pharmaceuticals. Substantially all of the Company’s identifiable
assets as at May 31, 2006 and 2005 are located in Canada.
Foreign Currency
Translation
Foreign currency transactions are
translated into Canadian dollars at rates prevailing on the transaction dates. Monetary assets and liabilities are translated into Canadian dollars at
the rates on the balance sheet dates. Gains or losses resulting from these transactions are accounted for in the loss for the period and are not
significant.
Use of
Estimates
The preparation of financial statements
in accordance with Canadian Generally Accepted Accounting Principles (“GAAP”) requires management to make estimates and assumptions that
affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the financial statements, and
the reported amounts of revenue and expenses during the reporting period. Actual results may differ from those estimates. Significant estimates include
the valuation of the convertible debentures, the fair value of stock options granted and warrants issued and the useful lives of capital and intangible
assets.
Measurement
Uncertainty
The preparation of financial statements
in accordance with Canadian GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and
disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenue and expenses during the
period. Actual results could differ from those estimates.
The Company has estimated the useful
lives of all depreciable assets and the recoverability of property and equipment and acquired technology using estimates of future cash flows and other
measures of fair values. Significant changes in the assumptions with respect to future business plans could result in impairment of property and
equipment or acquired technology.
Recent Canadian Accounting
Pronouncements Not Yet Adopted
Comprehensive Income and
Equity—In January 2005, the CICA released new Handbook Section 1530, Comprehensive Income, and Section 3251, Equity. Section 1530 establishes
standards for reporting comprehensive income. The section does not address issues of recognition or measurement for comprehensive income and its
components. Section 3251 establishes standards for the presentation of equity and changes in equity during the reporting period. The requirements in
this section are in addition to Section 1530.
Section 3855, Financial Instruments
— Recognition and Measurement — Section 3855 establishes standards for the recognition and measurement of all financial instruments,
provides a characteristics-based definition of a derivative instrument, provides criteria to be used to determine when a financial instrument should be
recognized, and provides criteria to be used to determine when a financial liability is considered to be extinguished.
F-9
Section 3865, Hedges —
Section 3865 establishes standards for when and how hedge accounting may be applied. Hedge accounting is optional.
These three Sections are effective for
fiscal years beginning on or after October 1, 2006. An entity adopting these Sections for a fiscal year beginning before October 1, 2006 must adopt all
the Sections simultaneously.
We have not yet determined the impact,
if any, of the adoption of these standards on our results from operations or financial position.
3. |
Changes in accounting policy |
These new accounting policies were
adopted during the year ended May 31, 2006. For the new accounting policies adopted during the year ended May 31, 2005, refer to note 2 under the
heading ‘Stock-based compensation’. There were no new accounting policies adopted during the year ended May 31, 2004.
Variable interest
entities
Effective June 1, 2005, the Company
adopted the recommendations of CICA Handbook
Accounting Guideline 15 (AcG-15),
Consolidation of Variable Interest Entities, effective for fiscal years beginning on or after November 1, 2004. Variable interest entities (VIEs) refer
to those entities that are subject to control on a basis other than ownership of voting interests. AcG-15 provides guidance for identifying VIEs and
criteria for determining which entity, if any, should consolidate them. The adoption of AcG-15 did not have an effect on the financial position,
results of operations or cash flows in the current period or the prior period presented.
Financial instruments —
disclosure and presentation
Effective June 1, 2005, the Company
adopted the amended recommendations of CICA
Handbook Section 3860, Financial
Instruments—Disclosure and Presentation, effective for fiscal years beginning on or after November 1, 2004. Section 3860 requires that certain
obligations that may be settled at the issuer’s option in cash or the equivalent value by a variable number of the issuer’s own equity
instruments be presented as a liability. The Company has determined that there is no impact on the financial statements resulting from the adoption of
the amendments to Section 3860 either in the current period or the prior period presented.
Accounting for convertible debt
instruments
On October 17, 2005 the CICA issued EIC
158, Accounting for Convertible Debt Instruments applicable to convertible debt instruments issued subsequent to the date of the EIC. EIC 158 discusses
the accounting treatment of convertible debentures in which upon conversion, the issuer is either required or has the option to satisfy all or part of
the obligation in cash. The EIC discusses various accounting issues related to this type of convertible debt. The Company has determined that there is
no impact on the financial statements resulting from the adoption of EIC 158 either in the current period or the prior period
presented.
Section 3831, Non-monetary
transactions
In June 2005, the CICA released a new
Handbook Section 3831, Non-monetary Transactions, effective for all non-monetary transactions initiated in periods beginning on or after January 1,
2006. This standard requires all non-monetary transactions to be measured at fair value unless they meet one of four very specific criteria. Commercial
substance replaces culmination of the earnings process as the test for fair value measurement. A transaction has commercial substance if it causes an
identifiable and measurable change in the economic circumstances of the entity. Commercial substance is a function of the cash flows
F-10
expected by the reporting entity. The Company has not entered into any non-monetary transactions and as such this section is not applicable.
4. |
Corporate changes |
In November 2005, as a means to
conserve cash and refocus operations, the Company scaled back some activities related to the Virulizin® technology and implemented a workforce reduction of approximately 39% or 22 employees.
In accordance with EIC 134 —
Accounting for Severance and Termination Benefits, during the period ended November 30, 2005 the Company recorded severance compensation expense for
former employees of $557 thousand. Of this expense, $468 thousand is presented in the income statement as general and administrative expense and $89
thousand as research and development expense. Accounts payable and accrued liabilities at May 31, 2006 include severance and compensation expense
liabilities relating to the Company’s November 2005 corporate changes of $154 thousand that are expected to be paid by December
2006.
5. |
Short term investments |
As at May 31 (amounts in
000’s)
| |
2006 |
|
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Less than one year maturities |
|
Greater than one year maturities |
|
Total |
|
Yield to maturity |
||||||||||||
Fixed income
government investments |
$ | 2,838 | $ | — | $ | 2,838 | 3.55–3.64% | ||||||||||||
Corporate instruments |
2,789 | — | 2,789 | 3.46–3.87% | |||||||||||||||
Balance |
$ | 5,627 | $ | — | $ | 5,627 | |||||||||||||
| |
2005 |
|
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Less than one year maturities |
|
Greater than one year maturities |
|
Total |
|
Yield to maturity |
||||||||||||
Fixed income
government investments |
$ | 3,229 | $ | — | $ | 3,229 | 2.37 | % | |||||||||||
Corporate instruments |
15,454 | — | 15,454 | 1.95–2.71% | |||||||||||||||
Balance |
$ | 18,683 | $ | — | $ | 18,683 | |||||||||||||
At May 31, 2006 and 2005, the carrying
values of short term investments approximate their quoted market values. Short term investments held at May 31, 2006 have varying maturities from one
to six months (2005 — one to six months).
F-11
6. |
Fixed assets |
As at May 31 (amounts in
000’s)
| 2006 |
|
||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Cost |
|
Accumulated Amortization |
|
Carrying Value |
||||||||||
Furniture and
equipment |
$ | 2,650 | $ | 2,136 | $ | 514 | |||||||||
Leasehold improvements |
908 | 537 | 371 | ||||||||||||
Balance |
$ | 3,558 | $ | 2,673 | $ | 885 | |||||||||
| 2005 |
|
||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Cost |
|
Accumulated Amortization |
|
Carrying Value |
||||||||||
Furniture and
equipment |
$ | 2,575 | $ | 1,517 | $ | 1,058 | |||||||||
Leasehold improvements |
908 | 385 | $ | 523 | |||||||||||
Balance |
$ | 3,483 | $ | 1,902 | $ | 1,581 | |||||||||
During the year ended May 31, 2005, a
write-down of $75,000 was taken on certain furniture and equipment whose carrying value was deemed to be unrecoverable and in excess of the estimated
future undiscounted cash flows expected from the use and residual value of the underlying assets. The impairment charge was reported in the
consolidated statements of loss and deficit in depreciation and amortization.
During the year ended May 31, 2006, a
write-down of $250,000 was taken on certain furniture and equipment whose carrying value was deemed to be unrecoverable and in excess of the estimated
fair value of the residual value of the underlying assets. The impairment charge was reported in the consolidated statements of loss and deficit in
depreciation and amortization.
7. |
Acquired patents and Licenses |
| As at May 31 (amounts in 000’s) |
|
2006 |
|
2005 |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
Cost |
$ | 12,228 | $ | 12,228 | ||||||
Accumulated amortization |
(11,573 | ) | (10,002 | ) | ||||||
Balance |
$ | 655 | $ | 2,226 | ||||||
Amortization of $1.6 million
(2005—$1.7 million, 2004—$1.7 million) has been included in the research and development expense reported in the consolidated statements of
loss and deficit.
F-12
8. |
Share capital |
(a) |
Continuity of Common Shares and Warrants |
| (amounts and units in 000’s) |
Common Shares |
Warrants |
|||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Number |
|
Amount |
|
Number |
|
Amount |
||||||||||||
Balance at May 31, 2003 |
145,285 | $ | 119,438 | — | $ | — | |||||||||||||
Share
issuance |
26,220 | 24,121 | 13,110 | 4,325 | |||||||||||||||
Exercise of
stock options |
289 | 171 | — | — | |||||||||||||||
Other |
— | (60 | ) | — | — | ||||||||||||||
Balance at May 31, 2004 |
171,794 | 143,670 | 13,110 | 4,325 | |||||||||||||||
Interest payment
(note 13) |
421 | 300 | — | — | |||||||||||||||
Issuance under
ACP (note 8 (d)) |
50 | 37 | |||||||||||||||||
Exercise of
stock options |
276 | 112 | — | — | |||||||||||||||
Convertible
debentures (note 13) |
— | — | 3,000 | 991 | |||||||||||||||
Warrants expired unexercised (note 8 (e)) |
— | — | (13,110 | ) | (4,325 | ) | |||||||||||||
Balance at May 31, 2005 |
172,541 | $ | 144,119 | 3,000 | $ | 991 | |||||||||||||
Interest payment (note 13) |
2,153 | 882 | — | — | |||||||||||||||
Balance at May 31, 2006 |
174,694 | $ | 145,001 | 3,000 | $ | 991 | |||||||||||||
(b) |
Contributed Surplus |
| As at May 31 (amounts in 000’s) |
|
2006 |
|
2005 |
|
2004 |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Beginning of
year |
$ | 6,733 | $ | 1,003 | $ | 1,003 | ||||||||
Forfeiture of
stock options |
932 | — | — | |||||||||||
Expiry of
warrants (note 8 e) |
— | 4,325 | — | |||||||||||
Expiry of compensation options (note 8 e) |
— | 1,405 | — | |||||||||||
End of year |
$ | 7,665 | $ | 6,733 | $ | 1,003 | ||||||||
(c) |
Continuity of Stock Options |
| As at May 31 (amounts in 000’s) |
|
2006 |
|
2005 |
|
2004 |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Beginning of the
year |
$ | 4,252 | $ | 2,777 | $ | — | ||||||||
Stock option
expense |
1,205 | 1,475 | — | |||||||||||
Forfeiture of stock options |
(932 | ) | — | — | ||||||||||
End of year |
$ | 4,525 | $ | 4,252 | $ | — | ||||||||
(d) |
Alternate Compensation Plans (“ACP”) |
In 2000, the Company established a
compensation plan for directors and officers, which allows the Company, in certain circumstances, to issue common shares to pay directors’ fees or
performance bonuses of officers in lieu of cash. The number of common shares reserved for issuance under this plan is 2,500,000. Since inception,
121,000 shares have been issued under this plan. For the year ended May 31, 2006, no shares were issued under this plan (2005 — 50,000, 2004
— nil).
The Company also established a deferred
share unit plan that provides directors the option of receiving payment for their services in the form of share units rather than common shares or
cash. Share units entitle the director to elect to receive, on termination of their services to the Company, an equivalent number of common shares, or
the cash equivalent of the market value of the common shares at that future date. The
F-13
share units are granted based on the market value of the common shares on the date of issue. As at May 31, 2006, 168,581 deferred share units have been issued (2005 — 99,708, 2004 — 68,183), with a cash value of $64 thousand (2005 — $71 thousand, 2004 — $57 thousand) being recorded in accrued liabilities.
(e) |
Share Issuance |
On June 11, 2003, the Company raised
gross proceeds of $32.8 million by way of a public offering of 26,220,000 units at a price of $1.25 per unit. Each unit consists of one common share
and one-half of one purchase warrant. Each whole warrant entitled the holder to purchase a common share at a price of $1.75 at any time on or before
December 10, 2004. In addition, the Company issued 1,835,400 compensation options with a fair value of $1.5 million for services in connection with the
completion of the offering. Each compensation option entitled the holder to acquire one unit for $1.27 at any time on or before December 10, 2004. The
Company incurred expenses of $4.4 million for the issuance, which include the non-cash charge of $1.5 million being the fair value of the compensation
option. The Company allocated $4.3 million of the net proceeds to the warrants, $1.4 million to the compensation option and $24.1 million to share
capital.
On December 10, 2004 the warrants and
options described above expired without being exercised. The expiry of these warrants and options had no impact on earnings or the net balance of
shareholders’ equity.
(f) |
Employee share purchase plan (“ESPP”) |
The Company’s ESPP was established
January 1, 2005. The purpose of the ESPP is to assist the Company in retaining the services of its employees, to secure and retain the services of new
employees and to provide incentives for such persons to exert maximum efforts for the success of the Company. The ESPP provides a means by which
employees of the Company and its affiliates may purchase common stock of the company at a discount through accumulated payroll deductions. Generally,
each offering is of three months’ duration with purchases occurring every month. Participants may authorize payroll deductions of up to 15% of
their base compensation for the purchase of common stock under the ESPP. For the year ended May 31, 2006, a total of 293,000 (2005 — 106,000)
common shares has been purchased under the ESPP, and Lorus has recognized an expense of $46 thousand (2005—$16 thousand) related to this plan in
the year-end financial statements.
9. |
Stock-Based Compensation |
(a) |
Stock Option Plan |
Under the Company’s stock option
plan, options may be granted to directors, officers, employees and consultants of the Company to purchase up to 25,920,797 common shares. Options are
granted at the fair market value of the common shares on the date immediately preceding the date of the grant. Options vest at various rates (immediate
to three years) and have a term of ten years. Stock option transactions for the three years ended May 31, 2006 are summarized as
follows:
F-14
| |
2006 |
|
2005 |
|
2004 |
|
|||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
Options (000’s) |
|
Weighted average exercise price |
|
Options (000’s) |
|
Weighted average exercise price |
|
Options (000’s) |
|
Weighted average exercise price |
||||||||||||||||
Outstanding at
beginning of year |
8,035 | $ | 0.96 | 6,372 | $ | 1.05 | 5,378 | $ | 1.05 | ||||||||||||||||||
Granted |
6,721 | $ | 0.58 | 3,173 | $ | 0.77 | 2,629 | $ | 1.16 | ||||||||||||||||||
Exercised |
— | — | (276 | ) | $ | 0.40 | (289 | ) | $ | 0.59 | |||||||||||||||||
Forfeited |
(4,456 | ) | $ | 0.83 | (1,234 | ) | $ | 1.05 | (1,346 | ) | $ | 1.29 | |||||||||||||||
Outstanding at end of year |
10,300 | $ | 0.70 | 8,035 | $ | 0.96 | 6,372 | $ | 1.05 | ||||||||||||||||||
Exercisable at end of year |
6,714 | $ | 0.79 | 4,728 | $ | 1.04 | 3,542 | $ | 1.01 | ||||||||||||||||||
The following table summarizes
information about stock options outstanding at May 31, 2006:
| Options outstanding |
Options exercisable |
||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Range of exercise prices |
|
Options Outstanding (000’s) |
|
Weighted average remaining contractual life (years) |
|
Weighted average exercise price |
|
Options exercisable (000’s) |
|
Weighted average exercise price |
|||||||||||||
$0.26 to
$0.49 |
3,945 | 7.79 | $ | 0.30 | 1,956 | $ | 0.31 | ||||||||||||||||
$0.50 to
$0.99 |
4,487 | 7.63 | $ | 0.76 | 3,002 | $ | 0.73 | ||||||||||||||||
$1.00 to
$1.99 |
1,580 | 6.90 | $ | 1.23 | 1,468 | $ | 1.23 | ||||||||||||||||
$2.00 to $2.50 |
288 | 4.38 | $ | 2.46 | 288 | $ | 2.46 | ||||||||||||||||
| 10,300 | 7.44 | $ | 0.70 | 6,714 | $ | 0.79 | |||||||||||||||||
For the year ended May 31, 2006
stock-based compensation expense of $1.2 million (2005 – $1.5 million) was recognized, representing the amortization applicable to the current
period of the estimated fair value of options granted since June 1, 2002.
In the year ended May 31, 2006,
employees of the Company (excluding Directors and Officers) were given the opportunity to choose between keeping 100% of their existing options at the
existing exercise price and forfeiting 50% of the options held in exchange for having the remaining 50% of the exercise price of the options re-priced
to $0.30 per share. Employees holding 2,290,000 stock options opted for re-pricing their options, resulting in the amendment of the exercise price of
1,145,000 stock options and the forfeiture of 1,145,000 stock options. This re-pricing resulted in additional compensation expense of $76 thousand
representing the incremental value conveyed to holders of the options as a result of reducing the exercise price, of which $52 thousand has been
included in the stock-based compensation expense during the year ended May 31, 2006. The balance additional compensation expense of $24 thousand will
be recognized as the amended options vest. This increased expense is offset by $113 thousand representing amounts previously expensed on unvested stock
options due to the forfeiture of 1,145,000 stock options, which was reversed from the stock-based compensation expense for the year ended May 31,
2006.
For the year ended May 31, 2005
additional stock based compensation expense of $208 thousand was recorded due to the shareholder approved amendment of the 1993 Stock Option Plan to
extend the life of
F-15
options from 5 years to 10 years. This additional expense represented the incremental value conveyed to holders of the options as a result of extending the life of the options.
For the year ended May 31, 2006, stock
option expense of $1.2 million was allocated $300 thousand to research and development and $900 thousand to general and administrative
expense.
The following assumptions were used in
the Black-Scholes option-pricing model to determine the fair value of stock options granted during the period:
| |
2006 |
|
2005 |
|
2004 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Risk-free
interest rate |
2.25-4.00 | % | 2.25-3.00 | % | 2.25-3.05 | % | ||||||||
Expected
dividend yield |
0 | % | 0 | % | 0 | % | ||||||||
Expected
volatility |
70-81 | % | 70-90 | % | 89 | % | ||||||||
Expected life of
options |
2.5–5
years |
1–5
years |
5
years |
|||||||||||
Weighted average
fair value of options options granted or modified in the year |
$ | 0.33 | $ | 0.54 | $ | 0.74 | ||||||||
The Company has assumed no forfeiture
rate as adjustments for actual forfeitures are made in the year they occur.
(b) |
Pro forma information—Stock-based compensation |
In periods prior to June 1, 2002, the
Company recognized no compensation expense when stock options were granted to employees.
For the year ended May 31, 2006, the
pro forma compensation charge for stock options granted prior to June 1, 2002 was nil (2005 – $27,000, 2004 – $551,000). These amounts have
no impact on loss per share figures.
10. |
Income Taxes |
Income tax recoveries attributable to
losses from operations differ from the amounts computed by applying the combined Canadian federal and provincial income tax rates to pre-tax income
from operations primarily as a result of the provision of a valuation allowance on net future income tax benefits.
Significant components of the
Company’s future tax assets are as follows:
| As at May 31 (amounts in 000’s) |
|
2006 |
|
2005 |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
Non-capital loss
carry forwards |
$ | 25,174 | $ | 23,081 | ||||||
Research and
development expenditures |
22,089 | 20,436 | ||||||||
Book over tax
depreciation |
1,995 | 1,529 | ||||||||
Other |
738 | 1,089 | ||||||||
Future tax
assets |
49,996 | 46,135 | ||||||||
Valuation allowance |
(49,996 | ) | (46,135 | ) | ||||||
| $ | — | $ | — | |||||||
In assessing the realizable benefit
from future tax assets, management considers whether it is more likely than not that some portion or all of the future tax assets will not be realized.
The ultimate realization of future tax assets is dependent on the generation of future taxable income during the periods in which those temporary
differences become deductible. Management considers projected future taxable income, uncertainties related to the industry in which the Company
operates and tax planning strategies in making this assessment. Due to the Company’s stage of development and operations, and uncertainties
related to the industry in which the Company operates, the tax benefit of the above amounts has been completely offset by a valuation
allowance.
F-16
The Company has undeducted research and
development expenditures, totalling $63.1 million for federal purposes and $58.1 million for provincial purposes and these can be carried forward
indefinitely. In addition the Company has non-capital loss carry forwards of $69.1 million for federal purposes and $70.1 million for provincial
purposes. To the extent that the non-capital loss carry forwards are not used, they expire as follows:
| Year of expiry (amounts in 000’s) |
|
Non-capital losses |
||||
|---|---|---|---|---|---|---|
2007 |
$ | 4,626 | ||||
2008 |
4,985 | |||||
2009 |
6,658 | |||||
2010 |
8,660 | |||||
2011 |
1,131 | |||||
2012 |
— | |||||
2013 |
— | |||||
2014 |
20,126 | |||||
2015 |
13,340 | |||||
2016 |
9,565 | |||||
| $ | 69,091 | |||||
Income Tax Rate
Reconciliation
| (amounts in 000’s) |
|
2006 |
|
2005 |
||||||
|---|---|---|---|---|---|---|---|---|---|---|
Recovery of
income taxes based on statutory rates |
$ | (6,469 | ) | $ | (7,971 | ) | ||||
Expiry of
losses |
1,252 | 780 | ||||||||
Change in
valuation allowance |
3,861 | 6,124 | ||||||||
Non deductible
accretion and stock-based compensation expense |
721 | 687 | ||||||||
Change in
enacted tax rates |
— | — | ||||||||
Other |
635 | 380 | ||||||||
| $ | — | $ | — | |||||||
Subsequent to year-end, federal
legislation was enacted to reduce tax rates applicable to future periods and extend the loss carry forward period. Had this legislation been enacted
prior to year-end the value of the future tax assets and the corresponding valuation allowance would have decreased to $45.5 million. In addition, the
losses currently expiring in 2016 would expire in 2026.
11. |
Research and Development Programs |
The Company’s cancer drug research
and development programs focus primarily on the following technology platforms:
(a) |
Immunotherapy |
This clinical approach stimulates the
body’s natural defenses against cancer. The Company’s lead immunotherapeutic drug Virulizin® completed a global Phase III clinical trial for the treatment of pancreatic cancer during 2005.
(b) |
Antisense |
Antisense drugs are genetic molecules
that inhibit the production of disease-causing proteins. GTI-2040 and GTI-2501, the Company’s lead antisense drugs, have shown preclinical
activity across a broad range of cancers and are currently in various phase I/II clinical trials.
F-17
(c) |
Small Molecules |
Anticancer activity was discovered with
an antifungal agent Clotrimazole (“CLT”). Based on the structural feature found to be responsible for the anticancer effect of CLT, chemical
analogues of CLT have been designed and tested. Our library of clotrimazole analogues has been licensed to Cyclacel Limited, as described in note
16.
Lorus scientists discovered novel low
molecular weight compounds with anticancer and anti-bacterial activity in pre-clinical investigations. Of particular interest were compounds that
inhibit the growth of human tumour cell lines, including hepatocellular carcinoma, pancreatic carcinoma, ovarian carcinoma, breast adenocarcinoma and
metastatic melanoma.
In addition to the above, Lorus has a
number of other technologies under pre-clinical development, including a tumour suppressor or gene therapy approach to inhibiting the growth of
tumours.
| Years Ended May 31 |
||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Research and Development (amounts in 000’s) |
|
2006 |
|
2005 |
|
2004 |
|
Period from inception Sept. 5, 1986 to May 31, 2006 |
||||||||||
Immunotherapy |
||||||||||||||||||
Expensed |
$ | 6,202 | $ | 11,891 | $ | 19,944 | $ | 74,958 | ||||||||||
Acquired |
— | — | — | — | ||||||||||||||
Antisense |
||||||||||||||||||
Expensed |
2,550 | 2,384 | 6,666 | 29,809 | ||||||||||||||
Acquired |
— | — | — | 11,000 | ||||||||||||||
Small
Molecules |
||||||||||||||||||
Expensed |
1,485 | 119 | 175 | 5,708 | ||||||||||||||
Acquired |
— | — | — | 1,228 | ||||||||||||||
Total expensed |
$ | 10,237 | $ | 14,394 | $ | 26,785 | $ | 110,475 | ||||||||||
Total acquired |
$ | — | $ | — | $ | — | $ | 12,228 | ||||||||||
Amortization of the acquired patents
and licenses is included in the ‘Expensed’ line of the table.
12. |
Supplementary cash flow information |
Changes in non-cash working capital
balances for each of the periods ended are summarized as follows:
| Years ended May 31 (amounts in 000’s) |
|
2006 |
|
2005 |
|
2004 |
|
Period from inception Sept. 5, 1986 to May 31, 2006 |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
(Increase)
decrease |
||||||||||||||||||
Prepaid
expenses and other assets |
$ | 611 | $ | 571 | $ | (593 | ) | $ | 61 | |||||||||
Increase
(decrease) |
||||||||||||||||||
Accounts
payable |
(514 | ) | (1,360 | ) | 1,111 | (689 | ) | |||||||||||
Accrued liabilities |
(559 | ) | (377 | ) | (647 | ) | 2,220 | |||||||||||
| $ | (462 | ) | $ | (1,166 | ) | $ | (129 | ) | $ | 1,592 | ||||||||
During the year ended May 31, 2006, the
Company received interest of $627 thousand (2005 – $679 thousand, 2004 — $1.2 million).
F-18
13. |
Convertible debentures |
On October 6, 2004, the Company entered
into a Subscription Agreement (the “Agreement”) to issue an aggregate of $15.0 million of secured convertible debentures (the
“debentures”). The debentures are secured by a first charge over all of the assets of the Company.
The Company received $4.4 million on
October 6, 2004 (representing a $5.0 million debenture less an investor fee representing 4% of the $15.0 million to be received under the Agreement),
and $5.0 million on each of January 14 and April 15, 2005. All debentures issued under this Agreement are due on October 6, 2009 and are subject to
interest payable monthly at a rate of prime + 1% until such time as the Company’s share price reaches $1.75 for 60 consecutive trading days, at
which time, interest will no longer be charged. Interest is payable in common shares of Lorus until Lorus’ shares trade at a price of $1.00 or
more after which interest will be payable in cash or common shares at the option of the debenture holder. Common shares issued in payment of interest
will be issued at a price equal to the weighted average trading price of such shares for the ten trading days immediately preceding their issue in
respect of each interest payment. For the year ended May 31, 2006, the Company has issued 2,153,000 (2005 – 421 thousand) shares in settlement of
$882 thousand (2005 – $300 thousand) in interest.
The $15.0 million principal amount of
debentures issued on October 6, 2004, January 14 and April 15, 2005 is convertible at the holder’s option at any time into common shares of the
Company with a conversion price per share of $1.00.
With the issuance of each $5.0 million
debenture, the Company issued to the debt holder from escrow 1 million purchase warrants expiring October 6, 2009 to buy common shares of the Company
at a price per share equal to $1.00.
The convertible debentures contain both
a liability and an equity element, represented by the conversion option, and therefore, under Canadian GAAP these two elements must be split and
classified separately as debt and equity. In addition, as noted above, the debenture holder received 1 million purchase warrants on the issuance of
each tranche of convertible debt. The Company allocated the total proceeds received from the issuance of the convertible debentures to these three
elements based on their relative fair values. The fair value of the purchase warrants has been determined based on an option-pricing model. The fair
value of the debt has been based on the discounted cash flows using an estimated cost of borrowing of 15% to represent an estimate of what the Company
may borrow secured debt without a conversion option or purchase warrant. The convertible debentures conversion option was valued using a trinomial
model. The resulting allocation based on relative fair values resulted in the allocation of $9.8 million to the debt instrument, $4.1 million to the
conversion option and $1.1 million to the purchase warrants. The financing fees totalling $1.1 million related to the issuance of the convertible
debentures have been allocated pro rata between deferred financing charges of $652 thousand, against the equity portion of the convertible debentures
of $322 thousand and against the purchase warrants of $87 thousand. This allocation resulted in net amounts allocated to the equity portion of the
convertible debentures and warrants of $3.8 million and $991 thousand respectively. The financing charges are being amortized over the five-year life
of the convertible debentures agreement. For the year ended May 31, 2006, the Company has recognized $87 thousand (2005 – $84 thousand) in
amortization expense. This amortization expense has reduced the value of the deferred financing charges to $481 thousand at May 31, 2006
(2005—$568 thousand).
Each reporting period, the Company is
required to accrete the carrying value of the convertible debentures such that at maturity on October 6, 2009, the carrying value of the debentures
will be their face value of $15.0 million. For the year ended May 31, 2006, the Company has recognized $790 thousand (2005 – $426 thousand) in
accretion expense. This accretion expense has increased the carrying value of the convertible debentures from $9.8 million to $11.0 million at May 31,
2006 (2005 – $10.2 million).
The lender has the option to demand
repayment in the event of default, including the failure to maintain certain subjective covenants, representations and warranties. Management assesses
on a quarterly basis whether or not events during the quarter could be considered an event of default. This assessment was
F-19
performed and management believes that there has not been an event of default and that, at May 31, 2006, the term of the debt remains unchanged.
At the end of the second quarter of
fiscal 2006, subject to the completion of a tax assisted financing transaction and based on mutually agreed upon terms with the holder, it had been the
Company’s intent to repay the debentures by October 1, 2006. However, during the third quarter of fiscal 2006, the conditions precedent of the
proposed tax assisted financing were not met and as such the transaction did not close and the Company’s agreement with the debenture holder to
repay the debentures was terminated. As such the debentures have been recorded as a long-term liability with the original due date of October 6, 2009.
The investor paid Lorus $100 thousand to help cover the costs incurred as part of the incomplete transaction. This $100 thousand has been recorded as a
reduction in professional fee expense.
14. |
Commitments and Guarantees |
(a) |
Operating lease commitments |
The Company has entered into operating
leases for premises under which it is obligated to make minimum annual payments of approximately $139 thousand in 2007, $118 thousand in 2008 and $8
thousand in 2009.
During the year ended May 31, 2006,
operating lease expenses were $130 thousand (2005 – $136 thousand, 2004 – $141 thousand).
(b) |
Other contractual commitments |
In December 1997, the Company acquired
certain patent rights and a sub-license to develop and commercialize the anticancer application of certain compounds in exchange for:
(i) |
A 20% share interest in NuChem; |
(ii) |
A payment of US $350 thousand in shares of Lorus, and |
(iii) |
Up to US $3.5 million in cash. |
To date, the Company has made cash
payments of US $500 thousand. The remaining balance of up to US $3.0 million remains payable upon the achievement of certain milestones based on the
commencement and completion of clinical trials. Additional amounts paid will be classified as acquired patents and licenses and will be amortized over
the estimated useful life of the licensed asset.
The Company holds an exclusive
world-wide license from the University of Manitoba (the “University”) and Cancer Care Manitoba (“CCM”) to certain patent rights to
develop and sublicense certain oligonucleotide technologies. In consideration for the exclusive license of the patent rights, the University and CCM
are entitled to an aggregate of 1.67% of the net sales received by the Company from the sale of products or processes derived from the patent rights
and 1.67% of all monies received by the Company from sublicenses of the patent rights. Any and all improvements to any of the patent rights derived in
whole or in part by the Company after the date of the license agreement, being June 20, 1997, are not included within the scope of the agreement and do
not trigger any payment of royalties. To date, the Company has not paid any royalties pursuant to the license agreement.
(c) |
Guarantees |
The Company entered into various
contracts, whereby contractors perform certain services for the Company. The Company indemnifies the contractors against costs, charges and expenses in
respect of legal actions or proceedings against the contractors in their capacity of servicing the Company. The maximum amounts payable from these
guarantees cannot be reasonably estimated. Historically, the Company has not made significant payments related to these guarantees.
F-20
15. |
Financial Instruments |
The carrying values of cash and cash
equivalents, short-term investments, amounts receivable, other assets, accounts payable and accrued liabilities approximate their fair values due to
the short-term nature of these instruments.
Fair value estimates are made at a
specific point in time, based on relevant market information and information about the financial instrument. These estimates are subjective in nature
and involve uncertainties and matters of significant judgment and, therefore, cannot be determined with precision. Changes in assumptions could
significantly affect the estimates.
Financial instruments potentially
exposing the Company to a concentration of credit risk consist principally of cash equivalents and short-term investments. The Company mitigates this
risk by investing in high grade fixed income securities.
The Company is exposed to interest rate
risk due to the convertible debentures that require interest payments at a variable rate of interest.
The fair value of the convertible
debentures at May 31, 2006 is $13.8 million.
16. |
Revenue |
During the year ended May 31, 2004, the
Company recorded license revenue of $546 thousand in connection with a worldwide exclusive license agreement entered into with Cyclacel Limited in the
United Kingdom for the out-licensing of the Company’s small molecule program. Additional license fees of up to $11.6 million may be earned if
Cyclacel achieves certain defined research and development milestones. No such milestones were achieved during the year ended May 31,
2006.
17. |
Canada and United States Accounting Policy Differences |
These consolidated financial statements
have been prepared in accordance with Canadian GAAP which differ in some respects from accounting principles generally accepted in the United States
(“US GAAP”). The following reconciliation identifies material differences in the Company’s consolidated statement of operations and
deficit and consolidated balance sheets.
(a) |
Consolidated statements of loss and deficit |
| Years ended May 31, |
|||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |
2006 |
|
2005 |
|
2004 |
||||||||||
Loss per
Canadian GAAP |
(17,909 | ) | (22,062 | ) | (30,301 | ) | |||||||||
Accretion of
convertible debentures (i) |
480 | 329 | — | ||||||||||||
Amortization of
debt issue costs (i) |
(108 | ) | (40 | ) | — | ||||||||||
Stock
compensation expense (ii) |
1,149 | 1,475 | — | ||||||||||||
Loss and comprehensive loss per US GAAP |
(16,388 | ) | (20,298 | ) | (30,301 | ) | |||||||||
Basic and diluted loss per share per US GAAP |
$ | (0.09 | ) | $ | (0.12 | ) | $ | (0.18 | ) | ||||||
Under US GAAP, the number of weighted
average common shares outstanding for basic and diluted loss per share are the same as under Canadian GAAP.
F-21
(b) |
Consolidated balance sheets: |
| May 31, 2006 |
|
||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adjustments |
|
||||||||||||||||||
| |
Canadian GAAP |
|
Convertible Debentures (i) |
|
Stock Options (ii) |
|
US GAAP |
||||||||||||
Deferred
financing charges |
481 | 164 | — | 645 | |||||||||||||||
Secured
convertible debentures |
(11,002 | ) | (3,260 | ) | — | (14,262 | ) | ||||||||||||
Equity
portion of secured convertible debentures |
(3,814 | ) | 3,814 | — | — | ||||||||||||||
Stock
options |
(4,525 | ) | — | 4,525 | — | ||||||||||||||
Contributed
surplus/Additional paid in capital (APIC) |
(7,665 | ) | (1,048 | ) | 876 | (7,837 | ) | ||||||||||||
Warrants |
(991 | ) | 991 | — | — | ||||||||||||||
Deficit
accumulated during the development stage |
164,552 | (661 | ) | (5,401 | ) | 158,490 | |||||||||||||
| May 31, 2005 |
|
||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Canadian GAAP |
|
Convertible Debentures (i) |
|
Stock Options (ii) |
|
US GAAP |
|||||||||||||
Deferred
financing charges |
568 | 272 | — | 840 | |||||||||||||||
Secured
convertible debenture |
(10,212 | ) | (3,740 | ) | — | (13,952 | ) | ||||||||||||
Equity
portion of secured convertible debentures |
(3,814 | ) | 3,814 | — | — | ||||||||||||||
Stock
options |
(4,252 | ) | — | 4,252 | — | ||||||||||||||
Contributed
surplus/Additional paid in capital (APIC) |
(6,733 | ) | (1,048 | ) | — | (7,781 | ) | ||||||||||||
Warrants |
(991 | ) | 991 | — | — | ||||||||||||||
Deficit
accumulated during the development stage |
146,643 | (289 | ) | (4,252 | ) | 142,102 | |||||||||||||
(i) |
Convertible debentures |
Under Canadian GAAP, the conversion
option embedded in the convertible debentures is presented separately as a component of shareholders’ equity. Under U.S. GAAP, the embedded
conversion option is not subject to bifurcation and is thus presented as a liability along with the balance of the convertible debentures. Under U.S.
GAAP, Emerging Issues Task Force No.00-19 and APB Opinion No. 14, the fair value of warrants issued in connection with the convertible debentures
financing would be recorded as a reduction to the proceeds from the issuance of convertible debentures, with the offset to additional paid-in capital.
The warrants have been presented as a separate component of shareholders’ equity for Canadian GAAP purposes. Under U.S. GAAP the Company has
allocated the total proceeds received from the issuance of the convertible debentures to the debt and warrant portions based on their relative fair
values. The fair value of the purchase warrants has been determined based on an option-pricing model. The resulting allocation based on relative fair
values resulted in the allocation of $13.9 million to the debt instrument and $1.1 million to the purchase warrants. The financing fees totalling $1.1
million related to the issuance of the convertible debentures have been allocated pro rata between deferred financing charges of $964 thousand and
against the purchase warrants of $97 thousand. This allocation resulted in the net amount allocated to the warrants of $1.0 million. The financing
charges are being amortized over the five-year life of the convertible debentures agreement.
F-22
Each reporting period, the Company is
required to accrete the carrying value of the convertible debentures such that at maturity on October 6, 2009, the carrying value of the debentures
will be their face value of $15.0 million. To date, the Company has recognized $407 thousand in accretion expense. This accretion expense has increased
the value of the convertible debentures from $13.9 million to $14.3 million at May 31, 2006.
(ii) |
Stock-based compensation |
Effective June 1, 2004, the Company
adopted the fair value based method of accounting for employee stock options granted on or after June 1, 2002, retroactively without restatement as
allowed under the transitional provisions of CICA Handbook Section 3870. As a result, the opening balances of deficit and stock options were increased
by $2.8 million at June 1, 2004. During 2006, the Company recorded stock compensation expense in the consolidated financial statements, representing
the amortization applicable to the current year at the estimated fair value of stock options granted since June 1, 2002.
During 2006, the Company recorded stock
compensation expense of $1.2 million (2005 – $1.5 million) in the consolidated statement of operations, representing the amortization applicable
to the current year at the estimated fair value of options granted since June 1, 2002; and an offsetting adjustment to stock options of $1.2 million in
the consolidated balance sheets. No similar adjustments are required under U.S. GAAP as the Company has elected to continue measuring compensation
expense, as permitted under SFAS No. 123, using the intrinsic value based method of accounting for stock options. Under this method, compensation is
the excess, if any, of the quoted market value of the stock at the date of the grant over the amount an employee must pay to acquire the stock.
Election of this method requires pro-forma disclosure of compensation expense as if the fair value method has been applied for awards granted in fiscal
periods after December 15, 1994.
The Company grants performance based
stock options as a compensation tool. Under Canadian GAAP, the accounting treatment of these options is consistent with all other employee stock
options. Under US GAAP, the option is treated as a variable award and is revalued, using the intrinsic value method of accounting, at the end of each
reporting period until the final measurement date. At each reporting date, compensation cost is measured based on an estimate of the number of options
that will vest considering the performance criteria and the difference between the market price of the underlying stock and the exercise price at such
dates. The compensation cost is being recognized over the estimated performance period. For the year ended May 31, 2006 the Company recorded stock
based compensation expense of $20 thousand under U.S. GAAP for performance based options.
During 2006, employees of the Company
(excluding Directors and Officers) were given the opportunity to choose between keeping 100% of their existing options at the existing exercise price
and forfeiting 50% of the options held in exchange for having the remaining 50% of the exercise price of the options re-priced to $0.30 per share.
Employees holding 2,290,000 stock options opted for re-pricing their options, resulting in the amendment of the exercise price of 1,145,000 stock
options and the forfeiture of 1,145,000 stock options. Under Canadian GAAP the accounting treatment of these options requires that any incremental
value resulting from the amendment be determined and recognized over the remaining vesting period. Under US GAAP, the amended options are treated as a
variable award and are revalued, using the intrinsic value method of accounting at the end of each reporting period until the date the options are
exercised, forfeited or expired unexercised. The Company recorded stock-based compensation of $36 thousand under US GAAP related to these amended stock
options.
Prior to the adoption of CICA Handbook
Section 3870, Lorus accounted for performance based stock options using the intrinsic value method, and a recovery of $43,000 was included in net
income in 2004 related to these options.
F-23
The table below presents the pro-forma
disclosures required under U.S. GAAP:
| |
2006 |
|
2005 |
|
2004 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Net loss to
common shareholders — US GAAP |
(16,388 | ) | (20,298 | ) | (30,301 | ) | ||||||||
Compensation expense under SFAS 123 |
(1,149 | ) | (1,475 | ) | (1,623 | ) | ||||||||
Pro-forma net loss to common shareholders — US GAAP |
(17,537 | ) | (21,773 | ) | (31,924 | ) | ||||||||
Pro-forma basic and diluted loss per share — US GAAP |
(0.10 | ) | (0.13 | ) | (0.19 | ) | ||||||||
(c) |
Consolidated statements of cash flows |
There are no differences between
Canadian and US GAAP that impact the consolidated statements of cash flows.
(d) |
Income taxes |
Under Canadian GAAP, investment tax
credits and other research and development credits are deducted from research and development expense for items of a current nature, and deducted from
property and equipment for items of a capital nature. Under US GAAP, these tax credits would be reclassified as a reduction of income tax expense. The
impact would be higher research and development expense and an income tax recovery of $205 thousand for the year ended May 31, 2006 (2005 – $400
thousand, 2004 – $180 thousand) with no net impact to net income or earnings per share.
(e) |
New accounting pronouncements not yet adopted |
(i) |
In December 2004, the FASB revised SFAS No. 123 to require companies to recognize n the income statement the grant-date fair value of stock options and other equity based compensation issued to employees, but expressed no preference for a type of valuation model (SFAS 123R). The way an award is classified will affect the measurement of compensation cost. Liability-classified awards are re-measured to fair value at each balance sheet date until the award is settled. Equity-classified awards are measured at grant-date fair value and the grant-date fair value is recognized over the requisite service period. Such awards are not subsequently re-measured. |
| In April 2005, the staff of the Securities and Exchange Commission issued Staff Accounting Bulletin No. 107 (SAB 107) to provide additional guidance regarding the application of SFAS 123R. SAB 107 permits registrants to choose an appropriate valuation technique or model to estimate the fair value of share options, assuming consistent application, and provides guidance for the development of assumptions used in the valuation process. Based upon SEC rules issued in April 2005, SFAS 123R is effective for fiscal years that begin after June 15, 2005 and will be adopted by the Company effective June 1, 2006. Additionally, SAB 107 discusses disclosures to be made under “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in registrants’ periodic reports. The Company has not yet determined the effect of this new standard on its consolidated financial position and results of operations. |
(ii) |
In December 2004, FASB issued Financial Accounting Standard 153: Exchanges of Nonmonetary Assets as an amendment of APB Opinion No. 29. The guidance in APB Opinion No. 29, Accounting for Nonmonetary Transactions, is based on the principle that exchanges of nonmonetary assets should be measured based on the fair value of the assets exchanged. The guidance in that Opinion, however, included certain exceptions to that principle. This Statement amends Opinion 29 to eliminate the exception for nonmonetary exchanges of similar productive assets and replaces it with a general exception for exchanges of nonmonetary assets that do not have commercial substance. Nonmonetary exchange has commercial substance if the future cash flows of the entity are expected to |
F-24
| change significantly as a result of the exchange. This statement is effective for years beginning after June 15, 2005. The Company has not entered into any non-monetary transactions and as such this section is not applicable. |
(iii) |
In May 2005, the FASB issued SFAS No. 154, Accounting Changes and Error Corrections (SFAS 154), which replaces APB No. 20, Accounting Change and SFAS No. 3, Reporting Accounting Changes in Interim Financial Statements — An Amendment of APB Opinion No. 28. SFAS 154 provides guidance on the accounting for and reporting of accounting changes and error corrections. It establishes retrospective application, on the latest practicable date, as the required method for reporting a change in accounting principle and the reporting of a correction of an error. SFAS 154 is effective for accounting changes and corrections of errors made in fiscal years beginning after December 15, 2005. Management believes that the adoption of this statement will not have a material effect on the Company’s consolidated financial condition or results of operations. |
(f) |
Consolidated statement of shareholders equity (deficiency) for the period from June 1, 1998 to May 31, 2006: |
| |
Number of Shares (000’s) |
|
Amount |
|
Contributed Surplus/APIC |
|
Deficit |
|
Total |
|
||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Balance May 31, 1998 | 36,785 | $ | 37,180 | $ | 667 | $ | (32,946 | ) | $ | 4,901 | ||||||||||||||||
| Exercise of special warrants | 5,333 | 1,004 | (1,217 | ) | — | (213 | ) | |||||||||||||||||||
| Exercise of stock options | 46 | 48 | — | — | 48 | |||||||||||||||||||||
| Issue of warrants | — | — | 1,217 | — | 1,217 | |||||||||||||||||||||
| Issue of special warrants | — | — | 213 | — | 213 | |||||||||||||||||||||
| Other issuances | 583 | 379 | — | — | 379 | |||||||||||||||||||||
| Deficit | — | — | — | (4,623 | ) | (4,623 | ) | |||||||||||||||||||
| Balance May 31, 1999 | 42,747 | $ | 38,611 | $ | 880 | $ | (37,569 | ) | $ | 1,922 | ||||||||||||||||
| Exercise of warrants | 12,591 | 7,546 | (534 | ) | — | 7,012 | ||||||||||||||||||||
| Issuance of special and purchase warrants | — | — | 8,853 | — | 8,853 | |||||||||||||||||||||
| Issuance of public offering | 15,333 | 41,952 | 659 | — | 42,611 | |||||||||||||||||||||
| Issued on acquisition | 36,050 | 14,000 | — | — | 14,000 | |||||||||||||||||||||
| Exercise of units | 893 | 1,821 | (321 | ) | — | 1,500 | ||||||||||||||||||||
| Issuance under alternate compensation plan | 18 | 15 | — | — | 15 | |||||||||||||||||||||
| Exercise of special warrants | 30,303 | 8,438 | (8,438 | ) | — | — | ||||||||||||||||||||
| Exercise of stock options | 1,730 | 1,113 | — | — | 1,113 | |||||||||||||||||||||
| Stock based compensation | — | 869 | — | — | 869 | |||||||||||||||||||||
| Deficit | — | — | — | (8,599 | ) | (8,599 | ) | |||||||||||||||||||
| Balance May 31, 2000 | 139,665 | $ | 114,365 | $ | 1,099 | $ | (46,168 | ) | $ | 69,296 | ||||||||||||||||
| Exercise of warrants | 168 | 93 | (25 | ) | — | 68 | ||||||||||||||||||||
| Issuance under alternate compensation plan | 28 | 49 | — | — | 49 | |||||||||||||||||||||
| Exercise of stock options | 2,550 | 1,866 | — | — | 1,866 | |||||||||||||||||||||
| Stock based compensation | — | 351 | — | — | 351 | |||||||||||||||||||||
| Deficit | — | 82 | — | (15,131 | ) | (15,131 | ) | |||||||||||||||||||
| Balance May 31, 2001 | 142,411 | $ | 116,806 | $ | 1,074 | $ | (61,381 | ) | $ | 56,499 | ||||||||||||||||
F-25
| |
Number of Shares (000’s) |
|
Amount |
|
Contributed Surplus/APIC |
|
Deficit |
|
Total |
|
||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Exercise of compensation warrants | 476 | 265 | (71 | ) | — | 194 | ||||||||||||||||||||
| Exercise of stock options | 1,525 | 1,194 | — | — | 1,194 | |||||||||||||||||||||
| Stock based compensation | — | (100 | ) | — | — | (100 | ) | |||||||||||||||||||
| Deficit | — | — | — | (13,488 | ) | (13,488 | ) | |||||||||||||||||||
| Balance May 31, 2002 | 144,412 | $ | 118,165 | $ | 1,003 | $ | (74,869 | ) | $ | 44,299 | ||||||||||||||||
| Exercise of stock options | 873 | 715 | — | — | 715 | |||||||||||||||||||||
| Stock based compensation | — | 558 | — | — | 558 | |||||||||||||||||||||
| Deficit | — | — | — | (16,634 | ) | (16,634 | ) | |||||||||||||||||||
| Balance May 31, 2003 | 145,285 | $ | 119,438 | $ | 1,003 | $ | (91,503 | ) | $ | 28,938 | ||||||||||||||||
| Share issuance | 26,220 | 24,121 | 4,325 | — | 28,446 | |||||||||||||||||||||
| Exercise of stock options | 289 | 171 | — | — | 171 | |||||||||||||||||||||
| Stock based compensation | — | (88 | ) | — | — | (88 | ) | |||||||||||||||||||
| Other issuances | — | 28 | — | — | 28 | |||||||||||||||||||||
| Deficit | — | — | — | (30,301 | ) | (30,301 | ) | |||||||||||||||||||
| Balance May 31, 2004 | 171,794 | $ | 143,670 | $ | 5,328 | $ | (121,804 | ) | $ | 27,194 | ||||||||||||||||
| Interest payment | 421 | 300 | — | — | 300 | |||||||||||||||||||||
| Exercise of stock options | 276 | 112 | — | — | 112 | |||||||||||||||||||||
| Expiry of compensation options | — | — | 1,405 | — | 1,405 | |||||||||||||||||||||
| Issuance under alternate compensation plan | 50 | 37 | — | — | 37 | |||||||||||||||||||||
| Issuance of warrants | — | — | 1,048 | — | 1,048 | |||||||||||||||||||||
| Deficit | — | — | — | (20,298 | ) | (20,298 | ) | |||||||||||||||||||
| Balance May 31, 2005 | 172,541 | $ | 144,119 | $ | 7,781 | $ | (142,102 | ) | $ | 9,798 | ||||||||||||||||
| Interest payment | 2,153 | 882 | — | — | 882 | |||||||||||||||||||||
| Stock-based compensation | — | — | 56 | — | 56 | |||||||||||||||||||||
| Deficit | — | — | — | (16,388 | ) | (16,388 | ) | |||||||||||||||||||
| Balance May 31, 2006 | 174,694 | $ | 145,001 | $ | 7,837 | $ | (158,490 | ) | $ | (5,652 | ) | |||||||||||||||
18. |
Comparative Figures |
Certain of the comparative figures have
been reclassified to conform to the current year’s method of presentation.
19. |
Subsequent Events |
(a) |
On July 13, 2006 we entered into an agreement with HighTech Beteiligungen GmbH & Co. KG (“HighTech”) to issue 28.8 million common shares at $0.36 per share for gross proceeds of $10.4 million. The subscription price represented a premium of 7.5% over the closing price of the common shares on the Toronto Stock Exchange on July 13, 2006. The transaction closed on August 30, 2006. In connection with the transaction, HighTech received demand registration rights that will enable HighTech to request the registration or qualification of the common shares for resale in the United States and Canada, subject to certain restrictions. These demand registration rights will expire on June 30, 2012. In addition, HighTech has the right to nominate one nominee to the board of directors of Lorus or, if it does not have a nominee, it will have the right to appoint an observer to the board. Upon completion of the transaction, HighTech will hold approximately 14% of the issued and outstanding common shares of Lorus Therapeutics Inc. |
F-26
(b) |
On July 24, 2006 Lorus entered into an agreement with Technifund Inc. to issue on a private placement basis, 5 million common shares at $0.36 per share for gross proceeds of $1.8 million. The transaction closed on August 31, 2006. |
(c) |
On September 19, 2006 the Company announced that Dr. Jim A Wright would step down as the President and Chief Executive Officer of Lorus effective September 21, 2006. The departure of Dr. Wright resulted in a liability based on a mutual separation agreement executed subsequent to the quarter end of approximately $500 thousand. The amount is expected to be paid by the end of the third quarter 2007. |
F-27